You could read whats below or look at the charts to answer: How does integer ratios relate to ions, polyatomic and monoatomic ions, and lewis dot structures?
You could read whats below or look at the charts to answer: How does integer ratios relate to ions, polyatomic and monoatomic ions, and lewis dot structures?
Chemistry In Focus
6th Edition
ISBN:9781305084476
Author:Tro, Nivaldo J., Neu, Don.
Publisher:Tro, Nivaldo J., Neu, Don.
Chapter5: Chemical Bonding
Section: Chapter Questions
Problem 4SC
Related questions
Question
100%
You could read whats below or look at the charts to answer: How does integer ratios relate to ions, polyatomic and monoatomic ions, and lewis dot structures?
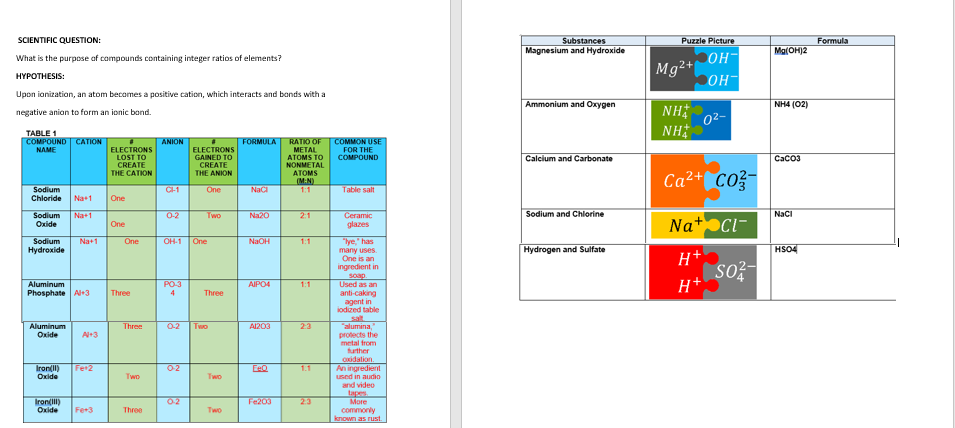
Transcribed Image Text:SCIENTIFIC QUESTION:
What is the purpose of compounds containing integer ratios of elements?
HYPOTHESIS:
Upon ionization, an atom becomes a positive cation, which interacts and bonds with a
negative anion to form an ionic bond.
TABLE 1
COMPOUND CATION
NAME
Sodium
Chloride Na+1
Sodium Na+1
Oxide
Sodium
Hydroxide
Aluminum
Phosphate Al+3
Aluminum
Oxide
Na+1
Iron(III)
Oxide
Al+3
Iron(11) Fe+2
Oxide
Fe+3
ELECTRONS
LOST TO
CREATE
THE CATION
One
One
One
Three
Three
Two
Three
ANION
CH-1
0-2
PO-3
4
ELECTRONS
GAINED TO
CREATE
THE ANION
OH-1 One
0-2
One
0-2
Two
0-2 Two
Three
Two
Two
FORMULA
NaCl
Na20
NaOH
AIPO4
A1203
FeO
Fe203
RATIO OF
METAL
ATOMS TO
NONMETAL
ATOMS
(MAN)
1:1
2:1
1:1
1:1
2:3
1.1
2.3
COMMON USE
FOR THE
COMPOUND
Table salt
Ceramic
glazes
"lye," has
many uses.
One is an
ingredient in
soap.
Used as an
anti-caking
agent in
iodized table
salt
"alumina,
protects the
metal from
further
oxidation
An ingredient
used in audio
and video
More
commonly
known as rust.
Substances
Magnesium and Hydroxide
Ammonium and Oxygen
Calcium and Carbonate
Sodium and Chlorine
Hydrogen and Sulfate
Puzzle Picture
он-
OH-
Mg²+
NH
NH
0²-
Ca²+ CO²-
Na+ Cl
H+
H+
so²-
Mg(OH)2
NHA (O2)
CaCO3
NaCl
HSO4
Formula
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry In Focus
Chemistry
ISBN:
9781305084476
Author:
Tro, Nivaldo J., Neu, Don.
Publisher:
Cengage Learning

Chemistry In Focus
Chemistry
ISBN:
9781305084476
Author:
Tro, Nivaldo J., Neu, Don.
Publisher:
Cengage Learning