You were asked to determine the acid content of vinegar and soda samples. You performed a simple acid-base titration using a solution of NaOH as the titrant with phenolphthalein as the indicator. To determine the concentration of the NaOH solution, you first performed standardization using KHP (FW: 204.22 g/mol, 99.94% purity). For analyzing the samples, he separately measured a volume of vinegar and soda then transferred them to a flask. Complete the table using the provided data. a) b) c) Standardization of NaOH Solution Trial 1 0.1012 Weight of KHP, g Initial volume of NaOH, mL Final volume of NaOH, mL Molarity of NaOH Average Molarity of NaOH Volume of vinegar, mL Initial volume of NaOH, mL Final volume of NaOH, mL Analysis of acetic acid in a vinegar sample Trial 1 Trial 2 1.500 0.500 14.98 26.85 24.84 30.15 Molarity of acetic acid Average Molarity of acetic acid 0.00 4.95 Trial 2 0.1004 5.00 9.99 Volume of soda, mL Initial volume of NaOH, mL Final volume of NaOH, mL Molarity of carbonic acid Average Molarity of carbonic acid Analysis of carbonic acid in a soda sample Trial 1 Trial 2 20.00 20.20 24.48 18.00 24.69 28.50 Trial 3 0.09987 10.01 14.93 Trial 3 1.000 30.87 37.50 Trial 3 20.00 29.48 33.87
You were asked to determine the acid content of vinegar and soda samples. You performed a simple acid-base titration using a solution of NaOH as the titrant with phenolphthalein as the indicator. To determine the concentration of the NaOH solution, you first performed standardization using KHP (FW: 204.22 g/mol, 99.94% purity). For analyzing the samples, he separately measured a volume of vinegar and soda then transferred them to a flask. Complete the table using the provided data. a) b) c) Standardization of NaOH Solution Trial 1 0.1012 Weight of KHP, g Initial volume of NaOH, mL Final volume of NaOH, mL Molarity of NaOH Average Molarity of NaOH Volume of vinegar, mL Initial volume of NaOH, mL Final volume of NaOH, mL Analysis of acetic acid in a vinegar sample Trial 1 Trial 2 1.500 0.500 14.98 26.85 24.84 30.15 Molarity of acetic acid Average Molarity of acetic acid 0.00 4.95 Trial 2 0.1004 5.00 9.99 Volume of soda, mL Initial volume of NaOH, mL Final volume of NaOH, mL Molarity of carbonic acid Average Molarity of carbonic acid Analysis of carbonic acid in a soda sample Trial 1 Trial 2 20.00 20.20 24.48 18.00 24.69 28.50 Trial 3 0.09987 10.01 14.93 Trial 3 1.000 30.87 37.50 Trial 3 20.00 29.48 33.87
Chapter15: Acid-base Equilibria
Section: Chapter Questions
Problem 13Q: An acid is titrated with NaOH. The following beakers are illustrations of the contents of the beaker...
Related questions
Question
SKIP IF YOU ALREADY DID THIS OR ELSE DOWNVOTE
ANSWER COMPLETELY AND I WILL UPVOTE
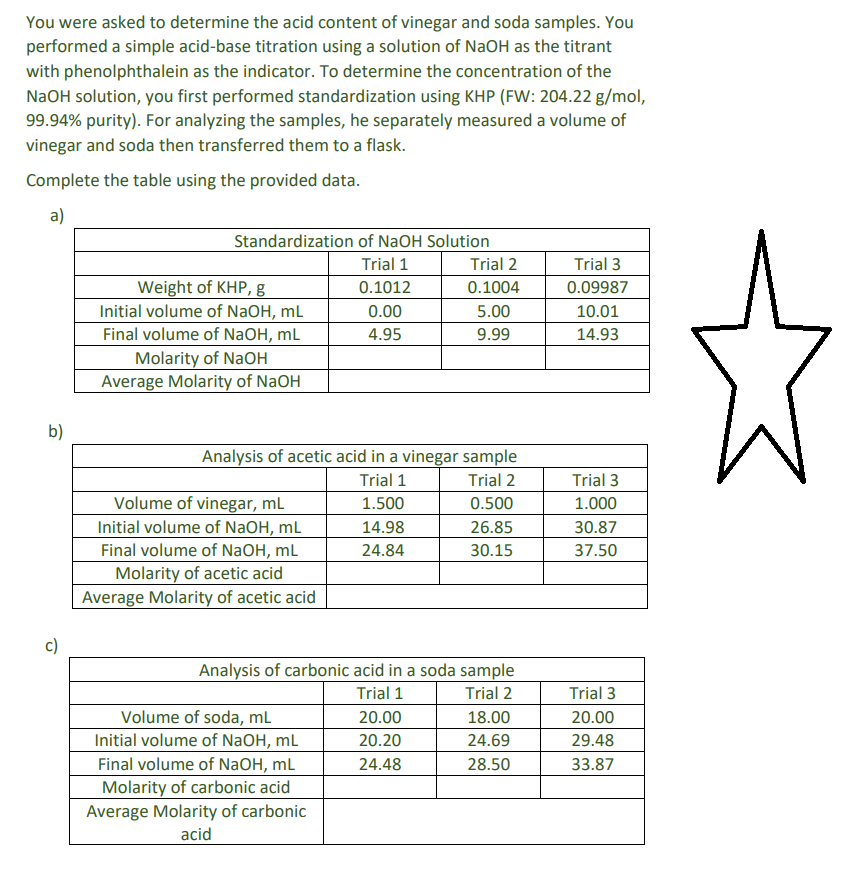
Transcribed Image Text:You were asked to determine the acid content of vinegar and soda samples. You
performed a simple acid-base titration using a solution of NaOH as the titrant
with phenolphthalein as the indicator. To determine the concentration of the
NaOH solution, you first performed standardization using KHP (FW: 204.22 g/mol,
99.94% purity). For analyzing the samples, he separately measured a volume of
vinegar and soda then transferred them to a flask.
Complete the table using the provided data.
a)
b)
Standardization of NaOH Solution
Trial 1
0.1012
0.00
4.95
Weight of KHP, g
Initial volume of NaOH, mL
Final volume of NaOH, mL
Molarity of NaOH
Average Molarity of NaOH
Analysis of acetic acid in a vinegar sample
Trial 1
Trial 2
1.500
0.500
14.98
26.85
24.84
30.15
Volume of vinegar, mL
Initial volume of NaOH, mL
Final volume of NaOH, mL
Molarity of acetic acid
Average Molarity of acetic acid
Analysis of carbonic acid in a soda sample
Trial 1
Trial 2
Volume of soda, mL
Initial volume of NaOH, mL
Final volume of NaOH, mL
Trial 2
0.1004
5.00
9.99
Molarity of carbonic acid
Average Molarity of carbonic
acid
20.00
20.20
24.48
18.00
24.69
28.50
Trial 3
0.09987
10.01
14.93
Trial 3
1.000
30.87
37.50
Trial 3
20.00
29.48
33.87
W
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning