
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Transcribed Image Text:KS- Latoya Johnson - Learn
=
O STATES OF MATTER
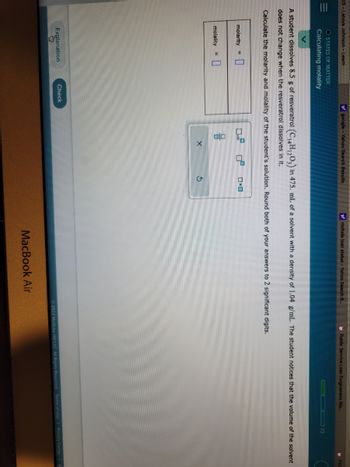
Calculating molality
molarity = 0
molality
Explanation
Say
y google - Yahoo Search Results
A student dissolves 8.5 g of resveratrol (C₁4H1203) in 475. mL of a solvent with a density of 1.04 g/mL. The student notices that the volume of the solvent
does not change when the resveratrol dissolves in it.
Calculate the molarity and molality of the student's solution. Round both of your answers to 2 significant digits.
Check
믐
X
OxO
y mohela loan status - Yahoo Search R...
$
Public Service Loan Forgiveness Wa....
MacBook Air
1/3
Ⓒ2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | A
Expert Solution
arrow_forward
Step 1
Gives mass of solute = 8.5g
Volume of solvent= 475 mL
Density of solvent = 1.04g/mL
Step by stepSolved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A student dissolves 6.6 g of resveratrol (C₁4H₁20₂) in 300. ml. of a solvent with a density of 1.19 g/mL.. The student notices that the volume of the solvent does not change when the resveratrol dissolves in it. Calculate the molarity and molality of the student's solution. Round both of your answers to 2 significant digits. molarity molality= 0 0 0.² ² 0.0arrow_forwardWhat is the molality of a solution made by dissolving 25.9 grams of CaCl 2 in 625 grams of water? 0.747 m 10.5 m 0.00594 m 0.00686 m 0.373 marrow_forwardA solution is made using 189.4 mL of methanol (density 0.7920 g/mL) and 300.0 mL of water (density 1.000 g/mL). What is the molality methanol in water?arrow_forward
- A student dissolves 1.9 g of styrene (C,H,) in 150. mL of a solvent with a density of 1.02 g/mL. The student notices that the volume of the solvent does not change when the styrene dissolves in it. Calculate the molarity and molality of the student's solution. Be sure each of your answer entries has the correct number of significant digits. molarity = x10 molality 미□arrow_forwardWhat is a colligative property? O A property that depends on the number of particles in solution, but not on the nature of the particles. O A property that relates the temperature of a gas to the pressure of the gas. O A property that depends on the specific chemical behavior of the constituent elements and not on the amount in solution. O A property that can only be measured when the moles of gas are constant. O A property that enhances the solubility of a molecule in water.arrow_forwardAn aqueous solution of isopropanol (MM = 60.10 g/mol) has a molality of 7.91 m and a density of 1.18 g/mL. What is the molarity of isopropanol in the solution?arrow_forward
- Calculating the freezing point of an aqueous solution 3. What is the freezing point of an aqueous solution that contains 3 moles of the ionic solute Cack per once kilogram of water? €a* + 2CI Cacl2 4. What is the freezing point of an aqueous solution that contains 2.30 moles of rock salt (NaCI) per one kilogram of water? 5. What is the freezing point of an aqueous solution that contains 1.3 moles of MgC12 per 1 kg water?arrow_forwardA student dissolves 17. g of sucrose (c,H,„0,,) in 250. mL of a solvent with a density of 1.04 g/mL. The student notices that the volume of the solvent does not change when the sucrose dissolves in it. Calculate the molarity and molality of the student's solution. Be sure each of your answer entries has the correct number of significant digits. molarity molalityarrow_forwardA student dissolves 8.0 g of phenol (CH₂OH) in 125. mL of a solvent with a density of 1.06 g/mL. The student notices that the volume of the solvent does not change when the phenol dissolves in it. Calculate the molarity and molality of the student's solution. Round both of your answers to 2 significant digits. molarity molality = x10 DO 0x0arrow_forward
- A solution was made by dissolving 10.000 g of NaCl (58.44 g/mol) in 150. mL (assume 1g/mL) of water. What is the molality of the solution?arrow_forwardA student dissolves 15. g of aniline (C6H5NH2) in 150. mL of a solvent with a density of 0.86 g/mL. The student notices that the volume of the solvent does not change when the aniline dissolves in it. Calculate the molarity and molality of the student's solution. Round both answers to 2 significant digits.arrow_forwardA solution containing 41.684 g of LiF (M.M. 25.939 g/mol) per liter has a density of1.391 g/mL. What is the molality of LiF in the resulting solution?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY