Q: when 200g of propane undergoes complete combustion, what volume of oxygen gas is required at SATP
A:
Q: Aqueous hydrobromic acid (HBr) will react with solid sodium hydroxide (NaOH) to produce aqueous…
A:
Q: A solution contains Cr³+ and Mg2+. The addition of 1.00 L of 1.55 M NaF solution is required to…
A: By determining total mass and total no. of moles of a mixture, we can calculate the mass of…
Q: 5. Use the titration curve to the right to answer the following questions A. What is the pH at the…
A:
Q: Car Bonyl was asked to identify all stereocenters in the following molecule and label them R or S as…
A: R and S configuration can be assigned to the compound using the Cahn-Ingold-Prelog sequence.
Q: how the volume of Clorox solution used is converted into grams and mmol. Clorox solution is 3mL
A: Volume of clorox solution = 3 mlMass = volume × densityMole = mass/molar mass
Q: Aluminum oxide forms when aluminum reacts with oxygen by the following balanced reaction: 4 Al(s) +…
A: We have to calculate the percentage yield for Al2O3 .
Q: 1. Which of the following solutions will be the best buffer at a pH of 9.26? (Ka for HC2H302 is 1.8…
A: Buffer solutions are solutions that resist pH change when small amount of acid or base is added to…
Q: The surface tension of ethanol (CHCH2OH) is 0.022 J/ m2 and the surface tension of hexane…
A: The feature of a liquid known as surface tension results from the cohesive forces between its…
Q: 3) What is the strongest intermolecular force in the following compound? a. London dispersion force…
A: The attraction forces between the different molecules are known as intermolecular forces. In general…
Q: Answer ASAP please! :) Thank you During the corrosion of Magnesium, the anode reaction is ... a) Mg…
A: In electrolytic cell, the reaction at cathode is the reduction reaction (accepting electrons).…
Q: Which compound will give a broad and intense band at 3100 cm-1 in its IR spectra? (a) Pentane (b)…
A: A question based on IR spectroscopy of organic compounds. 4 options are given from which the most…
Q: ify each of the molecules as one of the central electron geometries and molecular shapes. Molecule:…
A: -> electron geometry refers to arrangement of electron density around the central atoms. Geometry…
Q: Complete the following table. Be sure each of your answer entries has the correct number of…
A: We have to calculate the values in each unit i.e. in cal, kcal and in kJ.
Q: 1. The region of electromagnetic spectrum for nuclear magnetic resonance is: a) Microwave b) UV-rays…
A: EMR - ELECTROMAGNETIC RADIATIONS Type of EMR Region of spectrumMicrowave -…
Q: Select the compound with the highest boiling point at standard pressure. O O O O CH4 CH 3 CH 2 CH 3…
A: Boiling point is the temperature at which the substance transitions from a liquid to the gaseous…
Q: The ethyl alcohol content of beers produced is reported as ABV or alcohol by volume (vol/vol) If…
A:
Q: Calculate molar solubility of FeS. Ksp= 4.9 x 10^-18
A: Molar solubility refers to the maximum amount of solute that can dissolve in a given amount of…
Q: Subject :- Chemistry Identify the type of crystalline solid (metallic, network covalent, ionic, or…
A: Solid is mainly classified into two categories:- Crystalline and Amorphous.Crystalline solid is…
Q: Write the two pertinent balanced reactions (standardization and sample analysis) 2. Calculate the…
A: Standardization Reaction:5Na2C2O4 + 2KMnO4 + 8H2SO4 → 2MnSO4 + 10CO2 + 5Na2SO4 + K2SO4 + 8H2OSample…
Q: Phosphorus pentachloride decomposes according to the chemical equation PCI, (g) PCI,(g) + Cl₂(g) K.…
A:
Q: a. Choose the complete Lewis structure for the following partially completed Lewis structure. F…
A: Lewis structure is a way of representing the structure of a molecule in which bond pairs (shared…
Q: Potential Energy 1 02 1 you 2 3 1 1 1 1 Reaction Coordinate 4 Which interval on this diagram…
A: A potential energy diagram of a reaction is given.We have to tell which one interval represent the…
Q: In which step in the acid-catalyzed hydrolysis reaction mechanism of the ester is the direction of…
A: The reaction given is acid-catalyzed hydrolysis of ester, that converts ester into carboxylic acid.
Q: To 64 ml of an 0.015 mM solution of potassium bromide a 2.0 μM solution of silver(1) nitrate is…
A: the reaction between Potassium Bromide and Silver Nitrate gives white precipitate of Silver…
Q: Calculate the dissociation constant (Ka) of a weak acid HJ titrated with 0.100M NaOH given the…
A: To obtain the titration curve, plot the pH on y-axis and volume of NaOH on x-axis. The titration…
Q: (13) Which of the following ionic compounds will be more soluble in acid solution than in water?…
A: Answer :----The compound that is more soluble in an acid solution than in water is (c) Ba(OH)2…
Q: Compare and contrast the physical and chemical properties of: • propan-1-ol and propan-2-ol; •…
A: Propan-1-ol and propan-2-ol, also known as n-propanol and isopropanol, respectively, are two isomers…
Q: Molecular orbitals in metals... A. Overlap significantly to allow free electron movement through the…
A: Molecular orbital or band theory describes the electronic structure and electronic energy level in…
Q: Below you will see a wedge-dash diagram, and an incomplete Newman projection with the designated…
A: Staggered conformation in which the bigger groups are at anti-periplanar
Q: Obtain 2-methylbutanoic acid from diethyl malonate. Comment the reactions briefly.
A: Diethyl malonate is basically a 1,3 diester compound. It is an important starting material for the…
Q: The hydronium ion concentration in an aqueous solution at 25°C is 8.8×10-2 M. The hydroxide ion…
A:
Q: Carotenoids are: Select one: O Alkaloidal saponins O None O Steroidal saponins O Triterpenoidal…
A: Carotenoids are a class of organic pigments that are naturally occurring in various organisms,…
Q: Which bonds allow for a large, bonded network to be more directional and why? 1. Sigma bonds 2. Pi…
A: The question is based on the concept of chemical bonding.We need to explain which type of Bond forms…
Q: 16. The compound that corresponds to the following H-NMR spectrum is: (a) CH3CH₂CH₂Br 11 10 S R (b)…
A: 16.The given spectrum is shown belowWe have to determine the organic compound.The given options…
Q: What is the dihedral (torsion) angle between the two bromine atoms in the wedge-and-dash drawing…
A:
Q: which of the A) SP* B) Pi* c) sp² Following D) Sp²* is not possible
A: "sp2*" is not one of the acceptable alternatives for a hybridization state.In order to create new…
Q: This amino acid has a pK1 = 2.36 and pK2 = 9.68. Its pH is … a. 12.04 b. 6.02 c. 6.00 d. 7.32
A:
Q: Calculate the concentration of the products of the following equilibrium when the equilibrium…
A: Equilibrium constant= 12.3 Equilibrium concentration of NOBr2 = 2.17
Q: H₂C For the following structural formula, pick the structure that accurately represents its MOST…
A:
Q: A slice of cheese has a mass of 63 g and a volume of 41 cm3. What is the density of the cheese in…
A: The density of a substance is given by:The conversion of cm3 into mL is as:
Q: A chemist designs a galvanic cell that uses these two half-reactions: half-reaction NO3(aq) + 4 H…
A: The standard reduction potentials for half cell reactions given are NO3– (aq) + 4 H+ (aq) + 3 e–→NO…
Q: Sulfur atoms combine to form molecules that have different numbers of atoms depending on the…
A: given reaction S8 (g) ⇌ 4 S2 (g) at equilibrium positionThe equilibrium constant (KC ) =…
Q: Solve correctly please. (Gpt/ai wrong answer not allowed) what is the concentration of acetic acid…
A: Given buffer solution is of acetic acid and acetate buffer. We need to find the concentration of…
Q: The number of protons in the nucleus of an atom determines the species of the atom, i.e., the…
A:
Q: 1. Consider the first ionization energy for two atoms: Nitrogen (N) and Oxygen (O). Which one has…
A: The correct answer is 1,1 . Nitrogen.Nitrogen has a higher ionization energy compared to oxygen.…
Q: 1) Which of the following elements is a metal? A) nitrogen 2) Which of the following elements is a…
A: 1.nitrogen is a non metalic element Which belongs to group 15.2.fluorine is also a non metal which…
Q: Calculate the molar solubility of CaCO3, Ksp 8.7×10 ⁹.
A: When sparingly soluble salt is dissolve in water most of the salt remain undissolved in the…
Q: Question A10 Complete the missing blank in the following question: Rf value is (a) (c) (d) Distance…
A: A question based on analytical processing. 4 options statements about Rf value are given, from which…
Q: Which of the following molecules cannot be a hydrogen bond donor? Э !!!!! Э IIII H3C Э III NH CH3 NH…
A: -> Hydrogen bond can be formed when hydrogen attached with most electronegative atom hence,…
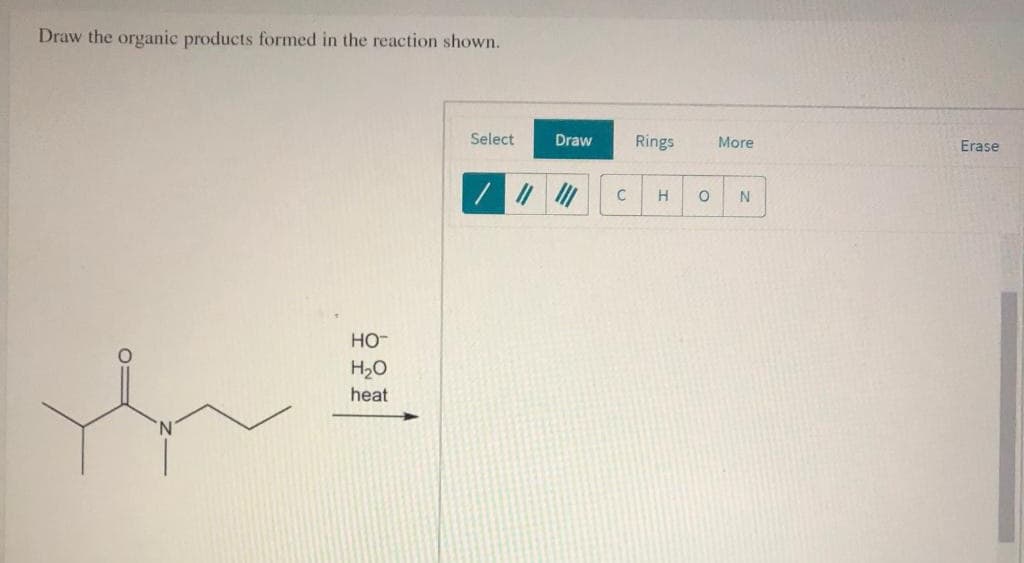
Step by step
Solved in 3 steps with 2 images


