. A protein has been sequenced after cleavage of disulfide bonds. The protein is known to contain 3 Cys residues, located as shown below. Only one of the Cys has a free - SH group and the other two are involved in an -s-s- bond. A Phe Cys « N-terminus Cys- - Met Cys- C-terminus The only methionine and the only aromatic amino acid (Phe) in this protein are in the positions indicated. Cleavage of the intact protein (i.e., with disulfide bonds intact) by either cyanogen bromide or chymotrypsin does not break the protein into two peptides. Where is the -s-s- bond (i.e., AB, BC, or AC)?
. A protein has been sequenced after cleavage of disulfide bonds. The protein is known to contain 3 Cys residues, located as shown below. Only one of the Cys has a free - SH group and the other two are involved in an -s-s- bond. A Phe Cys « N-terminus Cys- - Met Cys- C-terminus The only methionine and the only aromatic amino acid (Phe) in this protein are in the positions indicated. Cleavage of the intact protein (i.e., with disulfide bonds intact) by either cyanogen bromide or chymotrypsin does not break the protein into two peptides. Where is the -s-s- bond (i.e., AB, BC, or AC)?
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter31: Completing The Protein Life Cycle: Folding, Processing, And Degradation
Section: Chapter Questions
Problem 3P: Understanding the Relevance of Chaperones in Protein Folding Protein molecules, like all molecules,...
Related questions
Question
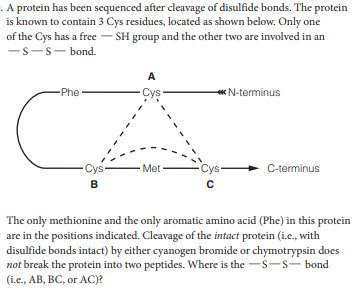
Transcribed Image Text:. A protein has been sequenced after cleavage of disulfide bonds. The protein
is known to contain 3 Cys residues, located as shown below. Only one
of the Cys has a free - SH group and the other two are involved in an
-s-s- bond.
A
Phe
Cys
« N-terminus
Cys-
- Met
Cys-
C-terminus
The only methionine and the only aromatic amino acid (Phe) in this protein
are in the positions indicated. Cleavage of the intact protein (i.e., with
disulfide bonds intact) by either cyanogen bromide or chymotrypsin does
not break the protein into two peptides. Where is the -s-s- bond
(i.e., AB, BC, or AC)?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning