1- A student spotted a mixture of benzophenone and biphenyl on a TLC plate. She then developed the TLC plate in hexane and obtained the following result: (10 pts) علی benzophenone biphenyl TLC Plate after Hexane a. Label the spots on the plate above to indicate which is benzophenone and which is biphenyl. b. Fill in below what the developed plate would have looked like if the student had run the plate in acetone (2-propanone) instead of hexane
1- A student spotted a mixture of benzophenone and biphenyl on a TLC plate. She then developed the TLC plate in hexane and obtained the following result: (10 pts) علی benzophenone biphenyl TLC Plate after Hexane a. Label the spots on the plate above to indicate which is benzophenone and which is biphenyl. b. Fill in below what the developed plate would have looked like if the student had run the plate in acetone (2-propanone) instead of hexane
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 3P
Related questions
Question
Help with question
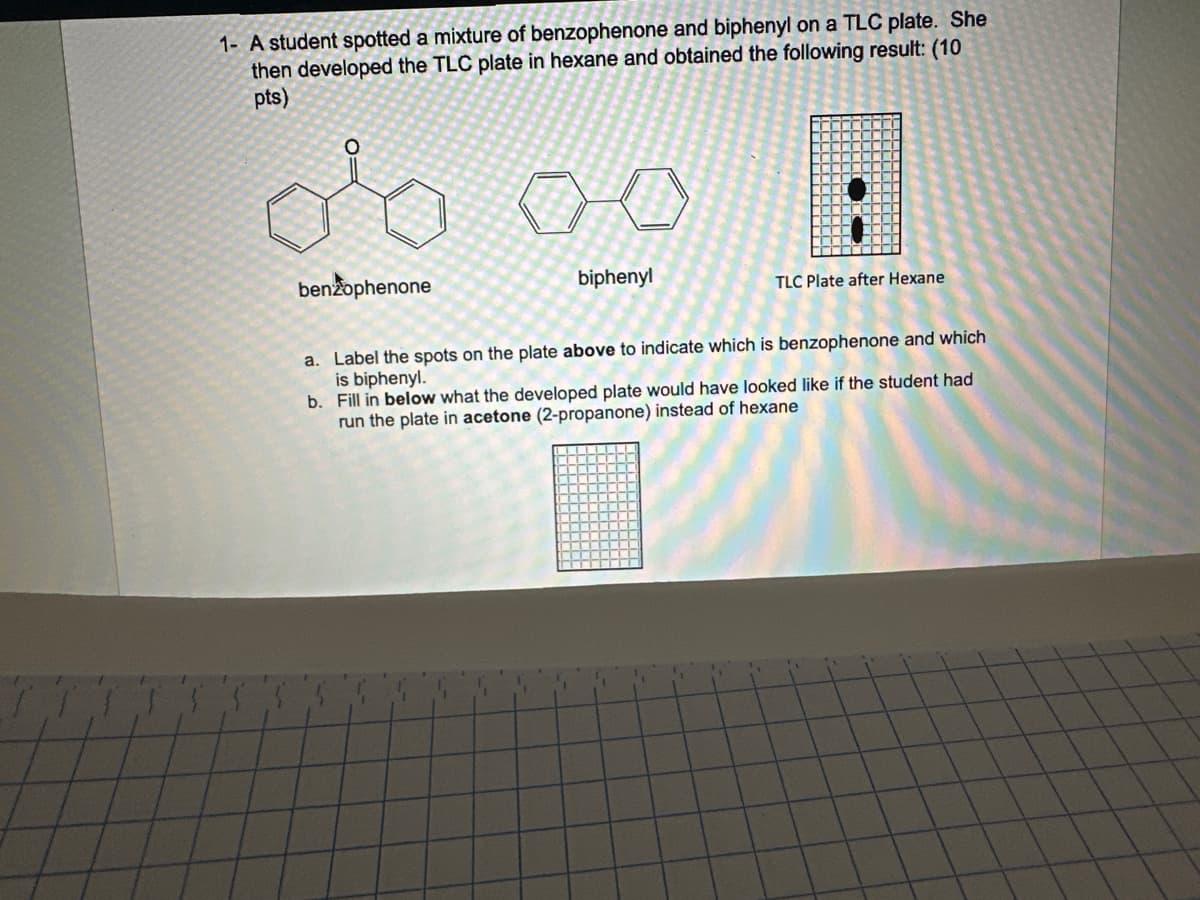
Transcribed Image Text:1- A student spotted a mixture of benzophenone and biphenyl on a TLC plate. She
then developed the TLC plate in hexane and obtained the following result: (10
pts)
علی
benzophenone
biphenyl
TLC Plate after Hexane
a. Label the spots on the plate above to indicate which is benzophenone and which
is biphenyl.
b. Fill in below what the developed plate would have looked like if the student had
run the plate in acetone (2-propanone) instead of hexane
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

