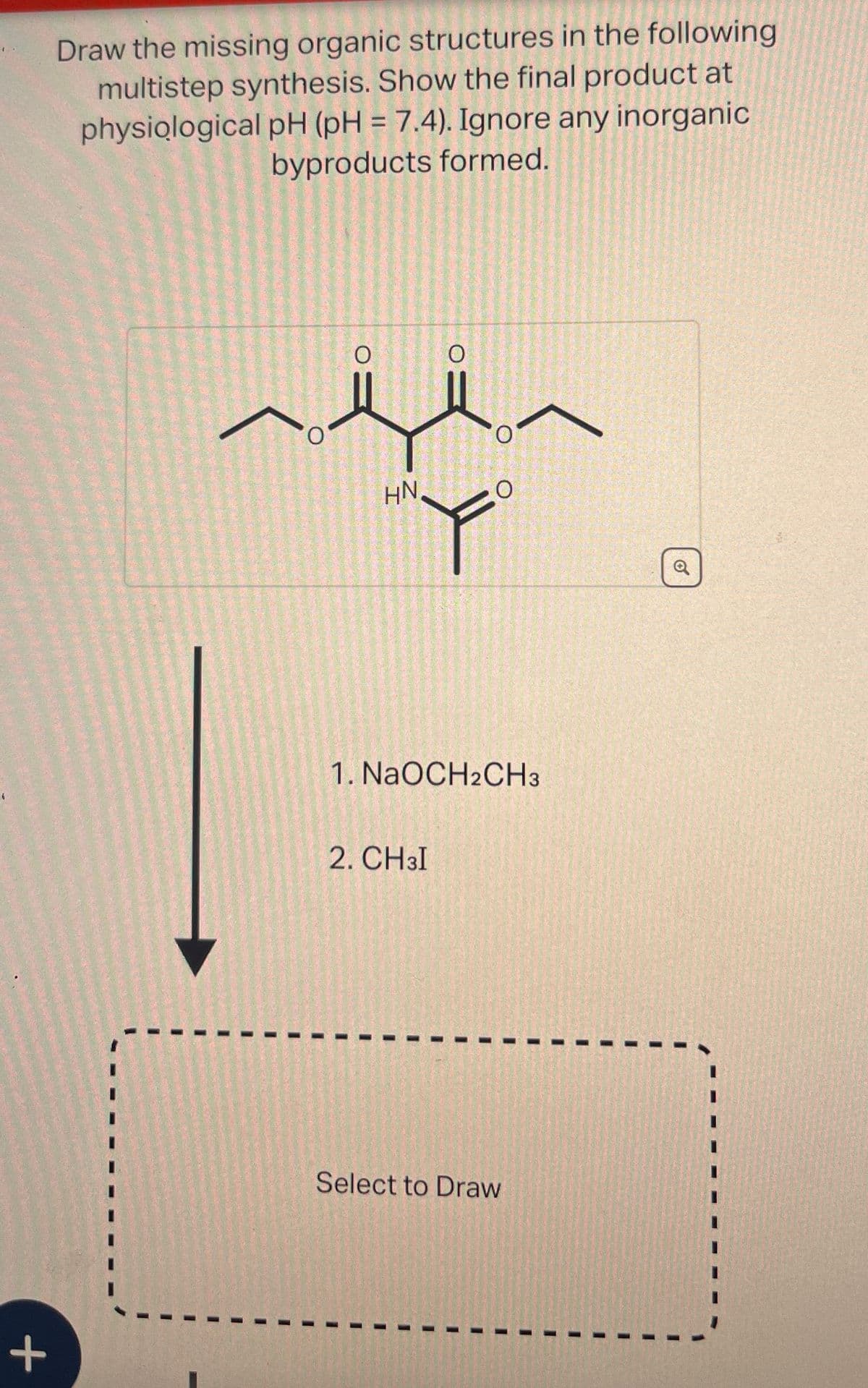
Draw the missing organic structures in the following multistep synthesis. Show the final product at physiological pH (pH = 7.4). Ignore any inorganic byproducts formed. + 0 O HN 1. NaOCH2CH3 2. CH3I Select to Draw
Q: None
A: The question is asking us to determine the nature of two cycloaddition reactions, specifically…
Q: None
A:
Q: None
A: Step 1: an electrocyclic reaction can either be classified as conrotatory or disrotatory , based on…
Q: Use Le Chatelier’s Principle to predict the color change, if any, when you add more HClsolution to…
A: The objective of the question is to predict the color change, if any, when more HCl solution is…
Q: A mixture of 2 mL of 3 M NaOH solution, 2.5 mL 95% ethanol, 0.212 g benzaldehyde, and 0.058 g…
A: The reaction is the Aldol condensation, 2C6H5CHO+CH3COCH3→C6H5CH=CHC(O)CH=C6H5+2H2Owhere…
Q: 8. Analyze the 13C NMR spectrum for the C14H10 compound shown below and determine a structure.…
A: The objective of this question is to analyze the 13C NMR spectrum for the C14H10 compound and…
Q: Choose the TRUE statement about the effect of the presence of a catalyst on reaction rate. In the…
A: The objective of the question is to identify the correct statement about the effect of a catalyst on…
Q: Draw structural formulas for the two compounds you could use to prepare the amine shown by reductive…
A: To form the given structure via reductive amination, we prepare it using a ketone and a secondary…
Q: 9. Determine the product of the following Diels-Alder reaction. CO₂Et 206°C
A: The single bonds are free to rotate. The rotations are made accordingly to facilitate the [4+2] type…
Q: [Assessments are ranked according to their difficulty; ●●●●= most difficult.] 16.40 (•) Predict the…
A: The question is asking to predict the products of the reactions of alkyl halides under various…
Q: 1. CN Br 2. LiAlH4 3. H₂O NH2 2a Primary amines can be prepared from nitriles by reduction with…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: T₂ H(T₂) = H(T) + [²C,dT T₁ T₂ A‚H°(T,) = A‚H°(T; ) + √„ª ACT (2C.6) Kirchhoff's law (2C.7a) P T₁ -…
A: Sure, I can help you with this. The text in the image pertains to thermochemistry, which is a branch…
Q: A 0.760-g sample of an unknown gas has a volume of 759 mL and a pressure of 399 mm Hg at 34.2 °C.…
A: The objective of this question is to calculate the molar mass of an unknown gas given its mass,…
Q: Using this reaction energy diagram, determine the activation energy for the reverse reaction. 140…
A:
Q: Draw the structure(s) of the major organic product(s), including counterions, of the following…
A:
Q: A sample of krypton gas at a pressure of 794 mm Hg and a temperature of 71 °C, occupies a volume of…
A: The objective of the question is to find the volume of a sample of krypton gas when it is cooled…
Q: Chemistry
A: The freezing points of the given substances are as follows: H₂O (Water): The freezing point is…
Q: Calculate and balance of equations that show how the substance dissolves in water.
A: The objective of the question is to balance the equations that show how the given substances…
Q: A sample of oxygen gas occupies a volume of 9.84 L at 46.0°C and 1.29 atm. If it is desired to…
A: The objective of the question is to find the temperature of the oxygen gas sample at the new volume…
Q: Consider a 1mg sample of cobalt- 60(). What is the expected exposure rate 1m away from the source…
A: the problem appears to be about calculating the expected exposure rate from a 1 mg sample of…
Q: A 1.44-g sample of an unknown gas has a volume of 896 mL and a pressure of 742 mm Hg at 37.9 °C.…
A: The objective of this question is to calculate the molar mass of an unknown gas given its mass,…
Q: Do the reactions below proceed in good yield from left to right as shown? a) b) 1 equivalent c) NH₂…
A: Step 1: Step 2: Step 3: Step 4:
Q: Please don't provide handwritten solution ...
A: The reaction above is light-initiated allylic bromination by N-bromosuccinimide (NBS).In this…
Q: Write the structural formula of the organic product for the given reaction between an alkyne and an…
A: Step 1:Alkylation of terminal alkyne (Or alkylation of acetylide anion): Terminal Alkynes (having…
Q: 2) Determine the molecular formula for following structure shown in the box: A) C10H18 B) C10H20 OOO…
A: Step 1:Molecular formula contains the total number of atoms of each type that are present in the…
Q: Equilibrium of Co2+and Cl-with CoCl42-a) Give a balanced reaction equation for the reaction between…
A: The objective of the question is to understand the chemical reaction and properties of the cobalt…
Q: The air pressure is approximately 0.35 atm at an altitude of 3.0 km. Most airplane cabins are…
A: The objective of this question is to understand the relationship between pressure and volume of a…
Q: Calculate the density of argon gas (in g/L) at 666 mm Hg and 41.3 °C. g/L
A: The objective of this question is to calculate the density of argon gas under given conditions of…
Q: b. Please provide a "curved arrow" mechanism for the following transformation. OH H+ Браон -OH
A: Step 1:Initial Structure (Left Side)-The left side of the image shows a complex molecule with…
Q: A reaction with an enthalpy of -16.9 kJ and and entropy of -265 J/K is being run at -115°C. What is…
A: Given: ΔH=−16.9kJ;ΔS=−265J/K;ΔG=???;T=−115°CStep 1: Write the equation we need.ΔG=ΔH−TΔS Step 2:…
Q: The equilibrium constant, K, for the following reaction is 3.75 × 1011 at 22 °C. 2A (g) + B2 (g) =…
A: The objective of the question is to determine the composition of the equilibrium mixture based on…
Q: The figure to the right illustrates the long-run average cost curve for a company that makes motors.…
A: When analyzing the long-run average cost curve of a company, we're essentially studying how average…
Q: A 0.175 mol sample of N2 gas is contained in a 4.00 L flask at room temperature and pressure. What…
A: The objective of this question is to calculate the density of nitrogen gas under given conditions.…
Q: A patient requires a dose of 99m-Technetium of 10 MBecquerel contained within 2 mL of injection. The…
A: The objective of the question is to calculate the amount of 99m-Technetium that needs to be eluted…
Q: In the reaction, sodium and chlorine combine to produce sodium chloride. In an instance when four…
A: Step 1:In the reaction, 2Na + Cl2 → ? NaClFirst balance the reaction, 2Na + Cl2 → 2 NaCl In the…
Q: In the reaction of potassium metal with chlorine gas to produce potassium chloride, which chemical…
A: The objective of the question is to identify which chemical is reduced in the reaction of potassium…
Q: What quantity of heat (in kJ) will be absorbed by a 33.1 g piece of aluminum (specific heat = 0.930…
A: Approach to solving the question:Please see attached photos for detailed solutions. Thank you.…
Q: Calculate the mass (in g) of SiO2 that reacts completely with 15.0 grams of carbon according to the…
A: Given mass of carbon= 15.0 gMolecular mass of carbon = 12.0 g/molNumber of moles of carbon= given…
Q: Enter electrons as e*. Use smallest possible integer coefficients for ALL reactions. If a box is not…
A: Step 1:Galvanic cell is an electrochemical cell that converts chemical energy into electrical energy…
Q: 2. i) Circle each stereogenic unit in the Structure provided and write its label directly below the…
A: Step 1: Step 2: Step 3: Step 4:
Q: P13.5: The biosynthetic pathway for the antibiotic compound rabelomycin begins with the condensation…
A: Step 1: Step 2: Step 3: Step 4:
Q: What masses of dimethylamine and dimethylammonium chloride do you need to prepare 5.00 L of pH =…
A: Step-1: pH = 12Volume= 5LConcentration of both = 0.5M.pKa of dimethylamine= 10.73. Step 2: The…
Q: None
A: Generally P exhibits valence 3, sulfur and oxygen exhibit valence 2. Among the given structures…
Q: why CO2 are nonpolar bonds
A: The question is asking why the bonds in a carbon dioxide (CO2) molecule are nonpolar. To answer…
Q: According to the HONC rule, oxygen atom has a neutral formal charge , when it forms 2 bonds around…
A: The HONC rule is a simple way to remember how many bonds are generally formed by the atoms of…
Q: How much energy would be produced from the reaction of 2.40 moles hydrogen with 3.95 moles chlorine
A: The objective of the question is to calculate the energy produced from the reaction of hydrogen and…
Q: 7. Copper metal crystallizes in a face-centred cubic structure. If the atomic radius of copper is 64…
A: The objective of this question is to calculate the volume of a unit cell of copper, which…
Q: Does any solid PbCl2 form when 0.30 L of 0.86M NaCl is dissolved in 0.25 L of 0.12 M Pb(NO3)2? Ksp…
A: Given:…
Q: Calculate the value of is for H+, He+ and Li+2 ions at r = 0, r = a, and r = 4a,, Rationalize the…
A: Step 1: Step 2: Step 3: Step 4:
Q: F3 Modify the structure to draw the organic product of the following reaction. Note that [O] is…
A: The given structure is CH2CHOHCH3, which is a secondary alcohol. The presence of [O] indicates that…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- The overall adenylate kinase reaction (2 ADP = AMP + ATP) is a combination of 3 reactions: ATP hydrolysis, pyrophosphate hydrolysis, and ATP formation. Show each step and provide the standard free energy (ΔG°′) for each and the overall reaction. If the adenylate kinase reaction is at equilibrium and intracellular [ATP] = 5 mM and [ADP] = 0.5 mM, calculate the concentration of AMP at pH 7 and 25 °C. Show all work. ATP (ADP+P) = -30.5 kj/mol; ATP (AMP+PP) = -45.6 kj/mol; PP = -19.2 kj/molFrom Table 1.1 indicates, among other things, the melting points of various fatty acids. Analyze the trend that the melting points of saturated fatty acids follow, from n-dodecanoic (CH3-(CH2) 10-COOH) to n-tetracosanoic (CH3-(CH2)22-COOH), and answer both next questions. Briefly justify your answers. i) What value would you expect the melting point of triacontanoic acid (CH3-(CH2) 28-COOH) to have? Choose one of the following options:a) More than 200 ° Cb) About 94 ° Cc) Less than 86 ° Cd) Exactly 86.1 ° C ii) What value would you expect the melting point of octanoic acid (CH3-(CH2) 6-COOH) to have? Choose one of the following options:a) 43.5 ° Cb) -20 ° Cc) About 20 ° Cplease please help me to answer it >>> i have an exam Human xanthine oxidase catalyzes the oxidation of hypoxanthine to xanthine and can furthercatalyze the oxidation of xanthine to uric acid. For the treatment of hyperuricemia and gout,several medications are used to inhibit the activity of xanthine oxidase and reduce theproduction of uric acid. You are a biochemist and just discovered a chemical that can inhibitthe activity of the human xanthine oxidase. When analyzing its mode of inhibition, you foundthat the enzyme inhibitor complex requires 450 J.mol-1 to dissociate and that it displayskinetics somehow similar to noncompetitive inhibition. You sent your inhibitor to the ministryof health for approval as a medication for gout. Based on the data provided, are they going toauthorize it as a medication or not? Explain?
- 1. draw the product in the ff reactions 2. show the ring closure scheme for the glucose molecule3) The enzyme aldolase catalyzes the conversion of fructose-1,6-diphosphate (FDP)to dihydroxyacetone phosphate (DHAP) and glyceraldehide-3-phosphate (G3P). Thereaction isFDP ⇄ DHAP + G3P , ∆rG0(298.15 K) = 23.8 kJ mol−1In red blood cells the concentration of these species are [FDP] = 35 µM, [DHAP] =130 µM, and [G3P] = 15 µM. (Remember that 1.0 µM = 1.0 × 10−6 mol L−1. Thestandard state for reactions in solution can be taken as c0 = 1.0 mol L−1).Calculate ∆rG in a red blood cell at 25 0C. Will the reaction occur spontaneouslyin the cell at this temperature?Which of the reactions are spontaneous (favorable)? 1)DHAP↽−−⇀glyceraldehyde-3-phosphateΔ?=3.8 kJ/mol 2)L-malate+NAD+⟶oxaloacetate+NADH+H+Δ?=29.7 kJ/mol- 3) glutamate+NAD++H2O⟶NH+4+α-ketoglutarate+NADH+H+Δ?=3.7 kcal/ 4) C6H13O9P+ATP⟶C6H14O12P2+ADPΔ?=−14.2KJ/mol 5) −Rh(I)C2H6Δ?=−150.97 kJ/molC2H4+H2→Rh(I)C2H6ΔG=−150.97 kJ/mol 6) C4H4O5⟶C4H2O4+H2OΔ?=3.1 kJ/mol
- In one of the steps in this pathway fructose 1,6-biphosphate (F1,6BP) is converted to gylceraldehyde-3-phosphate (G-3-p) and dihydroxyacetone phosphate (DHAP). This reaction is catalyzed by the enzyme aldolase. For this reaction at 25 celcuius and Ph7 we have: Keq= 10-4M and Delta G = +5456 cal/mol Calculate the following: The concentration of F1, 6BP, DHAP and G-3-P at equilibrium when the initial F1,6BP is (A) 1M, (b) 10-2 M, (c) 2 X 10*4 M and (d) 10-5 M. I been trying to solve this problem but I dont even know where to begin with. Atleast help me with problem A, I would be able to guide myself through.In step 7 of fatty-acid biosynthesis (Figure 29-5), dehydration of a -hydroxy thioester occurs to give trans-crotonyl ACP. Is the dehydration a syn elimination or an anti elimination?One of the steps in the metabolic degradation of guanine is hydrolysis to give xanthine. Propose a mechanism.
- Assume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material for the biosynthesis of mevalonate, as shown in Figure 27-7. At what positions in mevalonate would the isotopic label appear?When some sugars dissolve in water they spontaneously undergo changes in optical rotation called mutarrotation. The Mutarrotation of D-glucopyranose is catalyzed by acid and bases. 2-Hydroxypyridine is a more effective catalyst than phenol and pyridine for this reaction because: a.Both oxygen and N in 2-hydroxypyridine act as bases increasing the rapid interconversion of sugar b. The OH of 2-hydroxypyridine serves as the base while the current N as the acid. c. 2-hydroxypyridine acts both as a base to remove the proton from the hydroxyl group in the hemiacetal and as an acid to provide a proton to the oxygen in the hemiacetal. d.Phenol and pyridine are very expensive.When in contact with the body, Methanol is oxidised to Methanal by ____________ . Select your answer. KMnO4 Aldehyde Dehydrogenase Chromic Acid Alcohol Dehydrogenase





