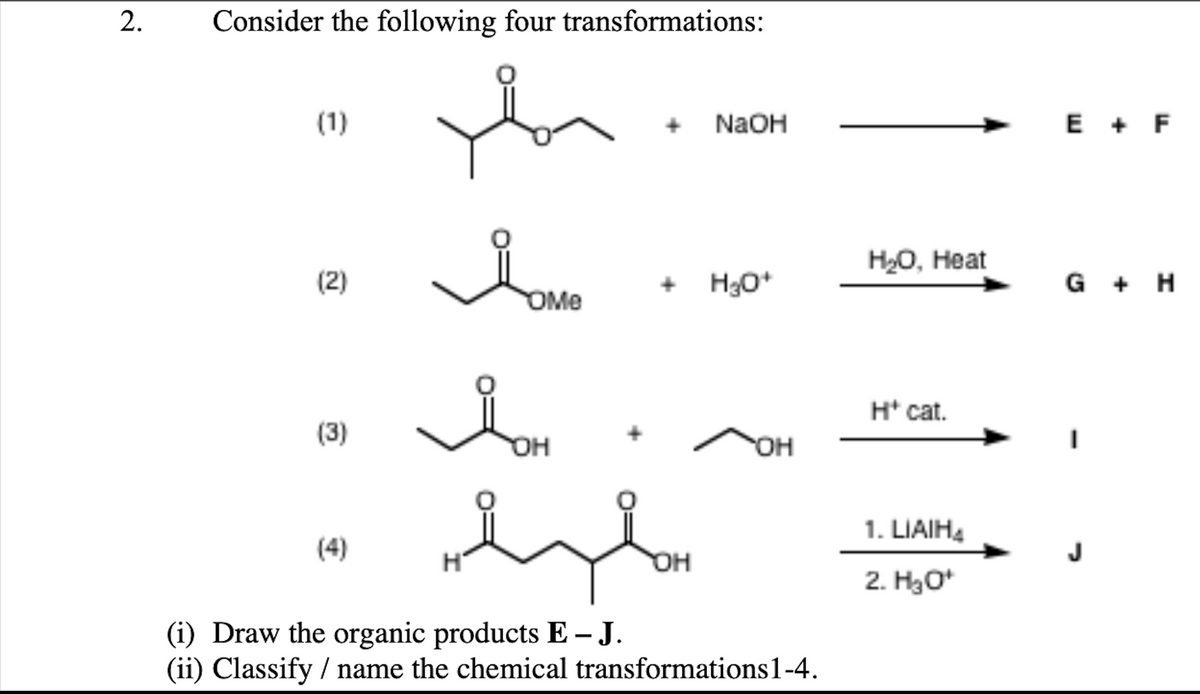
(1) NaOH E + F НО, Нeat (2) + H30* G + H OMe H* cat. (3) OH HO. 1. LIAIH. (4) OH 2. H3o* (i) Draw the organic products E – J. (ii) Classify / name the chemical transformations1-4.
Q: Q2: From the data shown below, eplain Lipinski's nule of 5 and determine whether or not meets the…
A:
Q: Find the products (A and B) for the following reaction sequence: 1.FeBr3, Br2 1. CO,, H3O+ 2. Mg,…
A:
Q: НО. НО OH A ОН D OH OH HO НО. OH OH OH + OH OH H* (Catalyst) H2O B Өон E НО НО НО НО. НО OH OH C F…
A:
Q: In cach reaction box, place the best reagent and conditions from the list provided. 1) Br 2) 3) 4)…
A:
Q: Synthesis #3 08-0 from Synthesis #4 Q CH3 from
A:
Q: synthesize bromobenzene from benzene?
A: 3.4 Bromination of benzene 3.5 friedel craft alkylation oxidation
Q: Is the following true or false? A highly selective reaction is one that proceeds with a high…
A: Selectivity of a substrate in an enzyme indicates the degree to which a substrate is bound and…
Q: 1. NACN, H* 2. H20°, Heat 3. PCC ?? он но но Он но
A: We have given the organic reaction and we have to find the major product of the reaction.
Q: Assume that student reacts 1.55 mL of 2-methylcyclohexanol (d=0.93 g/mL) with 0.50 mL of 85 %…
A: To express the efficiency of a reaction ,you can calculate the percent yield . % yield = actual…
Q: Start: LDA 0300H СМА
A: Correct answer is option C 2's complement conversion of 0300H content
Q: Cl i) mechanism: ii) most important determining factor(s): iii & iv) detailed, stepwise mechanism(s)…
A:
Q: enthaipy of fomation of pneno enthaipy formation of pienoi gaseous Carb -393.51 kJ mol1 2C6H5OH +…
A: Given-> 2C6H5OH +15O2 -----> 12CO2 + 6H2O I think it asked standard enthalpy of given…
Q: a)Determine AGrxn° values for the following Combustion reactions at 25°C b) Decide whether this…
A: Given data-
Q: EXPLAIN IN 3-5 SENTENCES: 1. What are the tissue sources of Alkaline phosphatase?
A: Since you posted multiple questions, as per guidelines we will solve only first one for you.…
Q: The bacterial load of a ketchup (pH-3.9) can be reduced from 6,000 units to 6 units in 15 minutes at…
A:
Q: Given: H2SO3(g) → H2O (g) + SO2 (g) Keq = 100 What will…
A: Equilibrium constant of a reaction, Keq can be written on the basis of law of action of mass. It is…
Q: 20% H2SO4 L added Au1 i
A: The values of added gold and emission intensity count can be given. To find the concentration of…
Q: LOH NaHCO, H3C- H3C HO. ČH3 H3C NaOH H3C. OH H3C H3C Table of nhysical constants for each reactant…
A: Given are two reactions,
Q: fattlreatin? %24 100 0.200 000 what is the rate ofreachor fa hydrasen 007.0 0.00 He स
A: Rate of reaction of hydrogen means how fast the Hydrogen is being consumed up in the reaction per…
Q: the reaction? cycloaddition CO₂Me CO Me HPh CO₂Me CO₂Me
A: Organic compounds can be defined as the compounds that contain carbon and hydrogen atoms in a…
Q: + O2 (g) → 2 NO2(g) Calculate AGrxn at: a) 25 °C
A: The question is based on the calculation of Gibbs free energy change. we have been provided a…
Q: ugen.wileyplus.com/edugen/student/ aintr.uni Klein, Organic Chemistry, 3e actice Assignment…
A: This is a enamine formation reaction followed by electrophilic attack.
Q: oue:-complete ithe given reactions with the mecessaryftarting material or greduct.? Naocts NaoHas…
A:
Q: Below you will find five reaction products (labeled product 1, product 2, etc.), but you will only…
A: Oxymercuration-demercuration: An alkene is treated with Hg(OAc)2. The product of this reaction is…
Q: I need help with the problem attached to the file
A: A chemical reaction is symbolic representation of the conversion of substances to new substances.In…
Q: 2. You omit the following steps. Circle the correct answer and briefly explain in the space…
A: the solution is shown below.
Q: 17- If 125 grams of solid sucrose (C12H22011) with 93% purity by mass is dissolved in 250.0 mL of…
A: Purity of sucrose = 93% Weight of sucrose = (93/100) x 125 = 116.25 gram Molar mass of sucrose =…
Q: e. -2257 kJ 4. Calculate AGrxn at 298 Ku 3 Oz(g) → 2 03(g P(O2) = 0.41 atm, a. +341 kJ b. +17.8 kJ…
A:
Q: OH C (i) H. NaCI CN racemic mixture 2 HNME2 (ii) C9H1,NO cP NH,Me2 Ph D E (i) ELOH OEt ONa но (iv)…
A: In an organic reaction, reactants usually undergo substitution, elimination, or addition reactions.…
Q: What is the effect of blending petrol with alcohol?
A: The answers are shown below. Good luck with the finals.
Q: Average Reaction Rate of Reaction Time (s) Molarity Molarity of Acetone (M) of HCI (M) Molarity of…
A: Given: Order for Acetone = 1 Order for H+ (HCl) = 1 Order for Iodine = 0 Rate of reaction for…
Q: GUSUIN CI- H. eviaoxe (MCPBA) elugelom ert notageta brs ainepse pnileil elom ert not eg CH3OH…
A: mcpba generally used for epoxidation. It also used in Bater villeger oxidation reaction.
Q: The basic hydrolysis of a nitrile yields first an amide and then a carboxylic acid salt plus ammonia…
A:
Q: Consider the following values of Keg for a reaction. Would you expect the reaction to be: dominated…
A: The equilibrium constant gives the ratio of the concentration of the products to that of the…
Q: (ii) CO₂Me 1) Zn/Cu, CH₂l2 2) Et₂0/reflux
A:
Q: ml Diluted Undiluted Diluted to mL Water Absorbance Unknown Conc. (M) Conc. (M) 0.5 4.5 0.4IS 1.25…
A: Given are the volumes of unknown sample which are diluted with given varying volume of water. The…
Q: Q. 8) Conversens hemi -acetal Catalyt auid then to acctal
A: The conversion of cyclohex-2-enone to hemiacetal and acetal has to be given.
Q: ......
A:
Q: What are the products of the following reactions?
A: Since you posted question with multiple sub-parts, we are entitled to answer the first 3 only.
Q: 28(g) Give name(s) of major organic product(s) of the following reaction. CH,CH, HBr, H,O reflux
A: The given starting material is an ether. The ethers are generally unreactive. Strong acid cleave the…
Q: methylbenzene + conc HNO, & conc H,SO, at 50°C to make 4-nitromethylbenzene
A: Detail mechanistic pathway is given below
Q: Question 7 (c) (i), (ii), (iii), and c(iv)…
A: The kinetics of a reaction can be studied experimentally. The equlibrium concentration of a reaction…
Q: redict the product of the intramolecular alkoxymercuration-demercuration below. 1. Hg(OAc) 2. NABH,…
A: Chemical reaction in which organic compounds take part are called organic reactions. Organic…
Q: но Ph A (i) `Ph LIAIH4 then aqueous workup H+/heat D
A: Ketone show Addition reactions With grignard reagent .
Q: You discovered a potential new drug molecule, which binds to its target with a binding free energy…
A: Binding free energy is defined as the sum of all the intermolecular interactions present between the…
Q: How to determine ΔHo for the attached reaction.
A: The enthalpy for the reaction is calculated as shown below.
Q: 1) Calculate the percent by volume of acetic acid by fermentation. Assumptions: No volume changed…
A:
Q: Construct the expression for Kc for the following reaction: 2 C₂H₄(g) + O₂(g) ⇌ 2 CH₃CHO(g)
A: The Expression of Kc is nothing but the expression of equilibrium when we talk in terms of…
Step by step
Solved in 2 steps with 1 images

- After graduating from your PhD, you are now working as a post-doctoral researcher in BASF , the world’s largest multinational chemical company. The management of your R&D department decided to look at the potentiality of chloroform-methanol mixture in removing the lipids of various biological systems. Your boss, a senior scientist at your department, thinking about the hydroxyl group on the alcohol and thet hree chlorines on the CHCl3 , suggested that the mixture is probably non-ideal. Being one of her brightest colleagues, she then tasked you to graph the partial molar enthalpies of the chloroform methanol mixture as a function of mole fraction. Ardently, you accepted the challenge and went back to your office. After several hours of searching, you finally come up with enthalpy of mixing data at 50oC fort his mixture (See the picture). Please provide the graphs that the boss has asked you for. Put both curves on the same graph. Note: provide your excel file for this5- Write down one paragraph (with proper equations) the steps for synthesizing a sample ofAspirin.6- Explain in details (one page hand writings) or one page typed the technique TLC andhow it is used in identification and characterization of organic compounds.A. Without the use of H2 reductions or PCC oxidation fill in the blank steps of the reactions. Final products should be the major organic product.
- I have this task in organic chemistry (book: Brown's introduction to organic chemisty, global edition). Task 10:42. In (a) I have to tell what the funcion of K2CO3 is in step 1. Is it that CO32- take the hydrogen atom in 1-napthol? Will it then be a SN2 mechanism? In (b) I have to name the amine used in step 2 to form Propanolol. But I can't really find out how to come up with an amine that will make that reaction. Here are two pictures of the task:Box 31-33 .Unless otherwise directed, show only the major product. For reagent or reactant questions, fill in the missing information.Orgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???
- Complete and balance the following methesis reaction in aqueous solution.Answer Entery Instructions• Parentheses are only required around polyatomic ions and only when more than 1 of those ions is present.K3PO4 (no parentheses required).Mg3(PO4)2 (parentheses required).• In each case one product is soluble (aq) and one insoluble (s). Use solubility rules to determine which product appears in each box.***Answer*** Na2S (aq) + ***Answer*** FeCl3 (aq) ➝ ***Answer*** ***Answer*** (s) + ***Answer *** ***Answer*** (aq)Which of the following statements is not correct? Oxymercuration-demercuration for alcohol synthesis is complicated by carbocation rearrangement. Oxymercuration-demercuration for alcohol synthesis follows Markonikov's Rule. Sodium borohydride is used in the last step of oxymercuration-demercuration to oxidize the hydroxyalkyl mercury compound. All of the above. Both the 1st and the 3rd statements.Table 6.4. Oxidation: Reaction with Baeyer’s reagent (cold, dilute, neutral, aqueous KMnO4) Write (+) if the reaction is positive, (-) if negative. SAMPLE OBSERVATIONS +/- cyclohexane cyclohexene benzene toluene acetylene Enumerate the classes of hydrocarbons that are oxidizable by Baeyer’s reagent. ________________________________________________________________________ ________________________________________________________________________ What is the structural feature of these classes of hydrocarbons? ________________________________________________________________________ ________________________________________________________________________
- 4. Compound A has the formula C 8H 8. It reacts rapidly with KMnO 4 to give CO 2 and a carboxylic acid, B (C 7H 6O 2), but reacts with only 1 molar equivalent of H 2 on catalytic hydrogenation over a palladium catalyst. On hydrogenation under conditions that reduce aromatic rings, 4, equivalents of H 2 are taken up and hydrocarbon C (C 8H 16) is produced. What are the structures of A, B, and C.Show the wurtz reaction of 1-fluoro-2-methylpropane and name the productsWrite the type of reaction that is occurring on the line provided then complete the reaction.Only include the major products and any byproducts (e.g. H2O) but no minor products.

