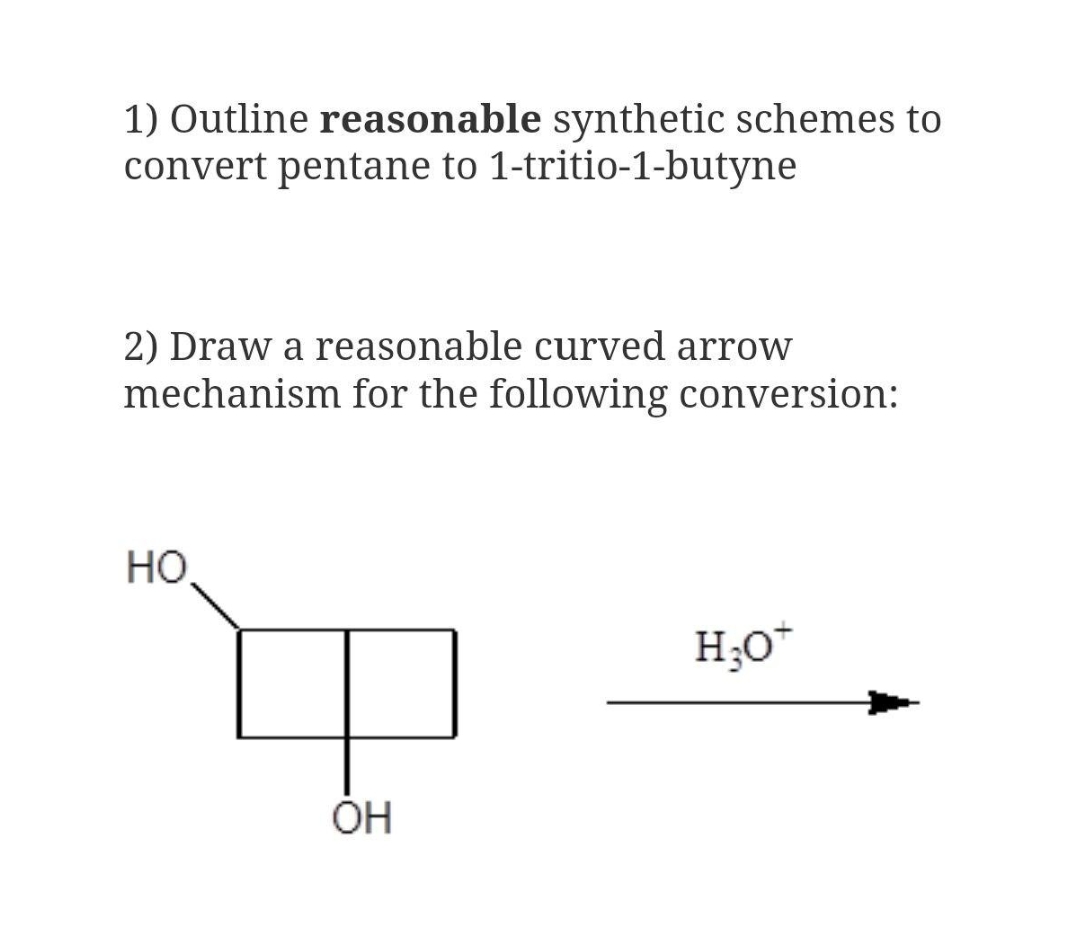
1) Outline reasonable synthetic schemes to convert pentane to 1-tritio-1-butyne 2) Draw a reasonable curved arrow mechanism for the following conversion: HO H;O* OH
Q: 2. If there are 1652 kJ of bond energy in methane, how much energy is there in a SINGLE Carbon Hydro...
A:
Q: 8. 1MIEndel eats a coökié at lunch. Which of the following actions during the digestive process is a...
A: Please find your solution below : Digestion is a chemical change in which the enzymes breakdown the ...
Q: Which of the following is the structure of the acid chloride that is formed in the reaction shown in...
A:
Q: Make an schematic diagram for procedure below: B. %SO3 determination Transfer all precipitate to t...
A: We need to make a schematic diagram for the given procedure.
Q: 2. A solution that may contain Cu, Bi?", Sn*, or Sb²+ is treated with thioacetamide in an acidic med...
A: We have find out group 2 element
Q: 15.0 g of carbon dioxide gas is stored in a cylinder (a malfunctioned fire extinguisher) with a volu...
A: Given, Mass of carbon dioxide (CO2) = 15.0g Volume (V) = 10.0L Pressure (P) = 3.00 atm R = 0.0821 L...
Q: Make schematic sketches of the microstructures that would be observed for conditions of very slow co...
A:
Q: CH Br CH,CH2C=CH NANH2 1. ( 2 =-CH3 (3) Lindlar's catalyst
A: When sodium amide reacts with alkyne, sodium replaces the terminal hydrogen. Further reacting with a...
Q: Calculate the following DNA concentrations and determine the purity of the samples: DNA Dilution Dil...
A: Formula used :- Concentration of double stranded DNA = 50 µg/mL × OD260 × dilution factor.
Q: Measurements show that the enthalpy of a mixture of gaseous reactants decreases by 308. kJ duríng a ...
A: Let us discuss the reaction and then solve the problem.
Q: Complete the mechanism for the formation of the major species at equilibrium for the reaction of 3-m...
A: Given,
Q: If the sample is compressed to a volume of 11.2 L, what is its pressure? A sample of gas has an init...
A: given :- initial pressure (P1) = 1.17 atm initial volume (V1) = 14.9 L final volume (V2) = 11.2 L ...
Q: The following Rf values were computed from a normal phase chromatography experiment. Arrange them fr...
A: In the normal phase chromatography it contains polar as mobile phase and non polar as stationary pha...
Q: What is the pH of a buffer solution would be if a little amount of acid is added? O basid acidic non...
A: A buffer solution is a solution which resist the change in pH if small amount of acid or base added ...
Q: Calculate the concentration of dissolved sulfate ion in solution if 75.0 mL of a 0.0250 M Ba(NO3)2 a...
A:
Q: A 2.00-L vessel contains 1.00 mol N2, 1.00 mol H2, and 2.00 mol NH3. The reaction is N2(g) + 3H2(g)...
A:
Q: *6. In a redox titration, 12.52 mL of a 0.3264 M KMNO4 solution were required to titrate 1.7832 g of...
A:
Q: Calculate the volume, in L, of water that must be added to dilute 24.6 mL of 10.6 M HCI to 0.105 M H...
A:
Q: The following mechanism has been proposed for the conversion of tert-butyl bromide to tert-butyl alc...
A:
Q: For the reaction conditions given below, what type of product will be produced? Ketone + Zn(Hg) +HCl...
A: Given :- Ketone + Zn(Hg) + HCl → To identify :- Type of product formed in above reaction
Q: The purple ring of Molisch reaction is due to none of the choices Furfural Furfural + α-Napthol α-N...
A:
Q: What are the advantages of performing ignition?
A:
Q: The formula for calculating the energies of an electron in a hydrogenlike ion is given in the follow...
A: For muli electron atom ,z is replaced with z-s. S is shielding constant value.
Q: Br2 1) Mg/ether 2) CH,CH,CH,CHO 3) H,O* H3C. FeBr3 d) CH3 Cro3 H3C. HNO, H,SO4, A H,SO4 e) CH3
A: Both are Multi step synthesis reaction . Detail mechanistic pathway is given below
Q: A loop of wire is placed in a magnetic field of uniform strength 3.0 T that is parallel to the norma...
A: Please find the below attachments.
Q: Determine A to complete the reaction below: CH,OH CH-OH HO A HO H. OH OH OH CH,OH CH-OH open-chain f...
A: Monosaccharide - it is the simplest carbohydrates that cannot be hydrolyzed further to give simpl...
Q: Which of the following reactions is an elimination reaction? A. -CH2-CH, Na OH -CH=CH2 H H. H. CI + ...
A: The basic organic reactions types are addition, elimination, substitution, and rearrangement reactio...
Q: CHy HyC Br Br CH2
A:
Q: I. Modified True or False Directions: Write CHEMISTRY if the statement is correct, if otherwise, cha...
A: Question 1 - True The entropy of the universe is always positive and it is increasing over time and...
Q: Which of the following is an example of a catalyst? dNTP's used as building blocks for a new DNA s...
A: Catalyst which affects the rate of reaction:
Q: *OCH3 Br DMSO HO
A: Charged species of alcohol(methoxide -OCH3) are strong nucleophiles. So which undergoes via SN2 reac...
Q: 云。 00 work 1. Saved Example 7- Molarity -use with mole value to calculate volume (L) value (Pool 1 o...
A: Molarity of sulphuric acid = 1.13 M Number of moles of solute = 0.131 mol Volume of the solution =...
Q: Mark only one oval. formation of white precipitate presence of oxalate ion disappearance of pink col...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: OH CrO H,SO, ОН CH3 f) H3C- Br2 -OCH3- FeBr, g) CH, NBS Na*O-t-Bu A h)
A:
Q: AH = - 1.2 kcal/mol; Kp = 16 nM %3D OH 1 HO AH = - 6.0 kcal/mol; N. Kp = = 76 nM OH 2 करले HOl. AH =...
A: Binding enthalpy Or ΔH for a binding process reflects the energy change of the system when it ligand...
Q: Indicate whether either an increase or a decrease of pressure obtained by changing the volume would ...
A:
Q: Time and concentration data were collected for the reaction 0.52. A products 0.48 0.44- t (s) [A] (M...
A: The change in concentration of a reactant or product in an infinitely small-time interval is known a...
Q: To answer this question, refer to the titration curve shown above and the points labelled A-G. Which...
A: We are given the titration curve for the titration of a weak acid, HA with strong base NaOH. The bal...
Q: What is the pKa of HC4H4O6 ? 2.17 10.26 O 6.59 8.15 4.34
A: generally pka gives the acidic nature of the molecule lower the pka more will be the acidic nature ...
Q: A particular mass of N2 occupies a volume of 1.00 L at –50 °C and 800 bar. Determine the volume occu...
A: Answer: Ideal gas law: PV=nRTPVT=nRhere:P=pressure of the gasV=volume occupied by the gasn=number of...
Q: B. How many grams of Oxygen gas will completely react with 3.8 moles of Propane? C. If 25 g of C3H8 ...
A: In the given question we have to solve the following problem of mole ratio. Given reaction equation,...
Q: Enter your answer in the provided box. Calculate the osmotic pressure of a 2.0 x 101 M solution of N...
A:
Q: 3. Determine the 3D structure of the molecule of formula C2H,O (H3CCHO). Indicate the geometry aroun...
A: CH3CHO - acetaldehyde Contains two carbon single bonded 1is methyl carbon, sp3 hybridised tetrahe...
Q: What fraction of iminodiacetic acid is in each of its four forms (H, A H,A, HA, A) at pH 3.357? The ...
A: pH=3.35=-log [H+]or, [H+]=10-3.35=4.47×10-4
Q: A certain gas obeys the van der Waals equation with a = 0.580 m6 Pa mol–2. Its volume is found to be...
A:
Q: At 77°C, 2.00 mol of notrosyl bromide, NOBr, placed in a 1.00-L flask dissociates to the extent of (...
A:
Q: Mark each statement as True (T) or False (F). i) CO belongs to the binary ionic compounds. ii) React...
A:
Q: These are practice questions. Please help! Thanks.
A: Volume of container = 8 Litre Moles of A = 2 mole Concentration of Compound = Moles of Compound/Tota...
Q: Consider the following data on some weak acids and weak bases: acid base K. name formula name formul...
A: Ka and Kb values for some weak acids and bases are given. Here we have to rank the solution in o...
Q: In a polar reaction mechanism, the atom that gives away electrons in an uncharged nucleophile will e...
A: Given, In a polar reaction mechanism, the atom that gives away electrons in an uncharged nucle...
Step by step
Solved in 3 steps with 3 images

- On the line drawn below, provide the systematic (IUPAC) name for the following structure. Besure to include stereochemistry (R/S, cis-trans, or E/Z) when appropriateOn the lines drawn below, provide the systematic (IUPAC) names for the followingstructures. Be sure to include stereochemistry (R/S, cis-trans, or E/Z) when appropriate. please show each step.1-methylcyclohexene is reacted with the following reactants. Write products. Write which stereochemistry is valid (syn/anti, racemization, antimarkovnikof, etc). 1- Br2/ H2O 2- CH2N2/heat
- Using any necessary organic and inorganic reagents, show how you can carry out the chemical conversions shwon below. Please answer parts a, b, and c.Bromide Bhas normal activity (for a secondary bromide) towards SN1 substitution, but A has much higher reactivity and Chas much lower reactivity.Five-membered aromatic heterocycles with one heteroatom undergo electrophilic substitutions preferentially at the α-position (C-2 and C-5) rather than at the β-position (C-3 and C-4). Explain?
- Which of the following statements about an -NH2 group is FALSE? a. meta director b. activator towards EAS c. increases electron density on the aromatic ring d. stabilizes positively charged intermediates by resonance or inductive effectsDraw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3 + H2O. Explain how these reactions illustrate that syn dihydroxylation is stereospecific.Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the help
- The treatment of isoprene [CH2=C(CH3)CH=CH2] with one equivalent ofmCPBA forms A as the major product. A gives a molecular ion at 84 in itsmass spectrum, and peaks at 2850–3150 cm−1 in its IR spectrum. The 1HNMR spectrum of A is given below. What is the structure of A?Complete the reaction map by providing the answer from A-E. Write the IUPAC name of the products. conc HNO B conc H,SO, B, D FeBrs conc HNO, E AIC conc H2SO. conc H2SO4Electrostatic potential maps of anisole and thioanisole are shown. Which do you think is the stronger acid, p-methoxybenzoic acid or p-(methylthio)benzoic acid? Explain.

