Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter13: Introduction To Symmetry In Quantum Mechanics
Section: Chapter Questions
Problem 13.41E: a Unlike methane, bromochlorofluoromethane (CHBrClF) is chiral. Determine all symmetry elements that...
Related questions
Question
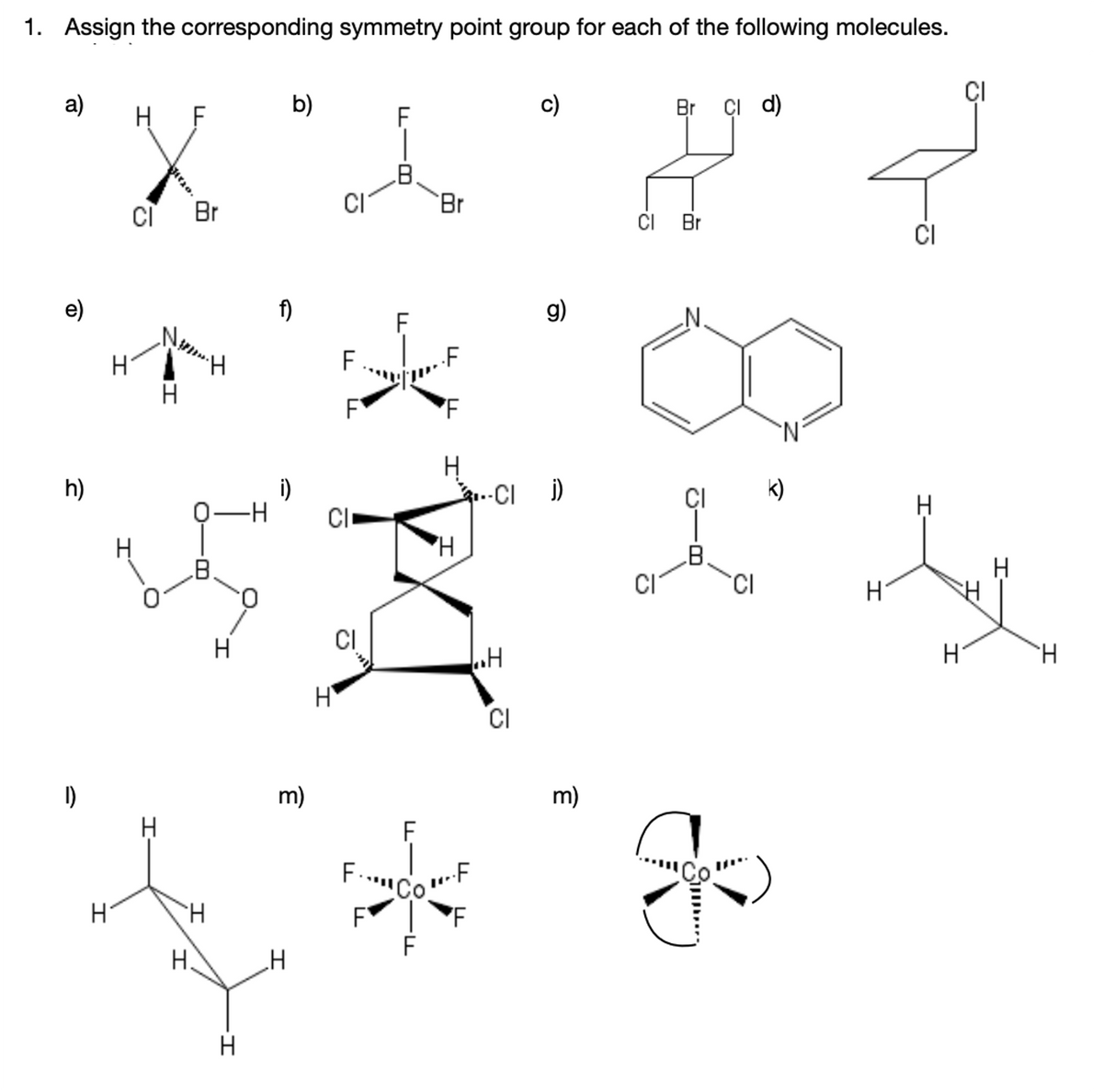
Transcribed Image Text:1. Assign the corresponding symmetry point group for each of the following molecules.
CI
a)
H F
b)
c)
Br çi d)
Br
CI
Br
CI Br
CI
e)
f)
g)
H.
H.
F
h)
i)
CI
..-CI )
ÇI
k)
H.
CI
0.
H.
CI
H
H.
CI
I)
m)
m)
CoF
H.
H.
H
H.
Si..
I'
u-8-
L -B
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning