1. What are the three kinds of enzyme-controlled reactions so that the chemical-bond energy from a certain nutrient is released to the cell in the form of ATP?
1. What are the three kinds of enzyme-controlled reactions so that the chemical-bond energy from a certain nutrient is released to the cell in the form of ATP?
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter25: Nitrogen Acquisition And Amino Acid Metabolism
Section: Chapter Questions
Problem 16P: A Deficiency on 3-Phosphogtycerate Dehydrogenase Can Affect Amino Acid Metabolism Although serine is...
Related questions
Question
100%
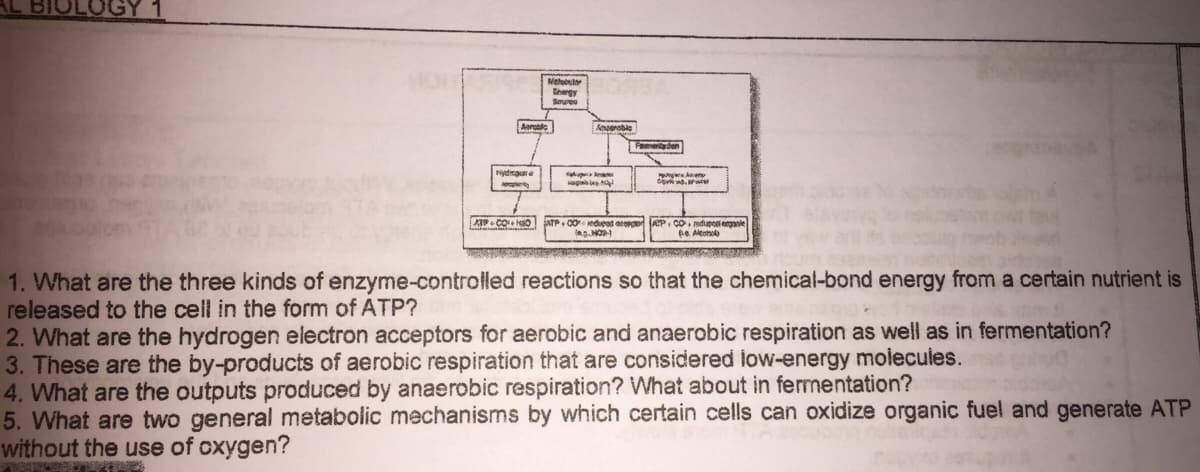
Transcribed Image Text:Metobsh
Shegy
Seyres
Aaretie
Feaden
ydaar e
agalep
ATP C0 edueod eo AP, Co mdupooi eganie
e Alconol
1. What are the three kinds of enzyme-controlled reactions so that the chemical-bond energy from a certain nutrient is
released to the cell in the form of ATP?
2. What are the hydrogen electron acceptors for aerobic and anaerobic respiration as well as in fermentation?
3. These are the by-products of aerobic respiration that are considered low-energy moiecules.
4. What are the outputs produced by anaerobic respiration? What about in fermentation?
5. What are two general metabolic mechanisms by which certain cells can oxidize organic fuel and generate ATP
without the use of oxygen?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning