1. What is the net charge of aspartic acid when the pH is 0.10? 2. Draw the pre-dominant form of aspartic acid when the pH is 2.1 3. Draw the zwitterionic form of aspartic acid. Give the approximate pH value wherein the zwitterionic form exist.
1. What is the net charge of aspartic acid when the pH is 0.10? 2. Draw the pre-dominant form of aspartic acid when the pH is 2.1 3. Draw the zwitterionic form of aspartic acid. Give the approximate pH value wherein the zwitterionic form exist.
Anatomy & Physiology
1st Edition
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Chapter2: The Chemical Level Of Organization
Section: Chapter Questions
Problem 41CTQ: The pH of lemon juice is 2, and the pH of orange juice is 4. Which of these is more acidic, and by...
Related questions
Question
Follow the instructions carefully.
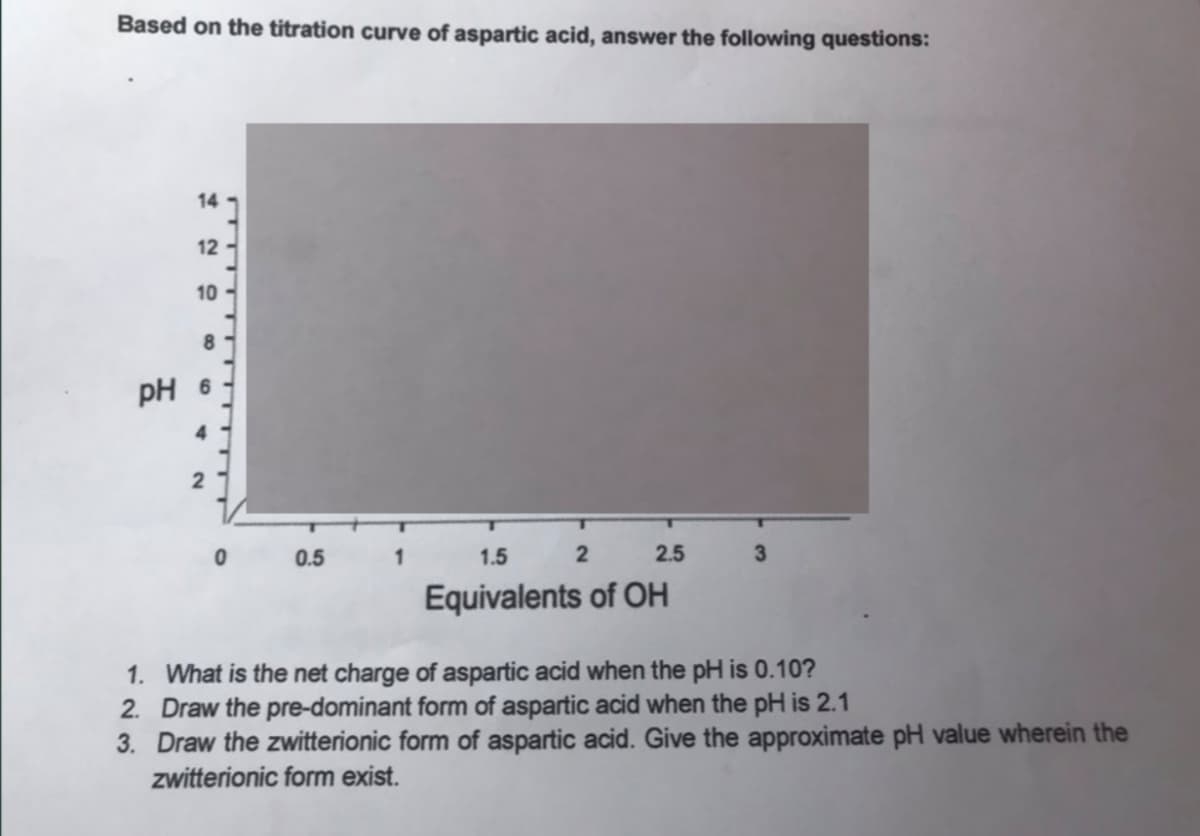
Transcribed Image Text:Based on the titration curve of aspartic acid, answer the following questions:
14
12
10
8
pH 6
0.5
1.5
2.5
3
Equivalents of OH
1. What is the net charge of aspartic acid when the pH is 0.10?
2. Draw the pre-dominant form of aspartic acid when the pH is 2.1
3. Draw the zwitterionic form of aspartic acid. Give the approximate pH value wherein the
zwitterionic form exist.
T T TT
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College