1. What section represents the sold phase? 2. What section represents the liquid phase? 3. What section represents the gas phase? 4. What letter represents the triple point? 5. What letter represents the critical point? 6. What is this substance's normal melting point? 7. What is this substance's normal boiling point? 8. Above what temperature is it impossible to liquity this aubstance no matter what the pressure? 9. At what temperature and pressure do all three phoses coexist? 10. s the density of the sold greater than or less than the density of the lquid? 11. Would an increase in pressure cause tnis substance to freaze or met?
1. What section represents the sold phase? 2. What section represents the liquid phase? 3. What section represents the gas phase? 4. What letter represents the triple point? 5. What letter represents the critical point? 6. What is this substance's normal melting point? 7. What is this substance's normal boiling point? 8. Above what temperature is it impossible to liquity this aubstance no matter what the pressure? 9. At what temperature and pressure do all three phoses coexist? 10. s the density of the sold greater than or less than the density of the lquid? 11. Would an increase in pressure cause tnis substance to freaze or met?
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter6: Equilibria In Single-component Systems
Section: Chapter Questions
Problem 6.60E
Related questions
Question
How do you read the phase Diagram to answer the questions?
Transcribed Image Text:O Phase Diagrams
O Phase Diagram-Freezin x
M My Classes | McGraw-H X
K Phase Diagram-Freezin x
2 Phase Diagrams
b How do you know wha x
+
A lakota.instructure.com/courses/22818/files/4306922?wrap3D1
E School Bookmarks
A Dingle House Irish.
É The College Board.
HAC a
O Knowledge Network
1. What section represents the sold phase?
2. What section represents the liquid phase?
3. What section represents the gas phase?
4. What letter represents the triple point?
5. What ietter represents the crifical point?
6. What is this subatance's normai metting polnt? .
7. What is this substonce's normal boling point?
8. Above what temperature is it imposible to liquity this substance no matter
what the pressure?
9. At what temperature and pressure do all three phoses coexist?
10. Is the density of the sold greater than or less than the density of the lquid?
11. Would an increase in pressure cause tnis substance to freaze or met?
O v 1 4:12
Transcribed Image Text:O Phase Diagrams
O Phase Diagram-Freezin x
M My Classes | McGraw-H x
K Phase Diagram-Freezin x
Phase Diagrams
b How do you know wha x
+
A lakota.instructure.com/courses/22818/files/4306922?wrap=D1
E School Bookmarks
* Dingle House Irish.
6 The College Board..
6 Knowledge Network
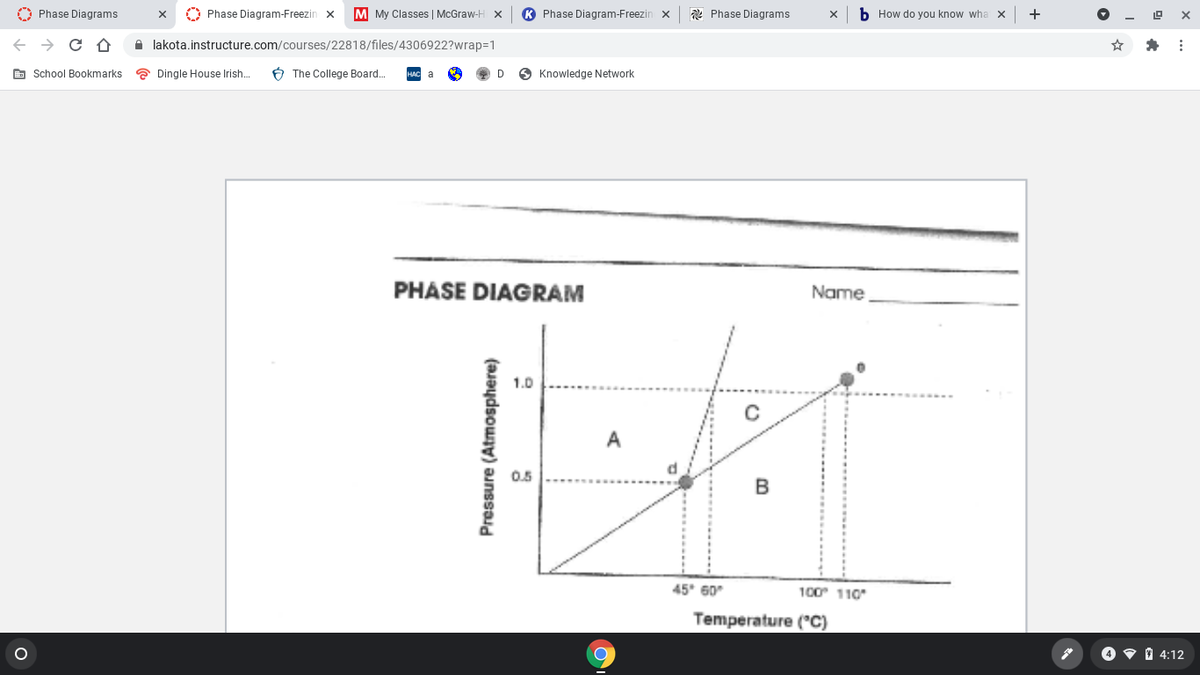
PHASE DIAGRAM
Name
1.0
A
0.5
45* 60
100 110
Temperature ("C)
O V 1 4:12
Pressure (Atmosphere)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,