Q: Use curved arrow notation to show how the first resonance structure can be converted to the second.
A:
Q: Draw in all hydrogens and lone pairs on the charged carbons in each ion. a. b. d.
A:
Q: d)Draw the resonance structures for the following: ÇHa H20
A: The above structure is for a protonated tertiary butanol. For a molecule to show resonance, the…
Q: Hydrocarbon A possesses a significant dipole, even though it is composed of only C-C and C-H bonds.…
A: The given compound contains two cyclic rings. The seven-membered ring contains seven pi electrons…
Q: H. HC= :NH HC : CH2
A:
Q: Part A H;C CH-N-CH, + HBr(aq) ? H3C Draw the molecule on the canvas by choosing b toolbars. The…
A: Ans
Q: B1) Draw all the Resonance structures for Cycloheptatrienyl anion, using arrows show the flow of…
A:
Q: Follow the curved arrows to draw a second resonance structure for each species. a. H-C=0: b. CH-…
A: (a) The resonance structure for given aldehyde is drawn in step two.
Q: Step 1 Draw step 1. Include lone pairs and formal charges in your answer.
A: Given reaction is dehydration of alcohol which happens in the presence of H2SO4 in which H+ and OH…
Q: Follow the curved arrows to draw a second resonance structure for each species.
A: a) Please find below the second resonance structure
Q: H. (+) H3C CH3 CI
A:
Q: In Naproxen, is the lone pair of the oxygen atom delocalize? I’m other words, can the oxygen…
A: In Naproxen, is the lone pair of the oxygen atom delocalize? In other words, can the oxygen…
Q: Considering structures A-D, classify each pair of compounds as isomers, resonance structures, or…
A: Isomers are the compounds having molecular formula different structures. Isomers are various type,…
Q: a) Add curved arrows to show how one resonance structure can be converted to the next
A: To draw resonance structures, the points to remember are: 1) The charge is converted to a bond and a…
Q: BEIOW, Consider the two OH compound A Justify your amswer: which compound do yO PKa : compound A or…
A: Applying concept of inductive effect and mesomeric effect i.e.resonance.
Q: Circle the molecule in each pair that is more water-soluble. 1 (b) (c) VS. VS. VS. M (e) vs. (f) ï ï…
A: We have to select the more soluble compound
Q: What is the resonance hybrid of the anion in the box below N - A В
A:
Q: Determine the correct first curved arrow movement of electrons in the following reaction. OH O A)…
A: In the question, it has been asked to find a possible reaction mechanism for the reaction that will…
Q: Give a reasonable set of resonance structures for each compound below. Use Resonance Arrows, and…
A: Resonance is the partial delocalisation of electron pairs which can be shown by arrows.
Q: 1. Draw curved arrows to show how to get from one resonance structures to the next ( points): A. В.…
A: Please find the attached images for the answer
Q: Add curved arrows to both structures to show the delocalization of electron pairs needed to form the…
A: The given molecule exhibits a continuous delocalisation of pi electrons (resonance).The resonance…
Q: Draw the asked compounds. 1. Cyclohexanone + AlCl3 -------> ? 2. Illustrate the resonance…
A: 1). Cyclohexanone have carbonyl group which have oxygen atom. Oxygen atom have lone pairs on it.…
Q: 7. a) Draw the resonance arrows to form the structure on the right, b) Circle the major resonance…
A: The transformation given is,
Q: Draw in all hydrogens and lone pairs on the charged carbons in each ion ?
A: A molecule in which carbon has a positive charge along with usually three groups attached is known…
Q: Draw a second resonance structure for each radical. Then draw the hybrid.
A: Radical, also called Free Radical, molecule that contains at least one unpaired electron.
Q: This is an organic chem question. draw 2 additional resonance structures for each part a and b
A:
Q: 1) Draw the resonance structures A, B, AND C below. 1) A B + H3C
A: Complete delocalization of pie electron, in a system , it is a interaction of lone pair and pie…
Q: a b CH CH₂ CH₂ CH₂
A:
Q: 4. Circle any n-conjugated portions in the molecules below. Draw all resonance structures for each…
A: Resonance structure comes into account when a single structure of the molecule not able to explain…
Q: ne resonance hybrid a
A: Rules to identify the best resonance contributors are as follows: Greatest number of full octets…
Q: Give a clear handwritten answer...
A: Here we have to write mechanism of the following reactions.
Q: For each pair of molecules, determine whether umpolung (polarity reversal) should be considered to…
A:
Q: (a) Add curved arrows to show how the starting material A is converted to the product B. (b) Draw…
A: Given, the structure of A is: The curved arrows to convert A into B is shown below:
Q: 5. Answer BOTH parts of this question. (a) Name molecules A and B shown in figure Q5.
A:
Q: See the Attachment & solev the followings (a) Add curved arrows to show how the starting…
A: (a) Conversion of A to B
Q: PLEASE Answer all subquestions 1) Draw resonance structures AND resonance hybrid for following…
A:
Q: (a) Label each carbon, oxygen, and nitrogen atom as being sp, sp, or sp hybridized. (b) Circle and…
A: In a carbon compounds the single bonded carbon atoms possess sp3, double bonded carbon atoms possess…
Q: A. Determine whether the curved arrow(s) shown below generate a valid or invalid resonance…
A:
Q: Am I drawing the resonance correctly for each? Thank you
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single…
Q: Draw a second resonance structure for each radical. Then draw the hybrid.
A: Resonance structure is the structure that gives stability to the molecule by the delocalization of…
Q: With reference to compound A drawn below, label compound B as an isomer, a resonance structure, or…
A: Isomer of any compound is the structure with same molecular formula and different arrangement of…
Q: Add a lone pair to NH,. Add a formal charge, Which atoms have a complete octet? if applicable. Rings…
A:
Q: including arrows For the following structures, complete resonance structures electron flow and…
A: The ion given is,
Q: Which species contributes more to the overall resonance hybrid? 00 right equally contribute left
A: Delocalization of pi electron from high charge density to lower charge density is known as…
Q: the reaction below: a. How many non-polar and polar bonds are in the reactants? b. How many…
A: Polar Bonds:- These bonds are present between atoms which have different electronegativity. Non…
Q: 1. Draw the best possible resonance contributor. Draw curved arrows to show electron movement.
A: Resonance structure : Delocalization of π electron density in adjacent conjugated system is called…
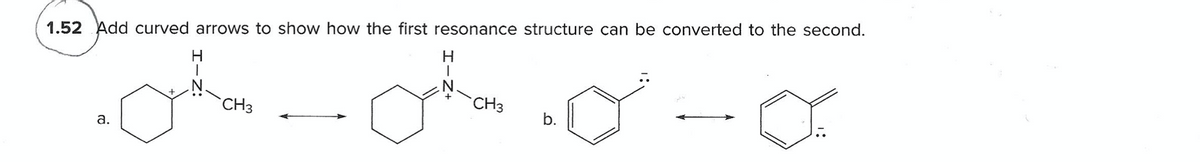
Q: Add curved arrows to show how the first resonance structure can beconverted to the second.
A: The conversion is as given below:
Q: H. C'
A: Rules for drawing resonance structure Only the electrons move, the nuclei of atoms never move and…
Q: P3A. Draw the resonance structures for the following compound and circle the one that contributes…
A: Resonance is the ability of The Pi bonds To Move Its electrons in the compound where it is possible.…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- I am having trouble on part c. It wants me to draw the resonance structure, but in mastering chem there’s no arrows available for some reason . Is there a way to draw it without the arrows and still be resonanceFor each proposed set of resonance structures: a. (E) Add curved arrows (starting from left) to show how each successive r.s. was generated. b. Cross out any resonance structures that are NOT important, and explain your reasoning.This is an organic chem question. draw 2 additional resonance structures for each part a and b
- Follow the curved arrows to draw a second resonance structure for each species.Which of the following species (B, C,D) is a valid resonance of A? Use curved arrows to show how A is converted to any valid resonance structure.Draw the curved arrows to show the resonance structure for the following molecules. ( there are 3 molecules )
- See attached image. For this molecule draw the 2 best resonance structures in the boxes. Circle the best.Add curved arrows to show how the first resonance structure can be converted into the second.Provided is the first and last resonance. Draw the missing resonance forms, then propose the corresonding resonance hybrids of each molecule. Show the movement of electrons using appropriate arrows.

