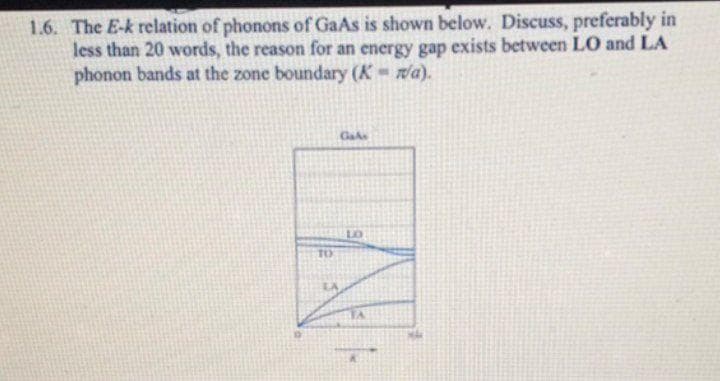
1.6. The E-k relation of phonons of GaAs is shown below. Discuss, preferably in less than 20 words, the reason for an energy gap exists between LO and LA phonon bands at the zone boundary (K a). TO LA
Q: 5) When you consider the voltammetric techniques, how is polarography different than the others?…
A: 5. THE SHAPE OF THE POLAROGRAPHY IS OF SIGMOID SHAPE IT IS ELECTROANALLY TECHNIQUE THAT IS APPLIED…
Q: * Balame Pedox R,D in auidic media -2 +2 → Mn + Co, -2 stiochiometric co-epiciantbof Mol4, soá, Mos"…
A: The reaction given is MnO4- + C2O42- ----------> Mn2+ + CO2 The oxidation number for O is…
Q: For dissolution of dopant ZrO2 into the host lattice of Y2O3, does the following defect reaction…
A: Chemical equation is the equation in which the chemical reaction is represented. All the reactant…
Q: For a Copper-Nickel (Cu-Ni) alloy of composition 65 wt % Ni and 35 wt % Cu at 1350 °C (Point B),…
A: Here we determine the phase composition of the alloy by using a tie-line in the case of the…
Q: 6. If there is one monolayer coverage of organic contamination on the wafer, how much is that…
A: In order to solve this question, we will be substituting one silicon atom with a carbon atom on the…
Q: What are the scientifically proven bioactivity of VCO that is beneficial to human health?
A: As per the guideline, Since you have asked multiple questions, we will solve the first question for…
Q: you've been tasked to compound 30 grams of a 0.5% Zinc oxide ointment. you have a stock preparation…
A: The formula used to calculate the amount need to be dilute is as follows: Amount need to be dilute =…
Q: Which is correct for polarography? I-polarography is an amperometry method conducted with a dripping…
A: Polarography technique is used to determine the molecules are oxidation or other reduction at…
Q: The Ksp values of two solid carbonates, A2CO3 and BCO3, were determined by preparing 250.0-mL…
A: Sparingly soluble salts are those that do not dissolve completely in solution rather maintain…
Q: In ammonia stripping, hitrogen is removed from wastewater by volatilization of NH3. Would ammonia…
A:
Q: A compound Y (150.0 g/mol) in a 1.00 cm quartz cell has an extinction coefficient (ε) of 23,150 at…
A: Answer = 509.12 Complete solution is given in next step.
Q: What is the use of Fisher-Johns apparatus, Gallenkamp apparatus, and automatic melting point…
A: To determine melting point the thermal analysis process is used. This analysis most frequently used…
Q: the mmiscibility gap for a gkiss men? chemitcally expRin the spinodal decomposition of the said…
A: As per the guideline, Since you have asked multiple questions, we have solved the first question for…
Q: Generally, anion exchange resins are negatively charged. Is it true or fasle?
A: This statement is false.
Q: and sheets of thermoplastic polymers. Elucidate the po giving TWO (2) suitable examples. Polymers…
A: polymers used in the production of films and sheets: Polyethylene, poly propylene and polyester.…
Q: analysis of a compound not distinguish between a monomer and dimer of the species? What technique…
A: Monomer is relatively small molecule, they can be binded covolently to other monomer to form…
Q: Provide the correct answers in the given statement with the given compounds in the image. A. In…
A: The reactions are as follows:
Q: explain why it would not be possible to determine the orders with respect to both iodide and…
A: To explain: why it would not be possible to determine the orders with respect to both iodide and…
Q: Give a lab cation separation scheme for Al3+, Ca2+, Ni2+ cations based on Ksp values and complex ion…
A: The detection of ions is done by qualitative analysis. The basic principle is the ions will undergo…
Q: Assume that there was an excess of (L)-isovaline on the Murchison meteorite. Discuss the…
A: Noorduin, et al., have demonstrated the enantiomeric enhancement upon crystallization by using amino…
Q: 3. There is a class of polyesters that have the general structure shown below. . For example, in…
A: The equation which relates the number average molecular weight and glass transition temperature is…
Q: (a) The melting point of NaCI was found to be 801 °C. Suggest, with reasons, whether the melting…
A: Following are the appropriate reason for the relationship between boiling point and bonding.
Q: 3) Figure below shows an x-ray diffraction pattem for single crystal scandium nitride (ScN), having…
A:
Q: The procedure for the synthesis of menthone indicates that you should use 10 cm³ of 6M sulphuric…
A: Molarity (M) :- The number of moles of solute dissolved in one litre of solution is defined as…
Q: 5. What is number average and weight average molecular weight give equations with molecular weight…
A: Answer - Number-average - number average and weight average molecular weight is that the number…
Q: What kind of differences are there in crystallizing in unit cell with 1. Chiral (enantiomerically…
A: A chiral compound is distinguished from its mirror images as these are non-superimposable. A chiral…
Q: hedict the poduct that will be frimed by the neachon below. Colzlag) + Mn (5)>
A: Here aqueous cobalt iodide CoI2 and solid manganese Mn have been reacted. We have to predict the…
Q: 2. How will the melting point of your semicarbazone derivative be af- fected if the derivative is…
A:
Q: please explain the solid solution to me completely and comprehensively. preferably with shape and…
A: Solid solution, mixture of two crystalline solids that coexist as a new crystalline solid, or…
Q: why is lupac for Hg2Cl2 Mercury (I) Chloride, instead of Mercury (II) Chloride?
A: Given, Molecules is Hg2Cl2 Why is IUPAC for Hg2Cl2 Mercury (I) Chloride, instead of Mercury (II)…
Q: You have 3 compounds (A, B, C) in your samples. Their polarity A > B > C. You want to separate these…
A: In HPLC technique, the separation of components on basis of polarity, size, charge takes place. The…
Q: Is a mobile phase composition 40:60 of acetone:hexane more polar than a mobile phase composition of…
A:
Q: Based upon a study of the iterature, discuss Ginberg's polarization then as it applies to the…
A: The ability of certain ligands to increase the rate of ligand substitution when positioned trans to…
Q: Discuss how the hollow cathode lamp can increase AAS selectivity.
A: Given : AAS selectivity To find : Hollow cathode lamp can increase the AAS selectivity Solution :…
Q: With reference to the polymerisation mechanisms explain why the polydispersities of the two samples…
A: Polydispersity index is defined as the ratio of weight average molecular mass and number average…
Q: 7. The lattice constants of GaAs:Px alloys are continuously tunable from 5.43 Å (pure GaP) to 5.65Å…
A: Given, the lattice parameter of pure GaP is 5.43 angstorm.the lattice parameter of pure GaAs is 5.65…
Q: Calculate the (Ex/GS) atomic ratio for 3s---3p (oppose) in Mg* at 3000k?
A: Given that For Mg+ Temperature = 3000K Orbital --- 3s - 3p
Q: Write the significances of Poly Dispersity Index (PDI) and calculate the PDI for given data. Number…
A: Ploy Dispersity Index (PDI) tells us the distribution of molecular weights in a polymer. It is given…
Q: Explain 'why glass cells are not used in, UV-spectroscopy.
A: To explain: Why glass cells are not used in UV-spectroscopy.
Q: Consider the mineral dissolution reactions below: FeSmackinawite -> Fe2+ + S2- CuScovellite ->…
A: Given: ΔGo of Fe+2 = -78.8 kJ/mol ΔGo of S-2 = 85.7 kJ/mol ΔGo of FeS = -100.4 kJ/mol…
Q: Consider the following series of poly(phenylene oxide) polymers A–D, and their glass transition…
A: The estimation of Tg depends on the versatility of the polymer chain - the more stable the chain,…
Q: You have ZrO2-Nb2O5 composite and HfO2-Ta2O5 composite. Can you distinguish the oxide phases in the…
A: ZrO2 ceramics used in the it is mixed with usef to varous appications. ZrO2 contains Nb2O5 they…
Q: (c)Explain why glass cells are not used in, UV-spectroscopy.
A: Uv or ultraviolet spectroscopy is a kind of absorption spectroscopy where light of wavelength…
Q: The K2S208 product may be contaminated with K2SO4 or KHSO4. Suggest a method for determining the…
A: PREPARATION OF K2S2O8: The preparation reaction of potassium peroxydisulfate is takes place with the…
Q: state the three different modes of GC/MS, and explain more for each.
A: GC-MS refers to Gas chromatography-Mass spectroscopy. It is an analytical method that combines the…
Q: Mesityl oxide show two band at 230 and 327 but its isomer does not show any band.
A: Mesityl oxide is colorless, volatile liquid having honey like order. It is an a α,β -unsaturated…
3
Step by step
Solved in 2 steps

- Consider the following series of poly(phenylene oxide) polymers A–D, and their glass transition temperatures. Explain how the differences in structure can account for the differences in TgWhat is the Madelung constant for MgO (rock salt) structure? Include only the first 3 terms. Give your answer to 3 significant figures.why is lupac for Hg2Cl2 Mercury (I) Chloride, instead of Mercury (II) Chloride?
- Discuss glass transition temperature and the factors affecting GTT and what are its significances. Give detailed answer please3..Suggest how to make syndiotactic/isotactic polystyrene on the one hand and on the other hand how to synthesize atactic polystyrene. Justify your statement.The UV–visible spectrum of GLB-1 is given in Figure 2. i.What colour(s) will the deoxy and oxy forms of GLB-1 be? Note, an electromagnetic spectrum is reproduced in the Assessment area on the S315 website. ii.By comparing the bands on the right-hand side (around 550 nm) with those of oxy- and deoxymyoglobin, suggest what type of transition gives rise to these bands.
- How much is the energy required for vacancy formation for Pb at its melting temperature of 620.6°F if the fraction of atom sites that are vacant is 2.41x10-5? Solve LegiblyUsing a simple infinite barrier approximation, calculate the “effective bandgap” of a 10 nm GaAs/AlGaAs quantum well grown along [001] direction. If there is a one-monolayer fluctuation in the well size, how much will the effective bandgap change? This example gives an idea of how stringent the control has to be in order to exploit heterostructures.We have a pink crystals of an ionic compound of an f- block element (M). The crystals house the octahydrate [ M(H2O)8]3+ and an anion which is not chloride and does not luminesce under UV. (A) First, draw and label a plausible solid-state geometry for the hydrated cation. (B)Provide ALL potential identities for the f- block element, M, based on the information supplied above, defending your choices and discussing the applicable chemical concepts. (C)What are the potential solid state MCI3 geometries and coordination numbers for ALL of the components proposed in section (B)? Explain briefly why, focusing on the key chemical principles. (D) In ion - exchange chromatography, which of the trivalent f-block ions indicated in component (B) would you anticipate to elute first? Explain your response, emphasising the important chemical concepts.
- 1.Why are the chlorophylls less mobile than the carotenes on the TLC plate? Give athorough explanation, including a sketch of how each type of molecule would interactwith the silica stationary phase material (you can use schematic structure formulas, ratherthan draw the whole molecules, which are quite large!). For instance, would one of thosetypes of molecules create more intermolecular force interactions than the other?Compute the repeat unit molecular weight for nylon 6,6Pls do Asap...! And give brief explanation ...!

