Q: Calculate the difference between AH and AU when 1.0 mol Sn (gray) of density 5.75 g cm³ changes to…
A: Enthalpy is a thermodynamic system attribute that is defined as the sum of the system's internal…
Q: What is the coefficient the should be placed in front of iron (II) oxide (Fe203)?* LizO+ FeCb LİCI +…
A: We have given the incomplete reaction which is Li2O + FeCl3 → LiCl + Fe2O3…
Q: Is the Cu detectable at a concentration of 0.10 mg/L in Flame-AAS? Please use data given.
A: Flame AAS is atomic absorbation spectroscopy. Which is used to detect the various sample i. e.blood…
Q: Why is it necessary to buffer a solution with sodium acetate in spectrophotometric determination of…
A: The UV-VIS spectroscopy is the most useful technique for the quantitative determination of various…
Q: :- tlene you able to see the borticles in the miacture f any CA, R&c) E- Hawe beam Jaf light vinible…
A: True Solution: A true solution is a homogeneous mixture of solvent and solute. Example: NaCl…
Q: In a cavity the total energ y U = aVT"i where d is îndependent of V and T. Show that F= + Tf (v) dnd…
A: Answer is explained below. There are some errors in the question, please follow the answer.
Q: An atomic absorption method for the determination of the amount of iron present in used jet engine…
A:
Q: Magnesium in 50ml of blood was extracted and mesured using falame emission AES An emission E 06 was…
A: In flame AES, the signal is proportional to the concentration of the analyte in the sample.
Q: Calculate the lowest energy Δo for a solution
A: Orgel diagram is only applicable for weak field ligands and it shows the relative energies of…
Q: (a) Illustrate a respective diagram showing Interstitials impurity and Frenkel defect.
A:
Q: 4) How many mmoles of SCN ion are there in 1.0 mL of 0.00200 M KSCN?
A: Recall the following expression of milimoles(mmoles) as follows Milimoles=Volumein…
Q: A solution of X of concentration 0.060 mol L gives an absorbance reading of D.65. What rancentration…
A: We will solve this question using beer- lambert law.
Q: Figure caption: EDS spectrum for a typical metal-rich spherule, K and L lines for noted elements are…
A: From EDS we can get the idea about the element present and it's amount . From here we can calculate…
Q: List as many experimental as you can it may be used to determine KSP for sparingly so you both salt.
A: Solubility product connects the species' solubility in saturated solution. The increment in any…
Q: Consider the data for the determination of phosphorus in urine as the phosphomolybdate complex,…
A:
Q: 1. Fill up the necessary "lettered" cells based on the properties given. Units must be based on the…
A: Ideal gases: Those gases that obey the ideal gas equation are said to be ideal gases. It is a…
Q: Phosphorus in urine can be done by treating with molybdenum (VI) and then reducing the…
A: To plot the corrected absorbance as a function of the concentration of P standard.
Q: The disadvantages of graphite furnace atomic absorption spectroscopy over flame atomic absorption…
A: Two important techniques for sample atomization are flame atomization and graphite furnace…
Q: solution needed to neutralize 500.0 mL of 0.30 M Ca(OH)2? 2.20 g NaOH are dissolved in-water 500 mL.…
A: Electronic configuration is defined as arrangement of electrons in shell , subshell and orbitals in…
Q: (b). Atomic abs orption spectroscopy (AAS) is used for the quantitative determination of chemical…
A: A question based on AAS, which is to be accomplished.
Q: If the equation produces from data is y = 0.343x - 1.021 and the absorbance for the unknown is…
A: Given data: Equation obtained from graph, y = 0.343x – 1.021 Absorbance (A) = 0.386
Q: Nickel bis-dimethylglyoximate complex was depősited with a weight of (4.869g) with the filter paper…
A: % of Ni ions = ? Mass of complex + filter paper = 4.869 g Mass of filter paper = 1.30 g
Q: Explain why Anodic Stripping Voltammetry (ASV) is considered superior over analytical techniques in…
A: Anodic stripping voltammetry is one of the most common analytical techniques for determination of…
Q: TA 100 mL natural water sample has very small concentration of Ag* (1x10-7 M). Is it possible…
A:
Q: Explain the effect of donor concentration (doping level) and temperature on electron mobility in…
A: Please find your solution below : The electron mobility tells that how quickly and electron can move…
Q: The alkali and few alkaline earth metals impart colour to a flame, a property used in the flame test…
A: The alkali and alkaline earth metals impart colour to a flame because of low ionization energy of…
Q: Calculate for the molar absorptivity coefficient of a 0.55 microgram per liter Cr(III) chelate…
A:
Q: How resolution of TOF can be improved?
A: We use TOF, time of flight, analysers to separate ions by time using magnetic or electric field.
Q: The detection limits reported for Aluminum with ICP-AES and GFAAS are 2 ppb and 5 pg, respectively.…
A: Because of its longer seeing path, axial view has larger detection limits. It's also less linear and…
Q: (a) Why is carbon used as an electrode in arc and spark sources? Explain briefly. (b) Compare…
A: Carbon used as an electrode in arc and spark sources: If we pass the high voltage of electric…
Q: 3. (a) Draw the variation of signal strength with applied voltage for a-particles and explain. (b)…
A: This Question is based upon Nuclear Chemistry.
Q: 1. Caleulate th equilibrium constant at 2s E For thereaction: 2H"+ " + Dacg) = Hzegs2D From the…
A: GIVEN:-
Q: A spectrophotometric method for the quantitative analysis of Ni2 in pharmaceutical formulation…
A: Since you asked multiple questions, we will answer only first question for you. In order to get…
Q: liter neces- 1. Calculate the minimum concentration of oxygen in milliliters sary to passivate iron…
A: Solution: In the given question we have to calculate the minimum concentration of oxygen.
Q: Use the Debye-Hückel equation to calculate the activity Ion Ion size (a, pm) coefficient (y) for…
A:
Q: Why is it necessary to buffer the solution with sodium acetate in the spectrophotometic…
A: To explain the reason of buffering the solution with sodium acetate in the spectrophotometric…
Q: Points to discuss: • Discuss the relationship between the concentration of the standard solution and…
A: Points to discuss: • Discuss the relationship between the concentration of the standard solution and…
Q: A solution of X of concentration 0.010 mol dm-3 gives an absorbance of 0.5. What concentration is a…
A: The Beer-lambert law is: A=εcl ......(1) Here, A is absorbance, c is concentration, ε is molar…
Q: -Explain The relation between the concentration and absorption based on beer's law in details.
A: Beer's Law According to this law, Absorbance is directly proportional to the path length and the…
Q: Make an accurate plot of Feversus a for a Mg" - Oʻpair. Consider the range of a from 0.2 to 0.7 nm.…
A:
Q: given that [Fe2+] = 0.60 M and [Cd2*] = 0.010 M? %3D %3D
A:
Q: 25 substarce D1toF(KS/mel) G(KJ(mcl) s°(SImel.Kl) P2 PC13 g POCsg Clay 144.3 -2881 -542.2 218.1…
A: Given: So(P2(g)) =218.1 J/mol.K So(Cl2(g)) =222.96 J/mol.K So(PCl3(g)) =311.7 J/mol.K
Q: Calculate the concentration if the molecular weight of compound 45 gram. mole , abs orptivity 14.6…
A: Introduction: This can be calculated from Beer's Lambert law. The law states the following equation,…
Q: A solution containing 0.00400 molar solution of ligand D had a percent transmittance of 33.3 in a…
A:
Q: What is the value of [S] with v = 0.5Vmax?
A:
Q: How inter- H-bonding can be ascertained by IR specctroscopy? and intra-molecular
A: A hydrogen bond is the electrostatic force of attraction between a hydrogen atom and a more…
Q: Q1: find the miller induces and d distance between plans if crystal constant for Si is 5.431 A
A: Miller indices is the raceprocal of the intercept of the axis .
Q: Show the PRODUCTS of the reaction that represents dissociation of PbCl2is) into its component ions.…
A: The dissociation occurs when a substance is put into a solvent, the substance dissociates and forms…
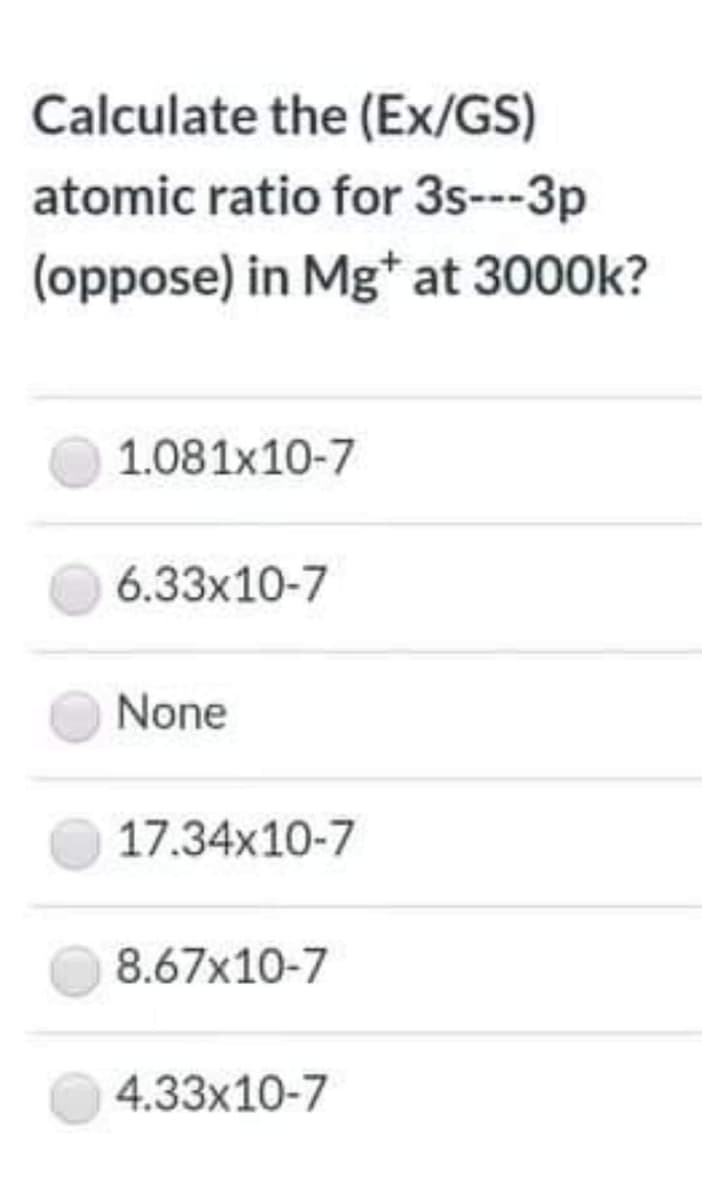
Step by step
Solved in 2 steps with 1 images

- 5. Calculate the Ionic strength of solution prepared by mixing of 0.2 M NaNO3and 0.02M of K2Cr2O7:a) s = [Ksp / 27]1/5 b) s = Ksp1//5c) s = Ksp ½ d) s = [Ksp/108]1/5e) s = 108 KspNickel bis-dimethylglyoximate complex was deposited with a weight of (4.869g) with the filter paper from the base medium from adding (10ml) of hot DMG to a salt of nickel chloride at weight (0.78g), find the percentage of nickel ion. Note that the weight of the filter paper (1.30g). At.Wt.: Ni= 58.69 g/mol, C= 12 g/mol, O=16 g/mol, H=1 g/mol, N=14 g/molwhat is the LFSE for [Fe(CN)6]4-? splitting parameter is 32800cm-1 pairing energy is 17600 cm-1
- Is the Cu detectable at a concentration of 0.10 mg/L in Flame-AAS? Please use data given.List as many experimental as you can it may be used to determine KSP for sparingly so you both salt.The accompanying data (1.00-cm cells) were obtained for the spectrophotometric titration 10.00 mL of Pd(II) with 2.44 10-4 M Nitroso R(O. W Rollins and M. M. Oldham, Anal. chem .,1971, 43, 262, DOI: 10.1021/ac60297a026). Calculate the concentration of the Pd(II) solution, given that the ligand-to-cation ratio in the colored product is 2:1
- Nickel bis-dimethylglyoximate complex was deposited with a weight of (2.721g) with the filter paper from the base medium from adding (10ml) of hot DMG to a salt of nickel chloride at weight (0.4g), find the percentage of nickel ion. Note that the weight of the filter paper (0.78g).In the development o f an analytical method for the quantitative analysis o f iron using Atomic Absorption spectrophotometry (AAS), a linear calibration curve for standards o f 0.00, 5.00, 10.00, 15.00 and 20.00 ppm was prepared. An iron ore sample with an expected iron content o f 40-60% w/w is to be analysed by this method. An approximately 0.5-g sample is taken, dissolved in a minimum amount o f concentrated HC1 and diluted to 1 L in a volumetric flask using distilled water. A 5.00-mL aliquot is removed with a pipette. With explanation, to what volume (10, 25, 50, 100, 250 500 or 1000 mL) should the 5.00 mL aliquot be diluted, to minimize the uncertainty in the analysis?Calculate the Molar absorptivity constant of a this metal complex solution read at 510 nm using a 50 mm cuvette. C = 1.00 mg/L as Fe Abs = 0.967 (Write answer in whole number, with no decimal and unit)
- (d) By using the data provided below, calculate the PillingBedworth ratio for the nickel (Ni) – nickel oxide (NiO) system and determine whether NiO is protective against corrosion. Atomic mass of nickel = 58.7 g.mol-1 Density of nickel = 8.91 Mg.m-3 Molecular mass of nickel oxide = 74.7 g.mol-1 Density of nickel oxide = 6.67 Mg.m-3 (e) A stainless steel pressure vessel that has fracture toughness, K1C, of 197 MN.m-3/2 is going to be exposed to a stress of 398 MN.m-2 . By using this information explain how designers can determine the thickness of the vessel so that catastrophic failure can be avoided.The molar absorptivity for cobalt and nickel complexes with 2,3-quinoxalinedithiol are 3.64x104L/M-cm for Co at 510nm, 5.52x103L/M-cm and 1.24x103L/M-cm for Co at 656nm and 1.75x104L/M-cm for Ni. A 250mg soil sample that was collected near an industrial metal-processing plant is dissolved, and masking agent is added to avoid interference from other metals present. Color is developed by adding the complexing agent and the sample is diluted to 100.00ml. The absorbance at 510nm is 0.517 and at 656nm it is 0.405 in a 1.00cm cell. Calculate the weight percent of Co and Ni in the soil.Calculate for the molar absorptivity coefficient of a 0.55 microgram per liter Cr(III) chelate solution that exhibits an absorbance of 0.9765 at 590 nm measured in a 1.00 cm cell.





