10/11 21 85% ppun21@ole.augie edu Highest Melting Point CaCl NH,OH 1 PCI3 KI 2 Cl2 CH4 * SUBMIT ANSWER 4 VIEW SOLUTION < 06/20 > F 20 QUESTIONS COMPLETED 56°F Cloudy rch DELL 立
Q: 1400 Mo [D] H. [C] Pressure (in mm Hg) 1000 [A] 800 600 400 [B] E. 200 - 20 60 100 140 180 260…
A: Melting point is defined as the temperature at which a solid substance melts. In the given phase…
Q: ess bi2 2. NaNH2, heat 3. HBr (excess) 1. iso-butyl iocde 2. Na, liquid NH3 3. CH3CO3H, then H3O* Li
A: Given are organic reactions.
Q: The elevation in boiling p CuC I, in 1 kg of water u: (molecular weight K%30.52 K mol-1) pe
A:
Q: Properties Nitric oxide Density (kg/m3) 1.34 Boiling Point (degree celcius) -152 Freezing point…
A:
Q: Predict the type of intermolecular forces that will be formed with the same kind (#1-3) and…
A: Intermolecular forces : The attracting and repellent forces that develop between the molecules of a…
Q: F. 150 135 120 105 90 75 C 60 45 30YA 15 Heat (kJ) Created by E. Lee for Virtual Virginia (2021)…
A: This curve is the example of heating curve. This is because in y direction temperature is increasing…
Q: 8- Arrange the following compound according in increasing melting point: KCI, He , H2O, HF A) Kcl >…
A:
Q: 120. 100. - 80. erature (°C) 60. 40. 20. 10. heat added (kJ/mol) 0. 20. 30. 40. aph to answer the…
A: As we know,at this point, melting of solid takes place.
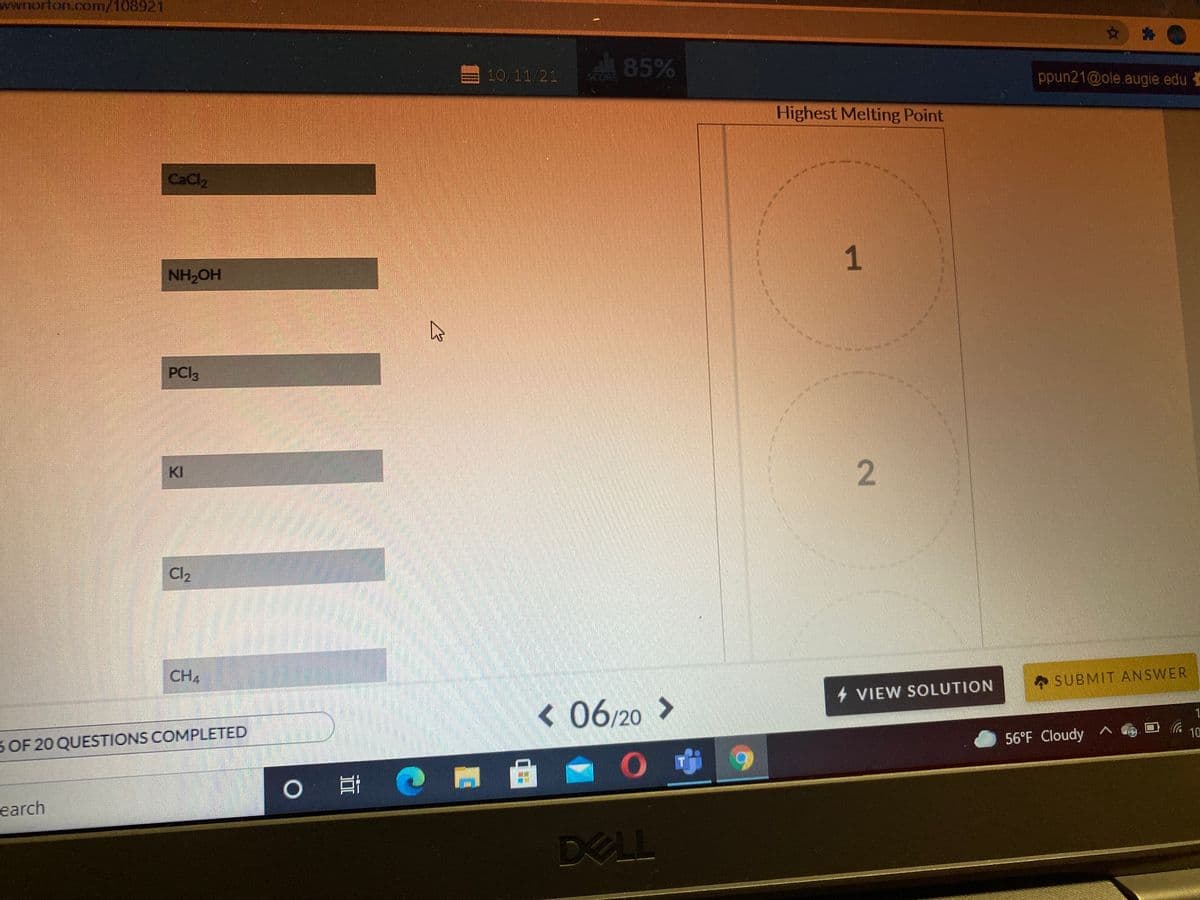
Q: ali 83% ppun21@ole. COR Melting Point Ranking Highest Melting Point CaCl2 2. KI PCI3 CH4 HO HN Cl2 4…
A: The temperature at which solid is changed into liquid is known as melting point of that substance.
Q: 5. In the Dumas method for determining the molar mass of an unknown volatile liquid, a sample of a…
A: Given that, the molar mass of an unknown liquid is determined by the Dumas method. The volume of the…
Q: h of the following compounds has the highest boiling point? hun 2 Не 4000 on 10 aton gen 4 Be B cN F…
A: Boiling point of compound is depends upon intermolecular interaction between molecules and structure…
Q: X. Rank in order of increasing melting point using the less than (<) inequality symbol. Label the…
A: Melting point of a compound depends on the length of the compounds , type of functional groups…
Q: CC x M Inbox O Post Atte x M MyOpen x M MyOpenh x Petition f X Watch the X Sy 2+2 - Ste x My Home x…
A: a) Compound having the greater value of boiling point has strong intermolecular force of attraction.…
Q: Calculate the amount of energy in (k]) given off to convert 346g of H20(g) at 182°C to H20(I) at…
A: Since the boiling point of water is 100 0C Hence the gaseous water will be first cooled to 100 0C…
Q: What is the direction of water in the set up? * 3 5 7 from 5 to 7 from 7 to 5 from 2 to 9 O from 9…
A: Since you have posted multiple questions, we are entitled to answer the first only. The experimental…
Q: 8- Arrange the following compound according in increasing melting point: KCI, He , H2O, HF A) Kcl>…
A: With increasing in the intermolecular interaction, melting point also increases as more energy is…
Q: 6. Complete the following table: Clussfications M-renaetal Ianie moleclac, Citape metullic Substance…
A: The physical state of an element depends on the nature of the substance. The physical state of the…
Q: Given the following data, what is the boiling point of CCI4 in °C? Round to the nearest integer (no…
A: The thermodynamic data given is,
Q: Aail ?Which one of the followings possesses melting point below 0 Phosphorus .a O Carbon .b O…
A: Nitrogen is the correct answer.
Q: 7. Melting points of Br2 and ICl are - 7.2 and 27.2°C, respectively and their electrons are same.…
A: Both Br2 and ICl are isoelectronic, but their melting points are different.
Q: dispersion hydrogen bonding ion-dipole dipole-dipole Motor oil (nonpolar) Lard (nonpolar) Potassium…
A: Motor oil is non-polar in nature. So in between a non-polar solute and non-polar solvent, dispersion…
Q: 2-lf you have 600 kJ of heat to spend, and you use it to melt lead, how many % ounce wheel weights…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: The heat of fusion of some component A at it normal melting point 242 K is 3.62 cal/g. The densities…
A: The relation between heat of fusion and pressure is shown below: P2 -P1 =∆Hfusion∆VfusionlnT2T1…
Q: 1. If a solid line represents a covalent bond and a dotted line represents intermolecular…
A: Hydrogen bonding is the Bonding, only formed between highly Electronegative elements like Fluorine,…
Q: Calculate the total heat required in the heating and complete melting of 1.00 mol solid Cu (MM =…
A: The mass of copper is = 1.00 mol The initial temperature of copper is = 100.0oC The final…
Q: Which of the following compounds has the highest boiling point? 2 H Не thum bo cton fue 10 Li Be F…
A: The boiling point is a physical property, that is, it will not change the chemical structure of the…
Q: Which of the following has the highest boiling point? O 0.20 m Na3PO4 O 0.45 m CH3CH2OH O 0.15 m…
A: The elevation in boiling point of any solution is given by elevation in boiling point = i X Kb X m…
Q: Identify the point(s) on the following diagram where the removal of heat will cause some of the…
A: Condensation is the gas to liquid
Q: Carbon tetrachloride was a common nonpolar solvent that is now being phased out due to carcinogenic…
A: Given: The heat added is 7.40 J. The initial volume of carbon tetrachloride is 39 mL. Initial…
Q: Methane burns and give off 192 Kcal/Mol, assuming 90% effeciency in heat transfer. what mass in KG…
A: Answer: In the process of heating up ice, there will be three steps, 1. Heating of ice up to its…
Q: the data in the table. Compound Melting point ("C) AH (kJ/mol) Boiling point (C) AH,ap (kJ/mol) HF…
A:
Q: Why is the boiling point of the halogen in each period greaterthan the noble gas?
A: The reason for ,the boiling point of halogen in each period is greater than noble gas has to be…
Q: The melting point of H2O(s) is 0 °C. Would you expect the melting point of H2S(s) to be greater or…
A: Intermolecular forces are the attractive force that withholds two molecules and the influence of…
Q: unknown liquid (MM = 83.21 g/mol) at 19.2 °C to a gas at s (g) 0.932 J/(g-°C) 93.5 °C? T. 57.3 °C AH…
A: We have to given three times heat. first we have to raise the temperature from 19.2°C to 57.3°C…
Q: TRUE OR FALSE 1. All the elements from left to right and bottom to up have the lowest…
A: 1. False Explanation - Electronegativity increases as we move from left to right and bottom to up…
Q: 3. Lauryl alcohol is obtained from coconut oil and is used to make detergents. A solution of 5.00g…
A: Given: mass of lauryl alcohol= 5.00g mass of benzene = 100 g = 0.100 Kg Boiling point of benzene =…
Q: I need help with question number 1
A: London Dispersion Forces: Weak intermolecular forces due to the formation of dipoles between…
Q: What happens to the equation AG = AH - TAS when the temperature is 0 K? %3D a) When T-0 then AG = AS…
A: ∆G is a thermodynamic function which is used to find the spontaneity of the reaction.
Q: 13. Which of the following compounds would be expected to bave the highest melting point? a) NC,…
A: The ionic compounds are held together by electrostatic force ,that is the two oppositely charge ions…
Q: 11. Complete the chart. **You should not have the same answer for all the IMF in the last column**…
A: The Lewis structures were given by Lewis who depicts the bonds and electrons present in the…
Q: Which of the following compounds has the highest boiling point? H Не Ihm bern bee 10 Li Be N F Ne…
A: Please find your solution below : The intermolecular forces in alcohols is hydrogen bonding which is…
Q: Octane(C8H18) is a hydrocarbon found in crude oil. Thermochemical data for octane is provided below.…
A: The phase of a substance changes when external energy or heat is provided to it. The value of…
Q: Arrange the following from the lowest melting point (1) to the highes melting point (4). HF [ Choose…
A: The melting point depends on the inter molecular forces between the molecules. Higher the inter…
Q: Diethyl ether (molar mass 74.12 g/mol) boils at 34.6 °C and water (molar mass 18.02 g/mol) boils at…
A: a. At 25 degree Celsius, diethyl ether has higher vapour pressure than water.
Q: Evaluate the boiling point temperature for benzene (CgHg) at 350 atm, given that benzene has the…
A:
Q: Given the following experimental data regarding "determination of molar mass of volatile liquid":…
A: Given the following experimental data regarding "determination of molar mass of volatile liquid":…
Q: nsider the data in the table. Compound Melting point (°C) AHfus (k.J/mol) Boiling point (°C) AH,ap…
A:
Q: Which of the following would have a higher boiling point: NaCl or Cl2? Explain your answer.
A: The boiling point is the temperature at which the vapor pressure of a substance becomes equal to the…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- a concentrated H2SO4 has the following composition specific gravity 1.84 and purity of H2SO4 is 97 percent by weight. express in mole/LConsider the data in the table. Compound Melting point (°C) ????? (??/???)ΔHfus (kJ/mol) Boiling point (°C) ????? (??/???)ΔHvap (kJ/mol) HFHF −83.11 4.577 19.54 25.18 HClHCl −114.3 1.991 −84.9 17.53 HBrHBr −86.96 2.406 −67.0 19.27 HIHI −50.91 2.871 −35.38 21.16 Using the data in the table, calculate Δ?fus and Δ?vap for HF. Δ?fus= J/(K⋅mol) Δ?vap= J/(K⋅mol) Determine the entropy change when 8.70 mol HF(g) condenses at atmospheric pressure. Δ?= J/KGiven the following mixture of two compounds 30.00 g of X (liquid) (MW =58.00 g/mol)(density 1.088 g/mL) and 880.00 g of Y (50.00 g/mol))(density 0.808 g/mL), calculate the mass percent of X. Given the following mixture of two compounds 35.00 mL of X (MW =80.00 g/mol)(density 0.945 g/mL) and 735.00 mL of Y (80.00 g/mol))(density 0.810 g/mL). The vapor pressure of pure Y is 47.00 torr. Calculate the vapor pressure of the solution.
- Melting point (Tm): -94.9C Boiling point (Tb): 110.7C a.) Using the data, find the vapor pressure of toluene at 20C in units of mol/L b.) What is the enthalpy of vaporization (∆?vap ) for toluene in kJ/mol?10.11/ A 1.5-kg specimen of a 90 wt% Pb-10 wt% Sn alloy is heated to 250°C (480°F), at which temperature it is entirely an a-phase solid solution . The alloy is to be melted to the extent that 50% of the speci- men is liquid, the remainder being the a phase. This may be accomplished either by heating the alloy or changing its composi- tion while holding the temperature con- stant. (a) To what temperature must the speci- men be heated? (b) How much tin must be added to the 1.5-kg specimen at 250°C to achieve this state? 10.15) A magnesium-lead alloy of mass 5.5 ka consists of a solid a phase that has a compo- sition that is just slightly below the solubility limit at 200°C (390°F). (a) What mass of lead is in the alloy? (b) If the alloy is heated to 350°C (660°F). how much more lead may be dissolved in the a phase without exceeding the solubility limit of this phase? 10.27 A hypothetical A-B alloy of composition 55 wt% B-45 wt% A at some temperature is found to consist of mass…Show calculations for the experimental percentage of water for Trial 1Percentage of Water in Hydrate Average
- a. How much methylcellulose 1500 should be weighed? viscosity for a suspension is 750 cps. Note 25-50% of the Rx volume comes fro cherry syrup : the remainder of the volume come from the methylcellolose. b. what volume of water should be added to hydrate the methylcellulose?What happen to the separation raid (RS) I change the mobile phase from methanol/water 70/30 till methanol/water 80/20. How does polarity affect?solve as neatly as possible and show completesolution. Round your final answer to 4 decimal places and box / highlight all final answers. and all values must include proper units with proper conversion if needed in your solution. kindly folllow the format Given,Required,Solution 8. At 250C, the interfacial tension between water and n-octyl alcohol is 8.50 dyne/cm. If the surface tension of n-octyl is 27.53 dyne/cm and that of the water is 72.75 dyne/cm, predict whether water spreads on the alcohol by calculating a) Work of cohesion of n-octyl b) Work of adhesion and c) the spreading coefficient of water on the alcohol.
- Referring to the Al-Si Eutectic diagram discussed on the Phase Diagrams-Lever Rule handout, calculate the mass fraction of the phase, silicon, given a sample with 51.7 wt% aluminum at 576.99 ˚C.For a given liquid a plot of the lnP versus 1/T gives a straight line with a slope of -2829 K.What is the ΔHvap of this liquid? Your answer should be in kJ/mol and have 3 sig figsPolyethylene terephthalate is an example of synthetic material in Materials and Engineering. 1. Describe the relation of its building blocks in connection to its “bulk” (materials) properties. (No need for essay, just a brief scientific explaination!) If you may, please send link as well of the reference that you use for the answer).

