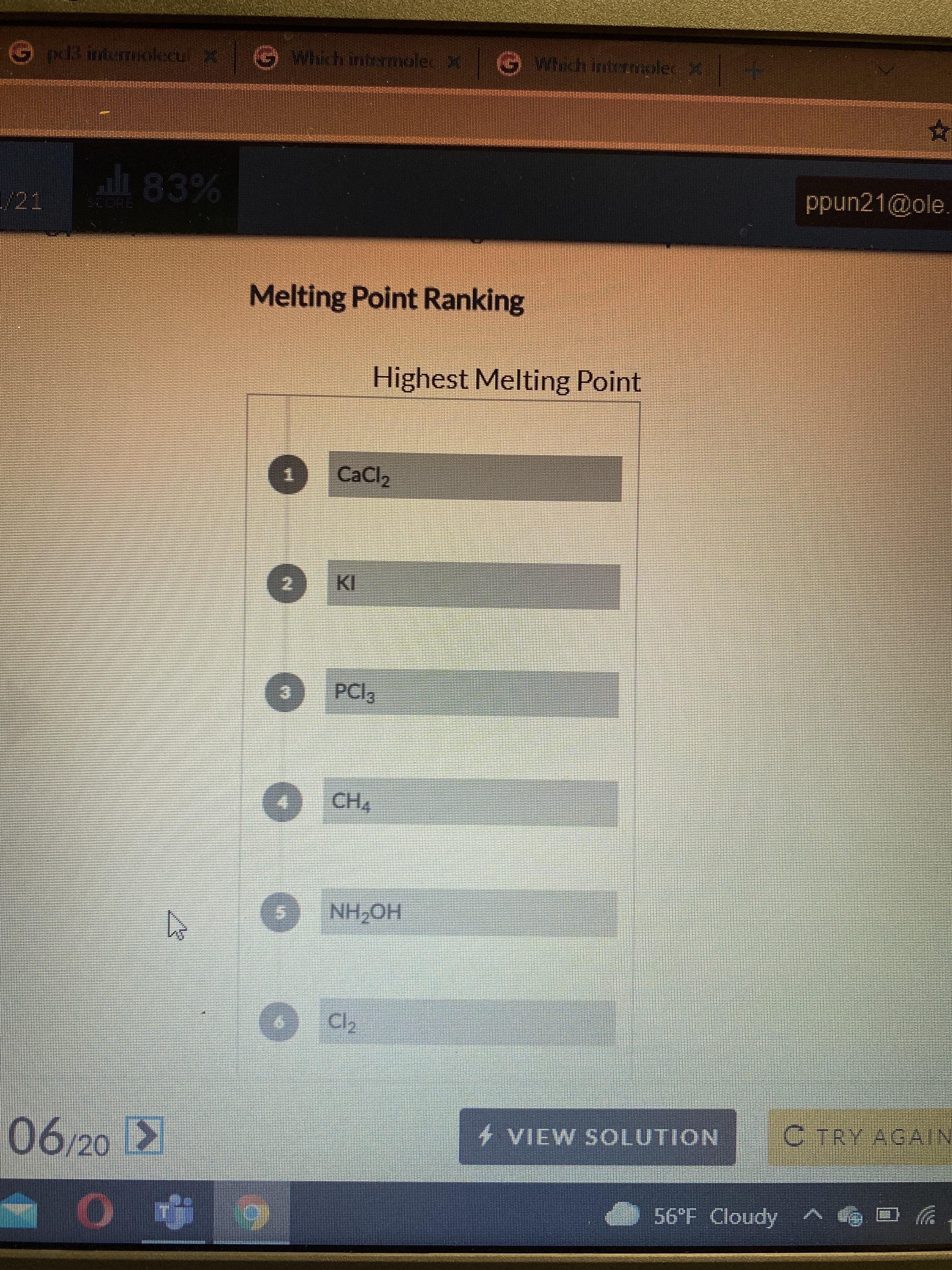
ali 83% ppun21@ole. COR Melting Point Ranking Highest Melting Point CaCl2 2. KI PCI3 CH4 HO HN Cl2 4 VIEW SOLUTION C TRY AGAIN 06/20 2 O v Apnop 1,95
Q: 1400 Mo [D] H. [C] Pressure (in mm Hg) 1000 [A] 800 600 400 [B] E. 200 - 20 60 100 140 180 260…
A: Melting point is defined as the temperature at which a solid substance melts. In the given phase…
Q: A solution is prepared from 2.00 x 10 2 g of sucrose (342g/mol) and 3.5 x 10 2 g water (18 g/mol).…
A: Given values- Weight of Sucrose = 200 gm Weight of water = 350 gm Vapour pressure of water = 17.5…
Q: When 6.943 g of lysine was dissolved in 109.3 g of benzene, the boiling point was 354.4 K. What is…
A: Given, When 6.943 g of lysine was dissolved in 109.3 g of benzene, the boiling point was 354.4 K.…
Q: The elevation in boiling p CuC I, in 1 kg of water u: (molecular weight K%30.52 K mol-1) pe
A:
Q: The freezing temperature of a solution produced by dissolving 25 g of a solid solute in 1.5 L of…
A: The boiling point of solution can be calculated by using following formula: Where ΔTF = freezing…
Q: Calculate Henry's law constant (mol/L atm) for O2 in blood which contains 0.25 g/L at 37°C and…
A:
Q: 17. Compute for the boiling point elevation of the solution. * OA. 100.08 °C OB. 103.25 °C C. 100.81…
A:
Q: 8- Arrange the following compound according in increasing melting point: KCI, He , H2O, HF A) Kcl >…
A:
Q: What is the boiling point of a mixture composed of 75.0 g HOCH,CH,OH (ethylene glycol) and 155 g…
A: Given Data: The mass of ethylene glycol is 75.0 g. The mass of H2O is 155 g.
Q: 1. Which aqueous solution will boil at the highest temperature? a. 1.0 m CaCl2 b. 0.5 m C6H12O6…
A: The addition of solute in a solution decrease the vapour pressure, thus increase the boiling…
Q: What is the boiling point of the automobile radiator fluid prepared by mixing 1.02 L of ethylene…
A: Numerical based on elevation in boiling point.
Q: Determine the melting point of an aqueous solution containing 193 mg of saccharin (C7H5O3NS) added…
A: Melting point: Temperature at which the solid and liquid forms of a pure substance can exit at…
Q: The vapor pressure of water at 20°C is 17.5 mm Hg. What is the vapor pressure of water in mm Hg over…
A: Given, a solution is prepared by dissolving sucrose in water. Sucrose is an example of non-volatile…
Q: 12. Two charged spheres 10 cm apart attract each other with a force of 3.0 x 10-6N. What force…
A:
Q: Calculate the cryoscopic and ebullioscopic constants for liquid bromine, Br2. You will need the…
A:
Q: 8- Arrange the following compound according in increasing melting point: KCI, He , H2O, HF A) Kcl>…
A: With increasing in the intermolecular interaction, melting point also increases as more energy is…
Q: Find the boiling point of water is of an at the to p mt. Fijiż which has at mospheric pressure of…
A:
Q: How much solute (lauric acid) do you need to add to the solvent wax (stearic acid) to make a 100g…
A: Well, In order to calculate the amount of lauric acid and solvent( stearic acid) we need to…
Q: Q: Show the effect each of the following on the Tm th crystalline melt temperature ? 1- Add salt…
A: Crystalline melting temperature of polymer is that temperature at which polymer changes from a semi…
Q: A certain substance X has a normal boiling point of 141.7 °C and a molal boiling point elevation…
A:
Q: Will water inside a sealed spherical glass boil (see bubble
A: When water is in inside a spherical sealed glass , And temperature of glass is raised for trying to…
Q: Use the graph below to answer the following questions. 2. What is the vapor pressure of CHCI3 at…
A: 2) Vapour pressure of CHCl3 at 50∘C = 70 kPa 3) Boiling point of water when external presssure is 30…
Q: Phase Diagram for Solution Critical Point 217.75 (Critical pressure) Normal freening point 1.00 New…
A: normal boiling point is the temperature at which the vapour pressure is equal to 1 atm or 760 mmHg.…
Q: You are handed a 15.85% by mass aqueous KCl (m.m. = 74.5513 g mol–1) solution (at 25 °C). Assume the…
A: Given, Mass percent of KCl in solution = 15.85% Molar mass of KCl = 74.5513 g/mole Density of the…
Q: Which of the principal characteristics of solutions can we see in the solutions of K2Cr2O7 as shown…
A: There are two types of solution mixtures, first is homogenous solutions and the second type is…
Q: Determine the solubility
A: Henry’s law states that dissolved gases in liquid are directly proportional to the partial pressure…
Q: From the following data, calculate the heat of solution for KBr. LicI LiBr KCI KBr Lattice…
A: Answer: Heat of solution of KBr will be ∆Hsolution=∆Hdissolution+Lattic energy∆Hsolution-lattice…
Q: om the following data, calculate the heat of solution for KBr. LICI LiBr KCI KBr Lattice…
A:
Q: 3. Consider the following chemical reactions: KCIO3 (s) -------> KCI(s) + O2(g) NANO3(s) NaNO2(s) +…
A: Mass of mixture of potassium chlorate and sodium nitrate = 5.00 gm Mass of residuals KCl + NaNO2 =…
Q: 17) A certain soft drink is bottled so that a bottle at 25 degrees Celsius contains CO2(g) at a…
A: Since, Henry Law, Concentration= Henry constant× pressure
Q: 4.57 g of C12H22O11 were dissolved in 100 g of H₂O. Density of solution is 1 g/ml. Calculate the…
A:
Q: The melting point of H2O(s) is 0 °C. Would you expect the melting point of H2S(s) to be greater or…
A: Intermolecular forces are the attractive force that withholds two molecules and the influence of…
Q: In fermentation distillation experiment, why does the boiling point increase, and the % ethanol…
A: A question based on physical chemistry, which is to be accomplished.
Q: A solution is made using 28.3 percent by mass CH₂Cl₂ in CHCl₃. At 30 °C, the vapor pressure of pure…
A:
Q: 1. What would it mean if the melting point had a wide range in temperature rather than a narrow…
A: Hello. Since your question has multiple sub-parts, we will solve the first three subparts for you.…
Q: 23. Using the x and y coordinates, what will happen if you add solute particles to the boundary…
A: In the given question, it is asked to tell the effect on temperature and pressure after adding any…
Q: 7. Which ionic solid is most soluble in water at 60°C? A. sugar (C12H22O11) B. KNO3 C. NANO3 D. NaBr…
A: according to the given graph above. at 60°C the solubility of Ce2(SO4)3 has the maximum solubility.
Q: 100 mL solution was prepared by dissolving 10.00 g of CaS04 (MW= 136. 14 g/mol) in water. The ensity…
A:
Q: The boiling point of 500.0L of water was determined to be 100.0°C. Ana has decided to increase the…
A:
Q: The freezing point (Ta for t-butanol is 25.50 C and Kr is 9.1"Cim. Uvually r-butanol absorbs water…
A: Lets mass of water = x gm Moles of water = MassMolar mass=x18 Molality = Mass of soluteMass of…
Q: 1. What would it mean if the melting point had a wide range in temperature rather than a narrow…
A: Since you have posted a question with multiple subparts, we will solve the first three subparts for…
Q: What volume of water at 20.0 °C (d = 0.9982 g cm3) is needed to dissolve 36.1 g of sucrose to make a…
A: What volume of water at 20.0°C (d = 0.9982 g cm 3) is needed to dissolve 36.1 g of sucrose to make a…
Q: Determine the freezing point and boiling point of the solution. (Assume density of water = 1.00…
A:
Q: A certain liquid X has a normal boiling point of 129.00 °C and a boiling point elevation constant…
A:
Q: The vapor pressure of pure toluene at 60 °C is 0.198 atm. A solution prepared by dissolving 2.2 mol…
A: We have given that Vapor pressure of pure toluene = Ptoluene° = 0.198 atm Moles of toluene = nt =…
Q: Jeneen v A certain liquid X has a normal bolling polnt of 120.70 °C and a bolling potnt elevation…
A: Given : Normal boiling point of liquid X = 120.70 oC Mass of solvent i.e liquid X = 800 g = 0.800 Kg…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- see the graph and determine/give the ff: a. 1 eutectic point (temp and composition) b. 1 peritectic point (temp and composition) c. melting point of Mg2SiO4Melting point (Tm): -94.9C Boiling point (Tb): 110.7C a.) Using the data, find the vapor pressure of toluene at 20C in units of mol/L b.) What is the enthalpy of vaporization (∆?vap ) for toluene in kJ/mol?Consider the data in the table. Compound Melting point (°C) ????? (??/???)ΔHfus (kJ/mol) Boiling point (°C) ????? (??/???)ΔHvap (kJ/mol) HFHF −83.11 4.577 19.54 25.18 HClHCl −114.3 1.991 −84.9 17.53 HBrHBr −86.96 2.406 −67.0 19.27 HIHI −50.91 2.871 −35.38 21.16 Using the data in the table, calculate Δ?fus and Δ?vap for HF. Δ?fus= J/(K⋅mol) Δ?vap= J/(K⋅mol) Determine the entropy change when 8.70 mol HF(g) condenses at atmospheric pressure. Δ?= J/K
- Barium titanate (BaTiO3) is an ionic solid with a density of 6.02 g/cm3. Its structure is described by orthogonal basis vectors (i.e. at 90 degrees to each other) with lattice parameters of a = b = 0.3992 nm and c = 0.4036 nm.a) What crystal system does barium titanate belong to, based on the above information?Explain your answer. b) Find the number of Ba, O, and Ti atoms per cubic centimeter in barium titanate (i.e.provide a value for each element). c) Sketch and label the (111) plane of BaTiO3 with its atoms and dimensions.1.At -185°C and 1 atm ethane (MW = 30.07 g/mol) exists as a solid with a FCC structure. Using the data provided below: E/k=194.1K, bo=0.1794 m3/kmol Determine the diameter of one molecule of ethane (in angstroms). 2. What is the void fraction? 3. Find the density of the solid etahne. 4. Calculate the distance of separation (in m) between two ethane molecules when the potential energy is at a minimum. 5. At the separation distance calculated in part (4), determine the minimum value of the potential energy between two molecules (in J).At 25 C, how high will water rise in a glass capillary tube with an inner diameter of 0.63 mm? Refer to Example 10.4 for the required information.
- The vapor pressure of 1-chlorotetradecane at several temperatures are tabulated below. T (ºC) P∗ mm Hg 98.5 1 131.8 5 148.2 10 166.2 20 199.8 60 215.5 100 Use the calculated values for ln(P∗)ln(P∗) to estimate the vapor pressure at 185°C.The sublimation pressures of solid Cl2 are 352 Pa at -112oC and 35 Pa at -126.5oC. The vapor pressures of liquid Cl2 are 1590 at -100oC and 7830 at -80oC. Calculate (a) △subH, (b) △vapH, (c) △fusH, and (d) the triple point. Answer: [31.4 kJ mol-1 ; 22.1 kJ mol-1 ; 9.1 kJ mol-1 ; -102oC]A liquid rises to a height of 1.50 cm in a capillary tube. What is the radius of the tube if the density and surface tension of the liquid are 1.80×10³ kg/m³ and 0.135 N/m respectively?
- #55. Distinguish between isotonic and isometriccontractions.??.a. How much methylcellulose 1500 should be weighed? viscosity for a suspension is 750 cps. Note 25-50% of the Rx volume comes fro cherry syrup : the remainder of the volume come from the methylcellolose. b. what volume of water should be added to hydrate the methylcellulose?Properties of ABE Materials 1.) prediction methods in determining density



