11 In an experiment to determine hydrolysable tannins in plants by absorption spectroscopy the following results were obtained: Absorbance 0.084 0.183 0.326 0.464 0.643 Concentration, mg ml 0.288 1.420 0.123 0.562 0.921 Use a suitable statistics or spreadsheet program to calculate a quadratic relationship between absorbance and concentration. Using R' and R values, comment on whether the data would be better described by a cubic equation. (Based on Willis, R. B. and Allen, P. R. 1998. Analyst 123: 435)
11 In an experiment to determine hydrolysable tannins in plants by absorption spectroscopy the following results were obtained: Absorbance 0.084 0.183 0.326 0.464 0.643 Concentration, mg ml 0.288 1.420 0.123 0.562 0.921 Use a suitable statistics or spreadsheet program to calculate a quadratic relationship between absorbance and concentration. Using R' and R values, comment on whether the data would be better described by a cubic equation. (Based on Willis, R. B. and Allen, P. R. 1998. Analyst 123: 435)
Chapter12: Spectrochemical Methods
Section: Chapter Questions
Problem 11P
Related questions
Question
I need the answer as soon as possible
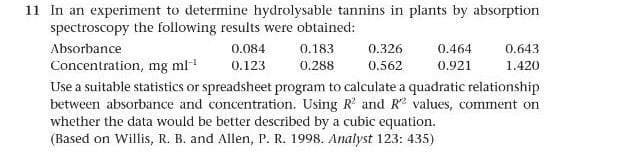
Transcribed Image Text:11 In an experiment to determine hydrolysable tannins in plants by absorption
spectroscopy the following results were obtained:
Absorbance
0.084
0.183
0.326
0.464
0.643
Concentration, mg ml
0.123
0.288
0.562
0.921
1.420
Use a suitable statistics or spreadsheet program to calculate a quadratic relationship
between absorbance and concentration. Using R' and R values, comment on
whether the data would be better described by a cubic equation.
(Based on Willis, R. B. and Allen, P. R. 1998. Analyst 123: 435)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
