11. Which of the following compounds would you expect to be soluble in water? a. LiCI b. CH4 C. HCI d. A and C e. A, B and C Give detailed Solution with explanation needed. don't give Handwritten answer
11. Which of the following compounds would you expect to be soluble in water? a. LiCI b. CH4 C. HCI d. A and C e. A, B and C Give detailed Solution with explanation needed. don't give Handwritten answer
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter10: Solutions
Section: Chapter Questions
Problem 74QAP: The freezing point of 0.20 m HF is -0.38C. Is HF primarily nonionized in this solution (HF...
Related questions
Question
None
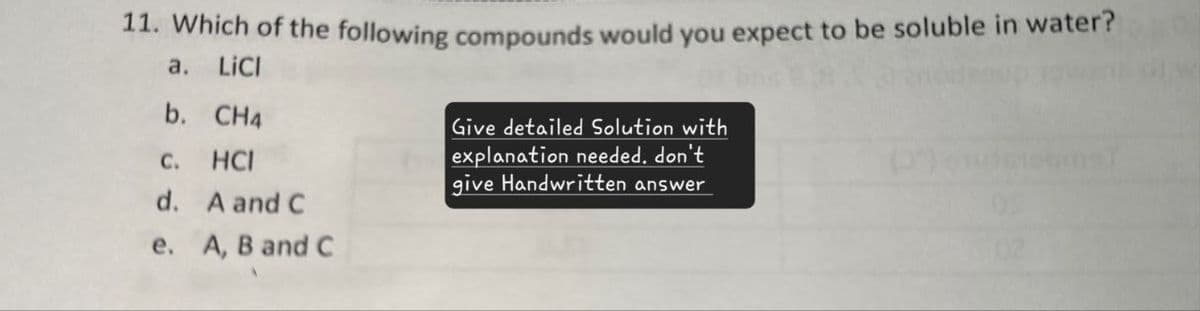
Transcribed Image Text:11. Which of the following compounds would you expect to be soluble in water?
a. LiCI
b. CH4
C.
HCI
d. A and C
e. A, B and C
Give detailed Solution with
explanation needed. don't
give Handwritten answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning



Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning



Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning