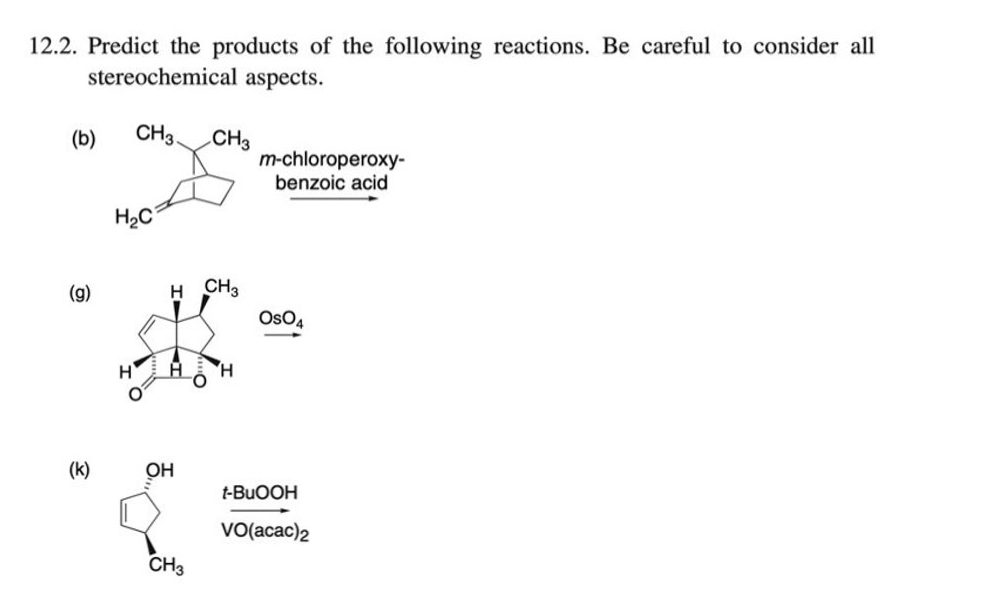
12.2. Predict the products of the following reactions. Be careful to consider all stereochemical aspects. CH3 CH3 m-chloroperoxy- benzoic acid (b) H2C (g) H CH2 OsO4 H. (k) OH t-BUOOH VO(acac)2 CH3
Q: Suppose 2.94g of H2 and 2.34g of He are trapped in 2.65L container at standard temperature.…
A:
Q: 1. Compute for the pH and generate a titration curve for the titration of 50.0 mL 0.0500 M NaOH with…
A: Here the analyte is NaOH and the titrant is HCl, both of which are strong and monoprotic. Hence we…
Q: OH viii. ii. Br +En 6. X. 7. ix.
A: Alcohol is converted into alkyl-bromide with HBr Dehydrohalogentation with bulkier base like…
Q: maWhat mass of nickel (in gm) could be formed in a solution of nickel (II) chloride by passing 0.25…
A: First we calculate charge using value of current and time. From charge , we can calculate moles of…
Q: 1 3. 7 8 A student dissolves 13. g of phenol (CH,OH) in 300. mL of a solvent with a density of 0.93…
A: Known Weight of phenol = 13.0 g Volume of a solvent = 300 mL Density of solvent = 0.93 g/mL Since…
Q: a. 1.1 M NaOH H2N + NABH4 MEOH 2.1 M HCI
A: (NaBH4 + NaOH) solution is a powerful basic reducing agent. Sodium borohydride reduce aldehydes and…
Q: Consider the titration of 100.0 mL of 0.10OM NAOH with 1.00 M HBr. Find the pH at the following…
A:
Q: F D E
A: In the heating curve, as heat is added to substance, its physical states changes. As we know, in…
Q: Balancing redox reactions in acidic solution 1) H2C204 + MnO4- CO2 + Mn2+ a) The balanced…
A: We are authorised to solve only one question at a time. Please post rest of the questions…
Q: 0 000 If the molecule or polyatomic ion is polar, write th decided the hydrogen atom was closest to…
A:
Q: The chemical formulae of some acids are listed in the first column of the table below, and in the…
A: Acid Strong or WeakH3PO4…
Q: Question 15 2 pts Complete and balance the following redox equation. The sum of the smallest…
A: Balanced redox equation in acidic medium is written in the following step.
Q: a)Which compound/s will produce an orange precipitate upon reaction with 2,4-DNPH? (b)Which…
A: Here we are required to distinguish several compounds using different chemical tesr
Q: Structures of three coenzymes involved in Phase II conjugation reactions are shown below. For each…
A: For catalytic activity coenzymes are required by many molecules.
Q: What is the approximate concentration of free Al3+ ion at equilibrium when 1.54x102 mol aluminum…
A:
Q: Part 1B: pH of Acetic Acid Solutions (Note: HAc = HC2H3O2, acetic acid). 1.0 M HC2H3O2 0.10 M…
A:
Q: so what is the answer whcih later
A: To provide the answer.
Q: 4. The value of Kp for the reaction below is 1.6 at 308 K. NH,HS(s) = NH3(g) + H2S(g) Initially,…
A: Kp = Kc (RT)delta n delta n = Change in no of gaseous molecules = (2-0) = 2 R = 0.0821 Latm/molK
Q: 2 3 4 7 8. The vapor pressure of Substance X is measured at several temperatures: temperature vapor…
A: Clausius clapeyron equation will be used to calculate the enthalpy of vaporization.
Q: QUESTION 8: Metals react with oxygen gas to form metal oxide. For example, magnesium reacts as…
A: Given, Initially having 1.40 g of O2 Molar mass of O2 - 32 g/mol Molar mass of MgO - 40.13 g/mol
Q: Question 8 The following are spectrometric methods of analysis, except MS NMR Polarimetric O…
A: Mass Spectrometry or, MS is a technique which is used to determine the characteristics of the…
Q: b) Consider the following compound 2. OH HO НО НО OH Compound 2 ii. Propose a detailed reaction…
A: Detail mechanistic pathway is given below
Q: Instructions: Draw a Lewis structure for: (a) H;S, (b) OF2, (c) hydroxylamine (NH;0)
A: The Lewis structure Tells about the bonding of atoms to the central Atom.
Q: Q1. A. Balance the equation ( Oxidation Reduction ): - Fe*2 + MnO4 Mn+2 + Fe +3 {salt} B. Prove that…
A: Multiple question given here..
Q: ch the following methods of Anlaysis ♥ Fractional analysis, methylation, and periodate oxidation A.…
A: 1) Fractional analysis, methylation and periodate oxidation - B. chemical method
Q: Phenolphthalein is added to a certain mixture. When the mixture was heated, the color turned pink.…
A: Phenolphthalein is a very common indicator which is used for the detection of presence of acids or…
Q: Propose a mechanism for the reaction of BH3 with water. The products of the first reaction are H2…
A: We have to show the mechanism of the reaction of BH3 with water. The product of the first reaction…
Q: Balance the redox reaction {Cr2O72- + Fe2+ = Cr3+ + Fe3+} (acidic solution). Use the smallest…
A:
Q: A weak acid HA (pKa = 5.00) was titrated with 1.00M KOH. The acid solution %3D had a volume of 100.0…
A:
Q: Select the compound having the lowest dipole moment O a. NaF Ob. CH20 O c. HCN d. CH4 O e. SF4 O f.…
A: Which of the following having lowest dipole moment ?
Q: 2. How many moles of Ag Br can dissolve in 1.0 L of 0.40M NH;? Kr [Ag(NH3)2] *= 1.7 x 107 Ksp(AgBr)…
A: Given: Kf [Ag(NH3)2]+ = 1.7×107 Ksp(AgBr) = 5.0×10-13 We have to calculate the number of moles of…
Q: What are the practical uses of the points of the cloud, and the pour?
A: The answer is given below
Q: Question 1 <. A solution contains 21.65 grams of NaCl in 0.153 kg water at 25 °C. What is the vapor…
A:
Q: 47. Of the elements shown in the Periodic Table, which is the most reactive? III IV V VI VII I I Li…
A: Most reactive element is Potassium(K). This is alkali metal so reactivity is highest. Option (d) K…
Q: Given for nos. 1-2: The pH of a swimming pool was measured at 6.10. 1. What is its pOH? Answer:…
A: Given, pH = 6.10 1) we know, pH + pOH = 14
Q: The reaction described by the equation 0;(g) + NO(g) 0,(g) + NO,(g) has, at 310 K, the rate law rate…
A: we need to calculate the rate of the reaction
Q: weak acid HA (pKa = 5.00) was titrated with 1.00 M KOH. The acid solution had a volume of 100.0 mL…
A:
Q: O2022 McGraw Hill LLC. All R Check Explanation H. H. -ɔ- H H H. H. H. | | H. H -C C -C -C - C –H | |…
A: For the alkanes, the lonegst chain is the main chain. Rest attached to the main chain are called…
Q: QUESTION 3: What is the mass (in grams) of fluorine gas is needed to react completely with 17.8g of…
A:
Q: 2. Find the value for the equilibrium constant, K, at 25 °C (298 K) for the following reaction. The…
A: Given that - Temperature =298K Free energy (∆G°)= -13.6 kJ…
Q: How many moles of hydrochloric acid are present in 61 mL of 0.847 M hydrochloric acid? Report your…
A: Given :- molarity of HCl solution = 0.847 M Volume of HCl solution = 61 mL To calculate :- Number…
Q: QUESTION 2: How much Pareactant will be needed to produce 105.8g of tetraphosphorus decaoxide…
A:
Q: mWhat mass of nickel (in gm) could be formed in a solution of nickel (II) chloride by passing 0.25…
A: Given, I(current) = 0.25 amperes t(time) = 10 hours = 36000 seconds n(no of electrons transferred) =…
Q: I. 1. Cr,O, → Cr³* 2. NO → NO; 3. SO, 2 → SO2 4. MnO, → Mn,O, Balance each half reaction in basic…
A: A redox reaction is balanced by balancing the mass and charge. In an acidic medium, the charge of…
Q: ve the number of monochloro substituted products for the lowing reactants. Consider only…
A:
Q: Name these organic compounds: structure name CH,==CH – CH,– CH, CHE CH CH,– C= CH Explanation Check
A:
Q: Propose stepwise mechanism of following reaction.
A:
Q: 6. A gas mixture contains 5 x 10°mole CO and has a total of 1 mole. What is the CO concentration in…
A:
Q: Question 2 Н—с—оН 3. H2, Pd/BaSO, 1. NaCN H', H20 Н—с—оН 4. H-О 2. H30* -NH2H Н—с—Он H,C-OH Which…
A: It is application of kilani Fischer synthesis Here are required to show the product formed after…
Q: Which is correct and which is incorrect from the following sentences? Proposition True False (a) The…
A: The solution is shown below
Step by step
Solved in 2 steps with 1 images

- Can you assist with predicting the organic product(s) of the last two reactions and provide stereochemistry, if applicable.I am working on a practice assignment for my organic II course and am having difficulty with a question that asks to identify the reaction sequence used to synthesize isopropylcyclopentane. I would really appreciate the help!Hydrocarbon 10 and ester 12 react upon heating to give a cycloaddition product D. (i) Suggest a structure for product D and provide a curved arrow mechanism that accounts for its formation. (ii) Draw the molecular orbitals corresponding to the HOMO of 10 and the LUMO of 12 and show that these are of suitable symmetry for a concerted cycloaddition to take place. (iii) Use frontier molecular orbital theory to account for the observation that the formation of D is faster in the presence of AlCl3. please answer all thank you
- Grignard reagents react with oxirane (ethylene oxide) to form 1° alcohols but can be prepared in tetrahydrofuran solvent. Why is this difference in behavior observed? A. There is a better leaving group in the oxirane molecule. B. Steric hindrance in the case of tetrahydrofuran precludes reaction with the Grignard. C. It is easier to obtain tetrahydrofuran in anhydrous condition. D. Oxirane is a cyclic ether, while tetrahydrofuran is a hydrocarbon. E. The oxirane ring is the more highly strained.1. Give the major organic product(s) of the reaction and include all the stereochemistry as appropriate. Identify any meso compounds. 2. Indicate whether a solution of the product would be optically active or no not active and provide a simple reasoning to support your choice.Nitrating methyl benzoate. 1.Describe the mechanism of EAS clearly and in detail 2. What is the effect of subtituent of the outcome of EAS;include 3 classes of subtituents
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Propose structural formulas for compounds (2) and (3).
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estroneOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.How many chiral centers are present in estrone?Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)



