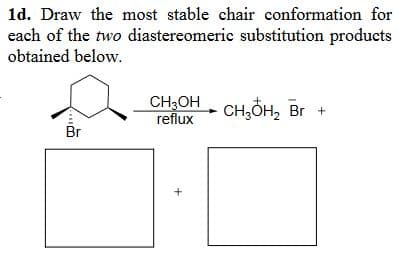
1d. Draw the most stable chair conformation for each of the two diastereomeric substitution products obtained below. CH3OH CH3OH2 Br + reflux Br +
Q: Predicting qualitatively how entropy changes with mixing and separation 1/3 For each system listed…
A: Step 1: Step 2: Step 3: Step 4:
Q: Calculate the pH when 15.0 mL of 0.200 M HBr is mixed with 30.0 mL of 0.400 M CH₃NH₂ (Kb = 4.4 ×…
A: The objective of this question is to calculate the pH of a solution when a strong acid (HBr) is…
Q: 1-bromo-5-chloropentane LDA 1 eq a) Hg(OAc)2 b) NaBH NaOH (1 eq) CHOO Note this product has a ring.…
A: Step 1:a) In this reaction, • LDA is the strong base which will abstract most acidic proton to…
Q: The IR provided is of monoacetylferrocene. Label each peak corresppnding to its appropriate…
A: In the IR spectrum of monoacetylferrocene, you'll typically observe several peaks corresponding to…
Q: Please correct answer and don't use hend raiting
A: The two structures are the cyclic forms of sugars in their pyranose forms. The sugar at the left is…
Q: Draw the starting alkyl chloride that would produce this alkyne under these conditions. Drawing 1.…
A: Step 1: Step 2: Step 3:E2 elimination It typically requires a strong base to abstract the proton and…
Q: Solids which have a structure similar to liquids, but without the freedom of motion of liquids are…
A: Solids are characterized by extended- three-dimensional arrangements of ions, atoms, or molecules.…
Q: The structure of the amino acid tyrosine is shown below in its completely protonated form. PK 16 OH…
A: The question is asking for the structure of the amino acid tyrosine in its completely protonated…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Can you please answer this question with steps. Thanks.
A: The objective of the first part of the question is to determine which ligand, X or Y, is the…
Q: The temperature is measured versus the energy added to 1 mol of a material. What energy is…
A: Step 1:From melting point to boiling point the energy (heat) is required to increase the temperature…
Q: Assign 1H NMR chemical shifts (ppm values) to the hydrogens indicated in…
A: In the given question, we have to assign the 1H NMR chemical shifts to the protons associated with…
Q: None
A:
Q: Please correct answer and don't use hend raiting
A: Step 1: An aldehyde or ketone in the presence of oxidizing agents like peracid gets converted to…
Q: Question 2 Consider the following reaction: 2NO(g) + O2(g) → 2NO2(g) + 112kJ (heat is released from…
A: Let's break down each option:a. positive and the reaction is endothermic: This option is incorrect…
Q: When the Pb2+ concentration is 5.00\times 10-4 M, the observed cell potential at 298K for an…
A: Step 1:Introduction to data :Given ,Concentration of Pb2+= 5.00 X 10-4Objective :Determine the…
Q: Please don't provide handwritten solution .....
A: 1. Calculate the molar concentration of ascorbic acid in the solution before it was evaporated.…
Q: At the end of the experiment you will be assessing the purity of your aspirin by measuring its…
A: Step :
Q: Part A Review Constants Draw the product formed when the compound shown below undergoes a reaction…
A:
Q: What is the cell potential for a voltaic cell having one electrode composed of a Zn strip dipped in…
A: 1. Given: [Zn2+](left)=0.034M;[Zn2+](right)=0.414MStep 1: Write the equation we need to…
Q: 1. Write two possible defect reactions and corresponding mass action expressions for the following:…
A: Given:Equilibrium constant is the ratio of concentration of products to the concentration of…
Q: None
A: Answer well explained above
Q: An analytical chemist is titrating 173.3 mL of a 0.1600M solution of diethylamine ((C₂H)2NH) with a…
A: The objective of this question is to calculate the pH of the base solution after a certain volume of…
Q: Polymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer…
A: Thank you.
Q: Payalben
A: NaOCH2CH3 in CH3CH2OH is used to substitute the -OR group of an ester with -OCH2CH3. The given ester…
Q: Figure 2: Re-arranging the expressions below, make a separate plot showing the concentration of the…
A: This code will generate a plot showing the concentration of the protonated form ([HA][HA]) and the…
Q: Consider the reactionI2(g) + Cl2(g)2ICl(g)Using the standard thermodynamic data in the tables linked…
A: Step 1: Step 2: Step 3: Step 4:
Q: Provide a synthesis of the final molecule from the initial molecule. Use reasonable reagent
A: Here's a step-by-step procedure for synthesizing 4-cyclopentene-1-carboxylic acid from malonic acid:…
Q: pH i got from previous question which was calculate the pH of a 1.00L buffer solution that is 0.100M…
A: To calculate the pH of the buffer solution after the addition of NaOH, we need to consider the…
Q: Draw the most reasonable resonance form for the CH_3N_2+ cation. Include all lone pairs and non zero…
A:
Q: Use the data in the table to fill in the questions
A: Cell Notation RulesThe anode (where oxidation occurs) is placed on the left and the cathode (where…
Q: None
A: In organic chemistry, chirality centers play a crucial role in determining the stereochemistry of…
Q: Predict the product of the following reaction: Cl₂, H HỌ CH3 C1 OH xi
A: Hence, option A is correct
Q: Sm. Don't provide handwriting solution
A: Step 1:1) what is the pH ?It is negative log of hydronium ion concentration. 1) reaction as…
Q: None
A:
Q: Create a titration curve for each trail of pH vs. volume of base added. calculate the volume of NaOH…
A: The objective of the question is to analyze the titration data of an unknown acid with a standard…
Q: Calculate the pH: - 0.20 M arginine hydrochloride - 50.0 mLs of 0.10 M arginine hydrochloride to…
A: The given solution is arginine hydrochloride solution. Ka1 of arginine = Part a:Concentration of…
Q: Write out the equilibrium expression for gypsum. Does gypsum, CaSO4 , precipitate?
A: However, since water (H2O) is a pure liquid, its concentration remains constant and does not appear…
Q: Need answer step by step I need answer expert solution
A: Step 1: Step 2: Step 3: Step 4:
Q: b write a mechanism for the reaction using curved arrows to show electron reorganization.…
A: Step 1: Step 2: Step 3: Step 4:
Q: For a molecule, the phosphorescence lifetime without quenching is 1.0 s while with a quencher…
A: Given:Phosphorescence lifetime without quenching (τ0) = 1.0 sPhosphorescence lifetime with quencher…
Q: Use the References to access important values if needed for this ques Hybrid orbitals are formed by…
A: The objective of this question is to determine the electron pair geometry, molecular geometry, and…
Q: Explain or Give conclusions abput the precipitation, identification and separation of Group I ANIONS
A: The objective of the question is to explain the precipitation, identification, and separation of…
Q: Which among the options for minimzing errors (Separation, Saturation-Mix Modification-Masking,…
A: The errors are the discrepancies b/w a measured value and the true value.
Q: Please answer this question with steps thanks.
A:
Q: Can you explain the mechanism for this question HO OH H2SO4 cat. H₂O
A:
Q: For the E2 reaction below, predict the major organic products. If no elimination products are…
A: do leave a thumbs-up, as it motivates us to assist you better and faster. Thank You.
Q: Polymers may be composed of thousands of monomers. Draw three repeat units (trimer) of the polymer…
A:
Q: None
A: For concentration cell, cathode and anode are same electrodeSo, Eo = 0Number of electron being…
Q: For the reaction2C2H6(g) + 7O2(g)4CO2(g) + 6H2O(g)H° = -2.86×103 kJ and S° = 92.7 J/KAt standard…
A: The objective of the question is to determine at what temperature range the given reaction would be…
Step by step
Solved in 2 steps with 1 images

- Two conformations of cis-l, 3-dimethylcyclobutane are shown. What is the difference between them, and which do you think is likely to be more stable?Lithocholic acid is an A–B cis steroid found in human bile. Draw lithocholic acid showing chair conformations, as in Figure 27-11, and tell whether the hydroxyl group at C3 is axial or equatorial.Indicate the relationship between each pair. Choose from: configurational stereoisomers,conformers, constitutional isomers, or different formulas (Each term is used at least twice.)
- Draw the Newman projections of the three p0ssible staggered c0nf0rmati0ns 0f 2,3-dimethylbutane, viewed through the C2—C3 b0nd. What are the relative energies 0f each conformation?Draw the most stable chair conformation possible for this compound. Are there any 1,3-diaxial interactions in the most stable chair conformation ( H-H not included ) and please fill in the blank: From the biomolecules surveyed, the compound is a(n)___________Eclipsed adn Anti C2H6 a)Perspective drawings of both conformers b)What is the relationship between the above two structures? How many different eclipsed or anti conformers can you make by rotating around teh C-C bond?
- label the stereocenters r,s,e,z and specify if any are meso. whats are the products to each orher? draw the chair conformationd and explain which one is the most stable for both products. are ha and hb homo/enantio/diasterio/hetero topic? also hc and hd.Draw 2 of the chair conformations for the compounds shown below and then cricle which are most stableThe diaxial conformation of cis- 1,3-dimethylcyclohexane is approximately 23kl/mol (5.4 kcal/mol) less stable than the diequatorial conformation. Draw thetwo possible chair conformations, and suggest a reason for the large energydifference.
- Consider 1-bromo-2-methylpropane and draw the following. Q) The staggered conformation(s) of lowest energyWhy is this chair conformation the most stable trans-1-chloro-3-flurocyclohexane? Wouldn't the conformation with Cl & F in equatorial positions be the most stable conformation?Which of the following molecules are chiral?I. cis-1,3-DibromocyclohexaneII.1-Bromo-1-methylcyclohexaneIII.trans-1-Bromo-3-methylcyclohexaneIV.cis-1-Bromo-3-methylcyclohexane



