Q: How does the Hollow Cathode Lamp work during the quantitative analysis of Iron (Fe) in Atomic…
A: Hollow cathode lamps (HCL) are discharge lamps designed for use in Atomic Absorption (AA)…
Q: Question 1 What arenium intermediates are formed in the ortho- and meta-substituted products of the…
A: Bromine electrophile is attack on ortho position gives carbocation which is stabilized by methyl…
Q: A sample of gas is cooled, releasing 202 Jof energy. The change in internal energy for the gas…
A: Given:: ∆H = -202 J ∆E = -417 J w = ?
Q: dation takes place alone, without an accompany lation happens in the salt bridge, and electrons…
A: In electrochemical cell, Oxidation takes place at anode and reduction takes place at cathode…
Q: For which of the following reactions would K be largest? 2H+(aq) + Pb(s) --> H2(g) + Pb2+(aq)…
A: The reactions given are, 2H+(aq) + Pb(s) → H2(g) + Pb2+(aq) 2H+(aq) + Sn(s) → H2(g) + Sn2+(aq)…
Q: 24 20.0 mL of a 0.100 M butanoic acid solution is titrated with a 0.250 M solution of Ba(OH)2…
A: 1.) In this base is added to a weak acid ( butanoic acid ) , so we would calculate moles of acid and…
Q: Match each reaction/phrase with the correct product/answer. Isobutyl alcohol and warm, basic KMNO4…
A:
Q: AR a - 4CDU-AD, 4CBa How many grams of AD, IsMW-285.72 g/mol would be made from the reaction of…
A: The balanced equation given is AB4 (aq) + 4 CD (aq) → AD4 (aq) + 4 CB (aq) Number of molecules of…
Q: From the following vapor pressure data for iodomethane, an estimate of the molar heat of…
A:
Q: Use the listing of standard reduction potentials on page 10 to determine the E° for Ce**(aq) + 4e →…
A: We would use two half reactions to calculate E° for given reaction .
Q: Calculate the basic dissociation constant for pyridine, C5H5N, if the acid dissociation constant for…
A: Given that, Ka =5.9×10-6 Kb= ?
Q: Consider the system at equilibrium. 2 CO(g) + 0,(g) = 2CO,(g) How will increasing the concentration…
A: Recall the reaction-- 2CO(g) + O2(g) <-----> 2CO2(g)
Q: The following information is given for lead at latm: T = 1740.00°C T = 328.00°C Specific heat solid…
A: negative sign represent the release of energy.
Q: Use the information provided to determine ΔH°rxn for the following reaction: ΔH°f (kJ/mol)…
A:
Q: Refer to the following phase diagram (not to scalet) for nitrogen 1.00 stm 0.127 631 632 TKelvin At…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: A Cr(s) | Cr(aq)|| Fe*(aq) | Fe(s) galvanic cell has a standard cell potential of 0.700 V. Calculate…
A: We have find out the ∆G and maximum amount of work done by the cell on the surrounding.
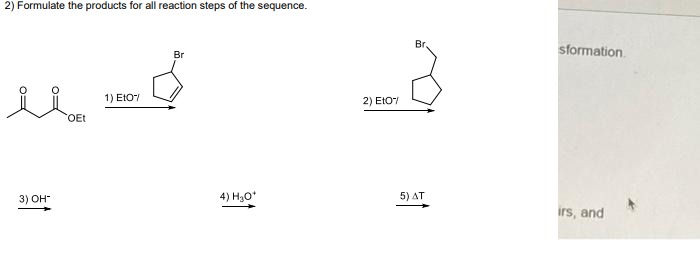
Q: Fill in ALL the missing parts to the organic transformations in this sequence - T tBuOK CH,CH;CH,…
A: Since you have posted a question with multiple sub-parts, we will solve the first three subparts for…
Q: 2.4x10-3 mol Sb Express your answer using two significant figures. m = Submit Request Answer bo
A: Detail mathematical calculation is shown below
Q: How much energy is required to vaporize 41.4 g of dichloromethane (CH2Cl2) at its boiling point, if…
A:
Q: What is the common factor between MS, SEM, and XPS? (Hint: It makes those techniques a good deal…
A: MS - Mass Spectrometry SEM- Scanning Electron Microscope XPS- X ray photoelectron spectroscopy
Q: How many signals would be in the 1C NMR spectrum of structure A below?
A:
Q: In Grignard reaction experiment, 0.6458 g of Mg (molar mass 24.305 g/mol, 2.03 ml of 2-bromopropane…
A: Given: mass of Mg = 0.6458 g molar mass of Mg = 24.305 g/mol Volume of 2-bromopropane = 2.03 mL…
Q: An enclosed sample of ideal gas undergoes an isochoric process at 6.24m. Its initial temperature is…
A:
Q: 6. Consider the following Newman Projections. Which is the most stable conformation? Which is the…
A:
Q: Describe the preparation of 2 L of 85 mM phosphate buffer, pH 6.5, using 120 mM NaH2PO4·6H2O and…
A: Given: total volume of buffer solution = 2 L Concentration of buffer solution = 85 mM Concentration…
Q: in the manufacture of ammonium chloride from the ammonium sulfate process which reacts with sodium…
A: Le Chatelier's principle: Le Chatelier's principle states that if a dynamic equilibrium is disturbed…
Q: mistry 2SO2 (g) + 02(g) = 2S03(g) Kc = 6.1 x 104 at 750K. At equilibrium PSO2 = 1. 22 bar and PO2 =…
A:
Q: Choose the reaction that illustrates ΔH°f for Mg(NO2)2. Choose the reaction that illustrates ΔH°f…
A: Standard Enthalpy of formation, ΔH°f :- The enthalpy change involved when one mole of a substance…
Q: Consider the reaction: 2NO(g) + 02(g)32NO2(g) AG° = -70.6 kJ Calculate the equilibrium constant of…
A:
Q: Consider the hypothetical reaction: A +B product Determine the unit of the rate constant, k, using…
A:
Q: (Q56) Consider yttrium iodate, yttrium fluoride, lanthanum iodate, and gallium (III) hydroxide:…
A:
Q: One method for producing hydrogen is to react coke (mostly solid carbon) with steam to make syngas,…
A: The equilibrium reaction given is, => Given: Initial pressure of H2O = 3.31 atm.
Q: For the compound below please choose the correct set of chair and flipped chair conformations: CI CI…
A: While drawing chair conformation for a given compound , we need to focus that stereochemistry of the…
Q: At 10°C, the vapour pressure of a liquid substance is 6.83 x 102 Pa. At 30°C, the vapour pressure is…
A:
Q: What is the effect of other substances found in tea leaves (especially cellulose) on the process of…
A: Interpretation- We have to tell about the effect of other substances found in tea leaves (especially…
Q: What is the meaning of each quantity? pre-exponential factor measure of particle size as a target…
A:
Q: What are the main components of fluoride ion-selective electrode?
A: The three main components of making a measurement at an ISE are an inner reference, or standard,…
Q: 8. Draw the structures of the conjugate acid and base for the following reaction. Provide the…
A: pKa is inversely proportional to acidity. So lesser pka, stringer the acid is.
Q: Using this data table classify each of the following sustances tested as either reducing sugar or…
A: Urine normally does not contain any sugar. Diabetic urine contains glucose.
Q: 19 Consider the reaction: Cl20 (g) + 3/2 O2 (g) --> 2CIO2 (g) Given the following data: AH°…
A:
Q: Dinitrogen tetroxide (N2O4) decomposes to nitrogen dioxide (NO2). If ΔH° = 58.02 kJ/mol and ΔS° =…
A:
Q: (Q54) Nickel iodate, Ni(IO3)2 is being precipitated out of a 1.00 L solution. If the concentration…
A: Given that, Nickel iodate, Ni(IO3)2 is precipitated Concentration of disdolved Ni2+ ion , [Ni2+] =…
Q: (Q60) One iron (III) cation can form a complex with a total of six thiocyanate (SCN-) anions. Which…
A: given :- one iron (iii) cation can form complex with 6 SCN- anions to write :- formula of complex
Q: Br CH;CHCH,Br 1.NaNH, , NH3 H,0* 2. (5 hrs) 3. Н-О (wash) Think about last weeks LAB! LOH *C=CCH3…
A:
Q: During the chemical reaction in an electrochemical cell, the anode is where oxidation happens.…
A:
Q: Any chemical or physical change that absorbs energy is an endothermic process. is an exothermic…
A:
Q: Significant Digits are required for all Questions below Al>(SO4)3(aq 1.45 g of Al2(SO4)3(aq express…
A: We have No of moles= mass/molar mass Molar mass of Al2(SO4)3 = 342g/mol Mass= 1.45g Moles =…
Q: 10.00 mL of a solution of NaOH was titrated with with 0.09546 M KHP solution. At the equivalence…
A:
Q: QUESTION 11 Which one is a wrong statement about the intermediate of nucleophilic acyl subsitution?…
A:
Q: balanced equation for the neutralization
A:
Step by step
Solved in 3 steps with 3 images

- q - GOPAL write full mechanism for atleast 4 and synthesus steps pleaseCreate a synthesis of the given product beginning with carbon fragments of three or fewer. Please make this legible and easy to follow, thanks.Full reaction pathway and mechanism of the synthesis of 2-chloropyridine, using chlorine (Cl2) as the reagent and water (H2O) as the solvent and start with pyridine. Also draw the overall reaction first above with a straight forward reaction arrow.
- For F, please draw the structures and indicate synthesis steps by arrows that show the reaction directionPlease help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)For D, please draw the structures and indicate synthesis steps by arrows that show the reaction direction.
- Chemistry please provide the flow of electrons aswell!! thank you! using the starting material (on the left) to determine the sythetic route which will be the most reaosnable and effective to theres none, you have to start witth the begin products to get to finish needed by using the minmium reagents and reactions needed to get to the final productWhat is the complete mechanism for this F!she rxn?The reaction below is homogenic homolysis heterolysis heterogenic

