Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the aldol…
A: The carbonyl electrophile and enolate nucleophile both are present in a single compound
Q: State two conditions which favor SN1 and SN2 reaction. ( Structure of alkyl halide and…
A: Nucleophillic substitution reactions proceed either by SN1 or SN2. SN1 is a type of nucleophilic…
Q: Why is this considered an Elimination E2 reaction? C₃H₈O (l) → C3H6 (g) + H2O (l) propan-1-ol…
A:
Q: H2C Br
A: E1 elimination is a two step process in the first step leaving group leaves and forms a carbocation…
Q: Choose the member of each pair that will react faster by the SN1 mechanism.(a) 1-bromopropane or…
A: 1-bromopropane or 2-bromopropane , which will react faster by the SN1 mechanism ?
Q: Select the member of each pair that undergoes SN1 solvolysis in aqueous ethanol more rapidly
A: Given pair is,
Q: Select the member of each pair that shows the greater rate of SN2 reaction with KI in acetone.
A:
Q: 3.Draw the crown ether that would be most appropriate for the reaction below. NaF benzene Br F.
A: The appropriate crown ether for the SN2 reaction is given below
Q: Which solvent would have a higher reaction rate for SN1? acetone ethanol Why does the solvent you…
A: Ethanol is a polar solvent that is used in SN1 reactions.
Q: Br Br X
A: SN1 reaction is known as unimolecular nucleophilic substitution reaction. In this reaction rate is…
Q: Which alkyl halide in each of the following pairs will react faster in an SN2 reaction with OH?…
A: SN2 reaction: It is nucleophilic substitution reaction in which attacking of nucleophile and removal…
Q: Which solvent conditions favor SN1 reactions over SN2? polar protic apolar protic polar aprotic O…
A: Sn1 reaction is unimolecular nucleophilic substitution reaction. Sn2 is bimolecular nucleophilic…
Q: Rank the following compounds in order of increasing SN2 reaction rate with a good nucleophile.…
A: We have to rank the given compounds in order of increasing SN2 reaction rate with a good…
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Predict reagents needed to complete this SN1 solvolysis reaction. Br
A: Detail mechanistic pathway for SN1 reaction is shown below
Q: On a single graph, draw the reaction coordinate diagram for the addition of one equivalent of HBr to…
A: Given reactions, (1) Addition of HBr to 2-methyl-1,3-pentadiene: (2) Addition of HBr to…
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone…
A: A carbonyl electrophile and an enolate (formed after the deprotonation of an aldehyde/ketone)…
Q: Select the member of each pair that shows the greater rate of SN2 reaction with KI in acetone.
A: The addition of nucleophile and removal leaving group takes place in simultaneously in SN2 reaction…
Q: Would you expect acetate ion (CH3CO2 -) to be a better nucleophile in an SN2 reaction with an alkyl…
A: The SN2/E2 reaction of alkyl halide is with neutral nucleophile/base, the increasing polarity of the…
Q: Classify following solvent as protic or aprotic HCONH2
A: Protic solvent They have presence have O-H and N-H bonds. Protic solvent can participate in hydrogen…
Q: Choose the starting materials needed to complete this Robinson annulation reaction. Assume an acidic…
A:
Q: Would you expect acetate ion CH3CO2 to be a better nucleophile in an SN2 reaction with an alkyl…
A: The SN2/E2 reaction of alkyl halide is with neutral nucleophile/base, the increasing polarity of the…
Q: O, N. Na OHCag) + Nacl
A: Acid-Base reaction: In chemistry acid-base reaction is the fastest reaction and salt will be formed…
Q: True or false? In an SN2 reaction with HBr, 1-propanol will react faster than 2-propanol to give the…
A: SN2 reaction refers to the substitution reaction between a nucleophile and an electrophile. A…
Q: 1. Based on the theoretical result, what is the order of reactivity of primary, secondary, and…
A: Since you have asked multiple questions, we will answer the first one for you. To get the remaining…
Q: SN1 reactions are better performed in protic solvents. Explain why the researchers could not perform…
A: In SN1 reaction, alky halide/sulphonate react with a weak nucleophile. In first step of reaction…
Q: 3. Rank the following in order by the rate in which they could participate in an SN1 reaction (“1"…
A:
Q: Which is the best solvent for an SN2 reaction? О СНЗОН O DMSO O hexane O H2O
A:
Q: How does doubling [RX] affect the rate of an E1 reaction?
A: The removal of hydrogen halides in a two-step mechanism where the rate of the reaction depends on…
Q: How does solvent affect rate of reaction if its increase or decrease in sn1 or sn2
A: In SN1 reactions, there carbocation intermediate so solvents of higher polarity stabilizes these…
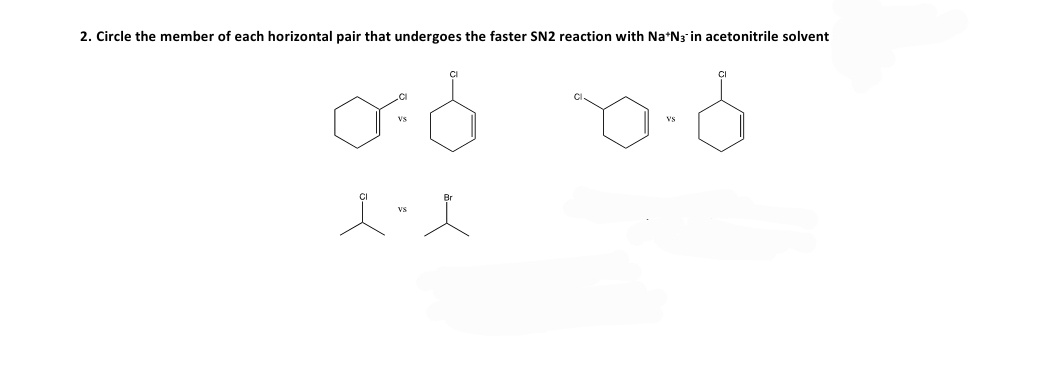
Q: Select the member of each pair that shows the greater rate of SN2 reaction with KN3 in acetone.
A: SN2 reaction rate depends upon the steric effect. Higher the steric hindrance, lower would be the…
Q: SN1 is always a competing reaction of E1. True or False?
A: SN1 Reaction: A tertiary alkyl halide substitutes nucleophile by the removal of halogen as leaving…
Q: Fill in the blanks: The E1 mechanism is most favored by a alkyl halide and a solvent.
A: E1 means elemination is Unimolecular and in this mechanism is most favourable in 3o type halocarbon…
Q: 8. Rank from best (#1) to worst (#3) substrate in an SN2 reaction and explain. Br Br
A: When an incoming nucleophile attacks the alkyl halide and substitutes the halide ion and the rate of…
Q: NH2 In which active solvent will you choose to extract from chloroform (CHC3)?
A: The separation of compound from the mixture on basis of their polarity is known as differential…
Q: What is the difference of SN1 and SN2 reaction
A: SN means 'nucleophilic substitution' and 1 defines the rate determining step is unimolecular and 2…
Q: 3) What is a nucleophile? Provide some examples of nucleophiles that would work well in the SN2…
A:
Q: Choose the member of each pair that will react faster by the SN1 mechanism.(a)…
A: Interpretation: The member of the given pair will react faster by the SN1 mechanism is to be chosen.…
Q: Show the difference between SN1 and E1 Reactions ?
A:
Q: In which solvent would an SN1 reaction be fastest? 100% ethanol 80% ethanol, 20% water 50% ethanol,…
A: SN1 reaction proceeds faster in 80% ethanol 20% water.
Q: Select the member of each pair that undergoes SN1 solvolysis in aqueous ethanol more rapidly
A: Given:
Q: Select the member of each pair that shows the greater rate of SN2 reaction with KI in acetone.
A: SOLUTION: Step 1: The given compounds are 1) 2-bromo pentane 2) 2-bromo-3-methylbutane.…
Q: Why is the reaction of the type shown below usually done? a.To make an aldehyde or ketone less…
A: We've to predict the function of the reagent in the given reaction.
Q: An alkene with electron withdrawing group is a good dienophile?
A: * Diels alder reaction occur between diene and dienophile. *Dienophiles are electron deficient…
Q: A set of three nucleophilic displacement reactions is shown below: CH3 SOH CH3CH2ĊCH3 SN1 reaction
A: GIVEN
Q: How does increasing base, solvent, or alkyl halide or aptly halide + base affect or decreasing them…
A: I am giving you all the information regarding SN1/SN2 or for E1 /E2. For direct answer prefer step…
Q: Questions 1 and 2 Solvents used are hexane,toluene,acetone,ethanol, and water
A: Determination of the polarity of the given compound: Trans-stilbene can be categorized as nonpolar,…
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone…
A: Aldol condensation: The alpha hydrogen of carbonyl compounds is acidic in nature and it is removed…
Step by step
Solved in 3 steps with 3 images

- Select the member of each pair that shows the greater rate of SN2 reaction with KN3 in acetone.What products are formed when the compound below are heated strongly in presence of an appropriate solvent? use curved arrows to show movement of electronsSN1 reactions are better performed in protic solvents. Explain why the researchers could not perform the reaction using 100% water as solvent instead of 50% aqueous acetone.
- SN1 is always a competing reaction of E1. True or False?complete arrow pushing mechanism for SN1 and SN2 reaction 1: SN1 = sodium iodide reacts with 2-bromobutane 2: SN2 = silver nitrate reacts with allyl chlorideWhich compound in each of the following pairs will react faster in SN2 reaction with —OH?(i) CH3Br or CH3I (ii) (CH3)3 CCl or CH3Cl
- Would you expect acetate ion CH3CO2 to be a better nucleophile in an SN2 reaction with an alkyl halide carried out in methanol or in dimethyl sulfoxide?How does changing the halide from (CH3)3CBr to CH3CH2CH2Br affect the rate of an E1 reaction?A nucleophilic addition is not considered SN1 or SN2 because: A.) The reaction proceeds through an E1 mechanism or E2 B.) There is no carbonyl substitution when doing the addition to this C.) The solvent for this reaction is non-aqueous D.) The solvent for this reaction is non-polar

