2. OH 1. 2. NH₂ a Proton transfer b = Lewis acid/base c= Radical chain substitution Biosynthesis of adrenaline (epinephrine) Mal Å 0,0CH,CH.CH- NH Submit Answer 1. Dry ether 2. Hyd Retry Entire Group Review Topka NH₂ d Electrophilic addition e E1 Elimination f = E2 Elimination Identify the mechanism by which each of the reactions above proceeds from among the mechanisms listed. Use the letters a-i for your answers. toy 9 more group attempts remaining OCCHICHICH 9= SN1 Nucleophilic substitution h= SN2 Nucleophilic substitution Previous
2. OH 1. 2. NH₂ a Proton transfer b = Lewis acid/base c= Radical chain substitution Biosynthesis of adrenaline (epinephrine) Mal Å 0,0CH,CH.CH- NH Submit Answer 1. Dry ether 2. Hyd Retry Entire Group Review Topka NH₂ d Electrophilic addition e E1 Elimination f = E2 Elimination Identify the mechanism by which each of the reactions above proceeds from among the mechanisms listed. Use the letters a-i for your answers. toy 9 more group attempts remaining OCCHICHICH 9= SN1 Nucleophilic substitution h= SN2 Nucleophilic substitution Previous
Chapter16: Chemistry Of Benzene: Electrophilic Aromatic Substitution
Section16.SE: Something Extra
Problem 30MP: The carbocation electrophile in a Friede1-Crafts reaction can be generated by an alternate means...
Related questions
Question
Chemistry
Please help.
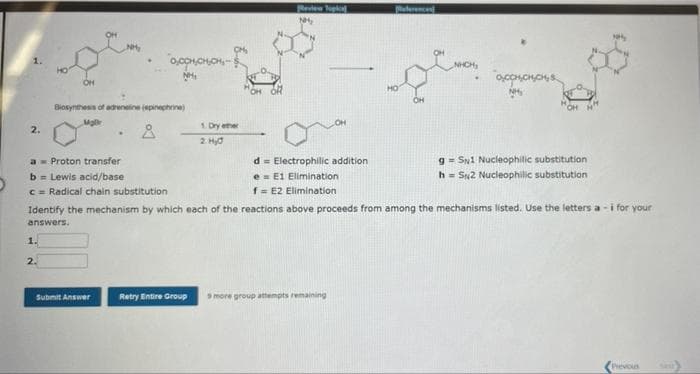
Transcribed Image Text:2.
OH
1.
2.
NH₂
Biosynthesis of adrenaline (epinephrine)
Mar
a Proton transfer
b = Lewis acid/base
c= Radical chain substitution
Å
Submit Answer
0,0CH,CH,CH-$
1. Dry ether
2. Hyd
Retry Entire Group
Review Topka
NH₂
d
Electrophilic addition
e
E1 Elimination
f = E2 Elimination
aferenced
Identify the mechanism by which each of the reactions above proceeds from among the mechanisms listed. Use the letters a-i for your
answers.
9 more group attempts remaining
ty
OCCHICHICH
gSN1 Nucleophilic substitution
h= SN2 Nucleophilic substitution
Previous
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning