2. Roughly sketch the hydropathy plot for the given hypothetical membrane protein. Follow the color assignments of the helical domains and properly label your plot and axes (NB: Only a rough sketch is being asked. No need be exact with the hydropathy indices and residue numbers). CO-
2. Roughly sketch the hydropathy plot for the given hypothetical membrane protein. Follow the color assignments of the helical domains and properly label your plot and axes (NB: Only a rough sketch is being asked. No need be exact with the hydropathy indices and residue numbers). CO-
Anatomy & Physiology
1st Edition
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Chapter3: The Cellular Level Of Organization
Section: Chapter Questions
Problem 6RQ: Because they are embedded within the membrane, ion channels are examples of ________. receptor...
Related questions
Question
Graph the given clearly and answer the question
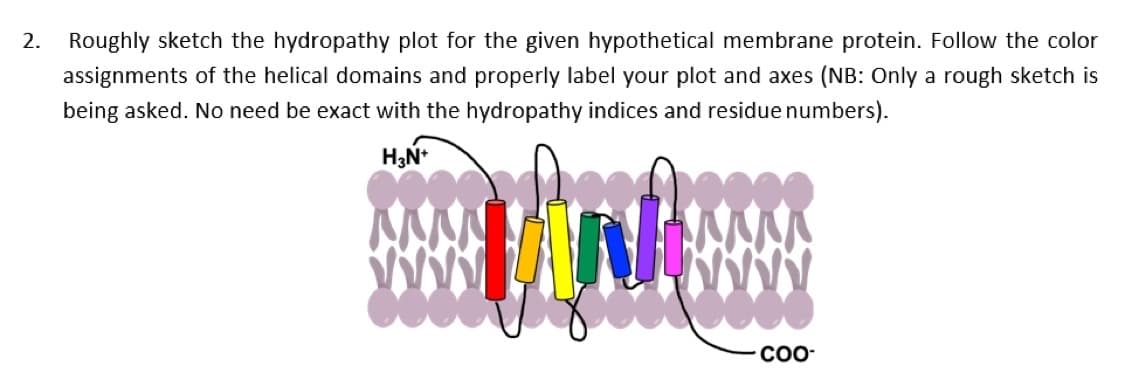
Transcribed Image Text:2. Roughly sketch the hydropathy plot for the given hypothetical membrane protein. Follow the color
assignments of the helical domains and properly label your plot and axes (NB: Only a rough sketch is
being asked. No need be exact with the hydropathy indices and residue numbers).
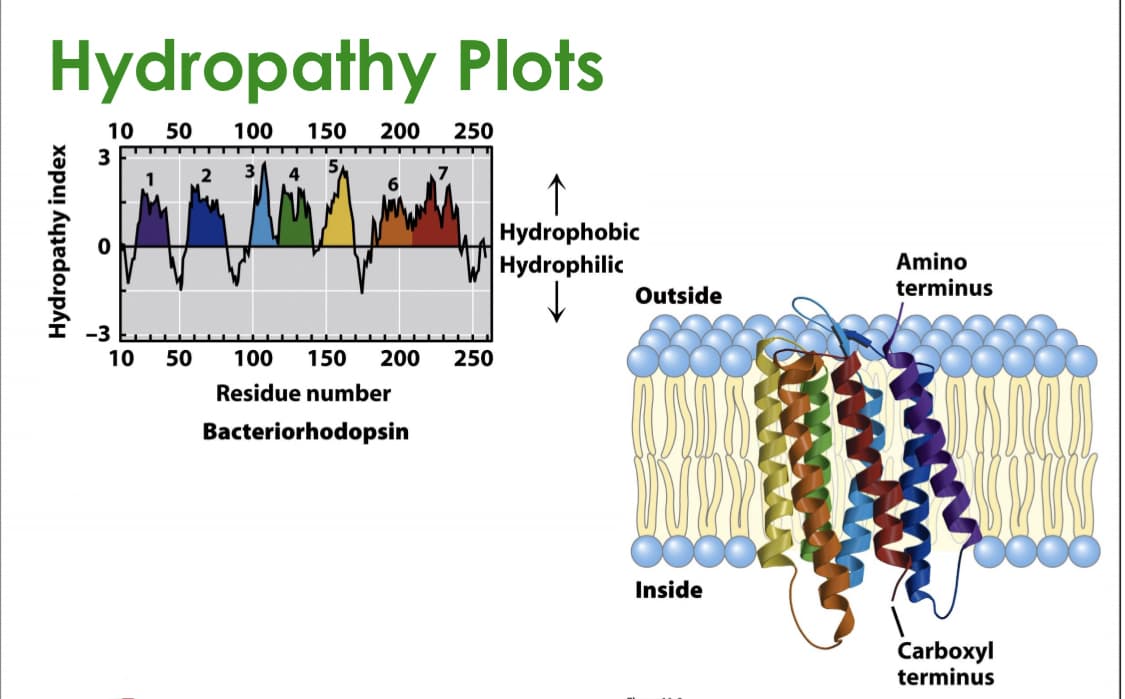
Transcribed Image Text:Hydropathy Plots
10
50
100
150
200
250
3
↑
Hydrophobic
Hydrophilic
Amino
terminus
Outside
-3
10
50
100
150
200
250
Residue number
Bacteriorhodopsin
Inside
Carboxyl
terminus
Hydropathy index
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax