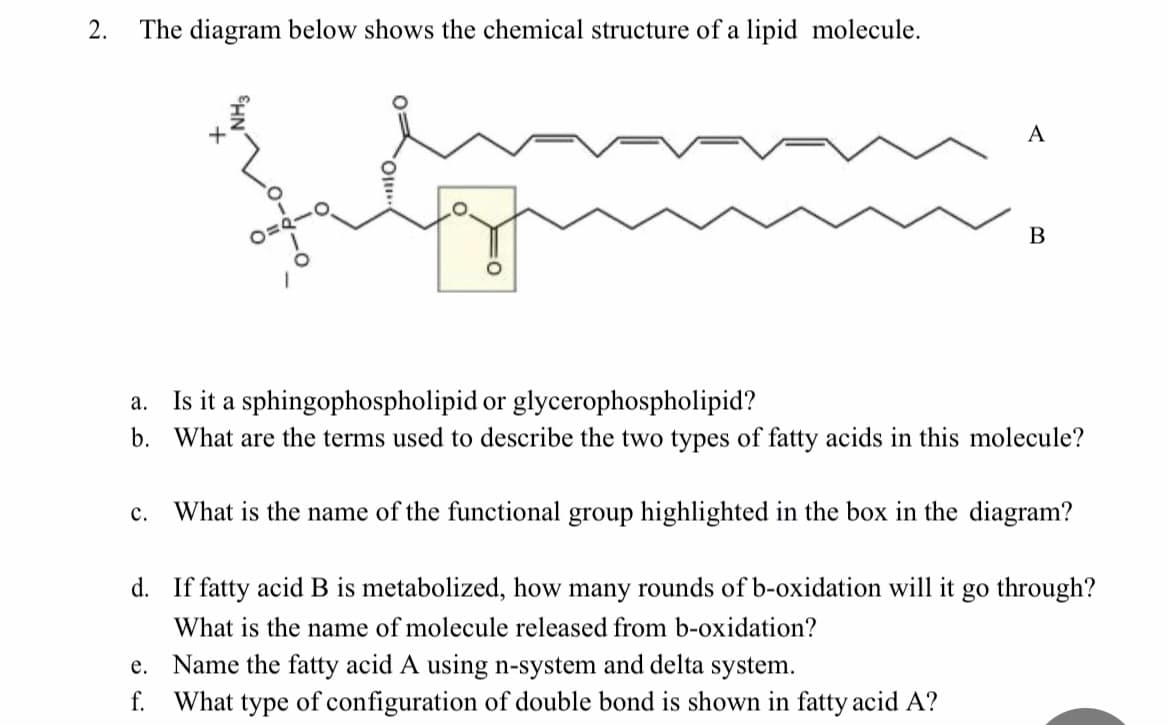
2. The diagram below shows the chemical structure of a lipid molecule. A В a. Is it a sphingophospholipid or glycerophospholipid? b. What are the terms used to describe the two types of fatty acids in this molecule? c. What is the name of the functional group highlighted in the box in the diagram?
Q: Enzymes use several types of catalytic mechanisms. Differentiate between acid-base catalysis, covale...
A: Enzymes are biocatalysts which increase the rate of biochemical reactions. Substrate molecules bind ...
Q: . The optimal conditions for salivary lysozyme (hydrolyzing glycoproteins of bacterial wall) are 37 ...
A: Lysozyme is a naturally occuring enzyme found in bodily secretion such as- tears,saliva and milk. L...
Q: critic the article. You are expected to have an (a) Introduction (b) Discussion (yo can attach relev...
A: The question is all about the carbohydrate i.e polysaccharides that consists of a unit sugar monosac...
Q: Hello, please answer the questions below about Vitamin C. Name of Vitamin: Vitamin C Classification...
A: Introduction: Vitamins are natural substances found in both plants and animals and it is considered ...
Q: How many times can catalase be used to hydrolyze hydrogen peroxide? never once twic...
A: Catalase is an enzyme found in the cell (e.g. it can be found in peroxisome). Catalase helps to conv...
Q: What is the order of the electron transport chain starting from FADH2 and ending with Oxygen
A: The energy released during the oxidation of glucose to carbon dioxide generates NADH and FADH2. The...
Q: In noncompetitive inhibition, can both the substrate and the inhibitor bind at the same time? I kn...
A: Inhibitors are substances which bind to the enzyme and slow down or inhibit the enzyme activity. Bas...
Q: 3.) Islandicin is a simple anthraquinone molecule. Propose a biosynthetic pathway of the compound. о...
A: Islandicin is an anthraquinone derivative molecule found in lichens and fungus like Penicillium. Thi...
Q: Shown are different derivatives of acetylcholine. N' A H2N H2N 1. Which one is expected to lose its ...
A: Acetylcholine: Acetylcholine is a choline and acetic acid derivative where both are bond by ester bo...
Q: 2. which of the following is the reason why hemolysis shoule be avoided? a. enzymes have lower con...
A: Q2. Hemolysis is defined as the rupture of erythrocyte membranes, which results in the release of ha...
Q: Application. One way to test for glucose oxidase and all the reagents necessary for the reaction: gl...
A: Introduction: Glucose is measured in both plasma and whole blood. The concentration of glucose in w...
Q: What are the main structural features of the mucupolysaccharides chitin? How do this aid in its func...
A: Chitin: Chitin is a nitrogenous polysaccharide found in the exoskeleton of a tarantula. It is the p...
Q: topic: Bradford Assay There are numerous methods of protein determination in use, but this module fo...
A: The Bradford assay is a dye-binding assay that is based on the changes in color of the Coomassie dye...
Q: Tricarboxylic acid cycle (a.k.a.krebs cycle)?
A: Krebs cycle is one of the important cycles and also called the citric acid cycle. It is the second s...
Q: 1.) What are the main structural features of the polysaccharides starch? 2.) How do this aid in its ...
A: Starch is a homopolysaccharide. It is the carbohydrate reserve of the plants. It is an important die...
Q: To which class does each enzyme belong? Explain hexokinase alanine aminotransferase alcohol deh...
A: The enzymes are divided into 7 classes mainly oxidoreductases, transferases, hydrolases, ligases, ly...
Q: Why are buffers important in biochemical experiments?
A: Buffer : solution which resists any pH change via the addition of any acidic, or the basic component...
Q: When glucagon binds to its receptor in liver cells, PFK-2/FBPase-2 becomes dephosphorylated. (circle...
A: The balance between the rate of glucose leaving and entering the blood circulation determines...
Q: During cell couting, when the data are plotted in an ordinary graph or on a cross-section paper, why...
A: For experiments involving growth of cells whether bacterial or eukaryotic the results of a growth ex...
Q: What are the general name for the monomers, dimers and polymers of carbohydrates?
A: Carbohydrate: Carbohydrates are biomolecule made from Carbon, Oxygen and Hydrogen atom and have chem...
Q: A mutant form of polypeptide hormone has the following amino acid composition: Asp, Arg, lle, Met,...
A: The amino acid composition of a mutant peptide is Asp, Arg, Ile, Met, Phe, Pro, Tyr, Val. It is an o...
Q: . Use the table of completely made-up data below to answer the following questions about a completel...
A: Enzymes catalyze the rate of conversion of Substrate into product and catalysis takes place in pocke...
Q: 4. For a simple enzyme-catalyzed reaction that follows Michaelis-Menten kinetics with Vmax = 75 µM/s...
A: Hi! Thank you for the question. We are authorized to answer two subparts at a time, since you have n...
Q: Does your protein 3GRS have a quaternary structure??? talk about the tertiary structure of 3GRS. h...
A: All molecular models (atomic coordinate file) based on the X-ray crystallographic data of the struct...
Q: 1.) What are the main structural features of the polysaccharides starch? 2.) How do this aids in its...
A: Polysaccharides are major classes of biomolecules. Polysaccharides are long chain of carbohydrate mo...
Q: (In 10 sentences) Can disaccharide be completely assimilated and used by human cells as a direct sou...
A: All disaccharides are dimeric product of monosaccharide linked together with glycosidic bond. Sucros...
Q: What is plant biochemistry?
A: Introduction - Plant Biochemistry is the study of different chemical reactions in the plant such as...
Q: Find the Gibbs free energy of mixing when 2 moles of ethanol is mixed with 5 moles of water at a tem...
A: Given; n1= no. of moles of ethanol= 2 mol n2= no. of moles of ethanol= 5 mol T=27oC =273+27=300K ...
Q: 2. State the four types of attractive interactions that give rise to tertiary protein structure. 3. ...
A: Protein structure has different levels of conformation. They are, the primary structure, secondary s...
Q: Crystal: Calcium oxalate or calcium phosphate in kidney stones If it is something harmful, what can...
A: A kidney stone is a solid, marble-like piece of material that forms in one or both kidneys. It forms...
Q: Draw each of the enzymatic step for each chemical conversion and write the enzyme together with the ...
A: Fatty acid biosynthesis is the pathway of synthesizing fatty acids from acetyl-CoA and NADPH cataylz...
Q: Identify the structure of the predominant form of the pentapeptide at pH = 12 if there is a pentapep...
A: The given peptide is composed of five amino acids and it can also be called a pentapeptide. Amino ac...
Q: Why might it be necessary to include the 50 mL cultures in order to express protein? This question ...
A: Hi, First I would like to thank for submitting the question. The question has one error as the word ...
Q: Directions: Supply the missing information. Amino Acid Sequences of in Cytochrome-c ANIMAL AMINO ACI...
A: Cytochrome C is a mitochondrial protein that is found in the mitochondria. It is highly water solubl...
Q: determine the principle involved or color reaction mechanism of each steroid. progesterone with sul...
A: Steroids are defined as an organic compound with four rings consisting of 17 C-atom called steroid n...
Q: 1. it is used to selectively bind K+ causing an impedance change that can be correlated to K concent...
A: Hi! Thanks for your question. As you have posted multiple questions and have not mentioned which o...
Q: a.) Explain how DFD meat has a high water holding capacity but is described as dry? b.) Why is it i...
A: DFD - It is a D-glucose linear polysaccharide. Hundreds to thousands of glucose molecules produce ea...
Q: Circle and label the hemiacetal functional group and the acetal functional group in each of the foll...
A: Carbohydrate is a macronutrient that the body uses to generate energy and form complexes that are ne...
Q: What are reducing sugars? Why is sucrose non-reducing while lactose also a dissacharide, is reducing...
A: Carbohydrates are biomolecules containing carbon, hydrogen, and oxygen. Carbohydrates are classified...
Q: 5. If enzymes are truly specific, they will catalyze the reaction of one substrate only. Predict whe...
A: Hi! Since you have posted multiple questions and have not mentioned which to answer, we will answer ...
Q: 5. At pH 7, the net charge of tyrosine is? А. -2 В. -1 С. 0 D. +1 E. +2
A: PH is defined as the negative logarithm of H+ ion concentration. The amount of H+ ions in a solution...
Q: Even under isotonic conditions, there is a slow leakage of ions into animal cells. How does the plas...
A: Osmosis is biochemical process by which water moves from a less concentrated solution to more concen...
Q: Given: One way to think about the hexokinase is that it couples the hydrolysis of ATP (AG° - -30.5 k...
A: Hexokinase: Hexokinase phosphorylate Glucose to Glucose-6-PO4 by utilizing one molecule of ATP. Here...
Q: Assume you have a long polypeptide chain of Gly-Val dipeptides linked end to end. Explain the likel...
A: Proteins have four levels of the structural organization including Primary, secondary, tertia...
Q: Review Figures 8.12 and 8.13. In cells, the primers forDNA synthesis are short strands of RNA, so ea...
A: A primer molecule is a short strand of RNA. During replication, the complementary nucleotides are ad...
Q: Discuss and describe the five major groupings of amino acids.
A: Amino acids are compounds containing carbon, hydrogen, oxygen, and nitrogen. They are the building b...
Q: Hello! Please give a slogan for Vitamin C, its food sources and a trivia or important information of...
A: A vitamin is an organic molecule that is an important micronutrient that an organism need in small a...
Q: Which of the following affects the rate of enzyme driven reaction? rate constant air pressure conce...
A: Enzymes: Enzymes are biocatalysts that fasten the rate of a chemical reaction. It is proteinaceous ...
Q: Catalase activity in catalase test can be determined by doing which of the following? measuring the...
A: Catalase Test: This test is used to demonstrate catalase activity. Catalase is an enzyme which catal...
Q: Sketch a titration curve for the following amino acids and indicate the pKa values for all titratabl...
A: pKa value: It is a negative log of ka value, which is acid ionization constant or acid dissociation ...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- 1. Which of the following best describes glycogen? A. a fibrous protein that helps in maintaining cellular structure B. a storage polysaccharide found in plants C. has alpha 1-6 glyosidic bonds. D. is formed by the hydrolysis of glucose molecules. 2. Which part of an amino acid gives it its unique identity? A. the long carbon-hydrogen tails of the molecule B. the carboxyl and amino groups C. the components of its side chain D. the glycerol molecule that forms the backbone of the amino acid1.Linoleic acid and linoleic acid cannot be synthesized in human tissues, because of _____. * a.Human cells cannot synthesize unsaturated fatty acids. b.Human cells can synthesize only mono-unsaturated fatty acids. c.The corresponding biosynthetic pathway was lost during evolution. d.Humans are diploid organisms; only polyploidy organisms have enough genes for the synthesis of these fatty acids 2.What product could be formed from the breakdown of amylopectin and cellulose? * a.Cellulose could be broken down into chitin b.Cellulose could be broken down into sucrose c.Amylopectin could be broken down into maltose d.Amylopectin could be broken down into glucosamine 3. Which characteristics distinguishes amylopectin and glycogen? * a.Only glycogen has a coiled shape. b.They have different degrees of branching c.Only amylopectin can form hydrogen bonds d.They have different types of glycosidic bond1. Make a series (at least 3) of drawings to show the three dehydration synthesis reactions that take place between the two Fatty Acids, the Phosphate Head and the Glycerol. Show the water molecules made. a) Label the following on the completed phospholipid: hydrophilic and hydrophobic parts of the molecule, fatty acids, phosphate group, and glycerol. b) Compare the structure of the phospholipid to that of a triglyceride. How are the two molecules Similar? How are they different? 2. Use the following vocabulary to label and draw a cell membrane: cytoplasm (cell lumen) extra-cellular fluid hydrophobic hydrophilic polar head non-polar tails a) How many O-H groups are there? And are the O-H areas of sucrose polar or non-polar? b) What is the term that is used to discuss how tightly or loosely an atom's nucleus has a hold of its valence electrons?
- 2. Draw the condensed structural formula for the triacylglycerol made from 3 saturated fatty acids that are 18 carbons long. Using your diagram from #2 as the reactant, draw the full equation (using condensed structural formulas) for this triacylglycerol undergoing enzyme hydrolysis, using lipase.1. a. Explain why the melting point of palmitic acid (16 carbons, no double bonds) is slightly lower thanthat of stearic acid (18 carbons, no double bonds). Explain why the melting point of oleic acid (18carbons, one double bond) is lower than that of stearic acid b. A mixture of lipids containing phosphatidic acid, cholesterol, testosterone, phosphatidylserine, andphosphatidylethanolamine was applied to a hydrophobic interaction chromatography column. Thecolumn was washed with a high salt buffer, and the lipids were then eluted with decreasing saltconcentrations. In what order would the lipids be eluted from the column? Explain your answer.1. Name the process by which rice can become porridge or congee. Give details of the process in relation to the example. 2.(a) What is the name of the fat after hydrogenation? Draw a hydrogenated fatty acid. (Please include the atoms involved in the molecule) 2.(b) Give an example of hydrogenated fats from vegetable oil. How are these fats harmful to our health? 4. Briefly describe the digestion of this form of carbohydrate described in (iv) in mouth and small intestine before absorption. 6. Part Stomach secretes a chemical that makes it to have a low pH. Name this chemical and give TWO functions of this chemical. 6.e One structural feature in F facilitates absorption of nutrients. Name this structure.
- 3. One of the triacylglycerols found in corn oil contains palmitic acid, linoleic acid, and linolenic acid. During the production of spreadable margarine, all of the double bonds in this triacylglycerol are converted from cis to trans isomers. Linoleic acid: CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOHLinolenic acid: CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7COOH Palmitic acid: CH3(CH2)14COOH a. Identify the unsaturated fatty acid(s) listed above and provide the associated omega designation. b. Draw an accurate representation of the structure of the triacylglycerol present in spreadable margarine. Circle all ester bonds in the structure you have drawn.Although the first two carbons of fructose and glucose are identical in structure to DHAP and GADP (from glycolysis), DHAP and GADP equilibriate on their in solution to favor the ketone over the aldehyde, while fructose and glucose do not. Why? a)The larger size of the molecule sterically hinders the isomerization b)The larger sugars have more OH groups which hydrogen bond and disrupt isomerization c)The larger sugars cyclize, and there is no carbonyl to isomerize in the cyclic form d)The larger sugars cyclize, and in the cyclic form the hydrogen bonding is very strong e)The larger sugars are less soluble in water than the smaller sugars1. Draw the structure of triglyceride containing the fatty acids palmitic, oleic, and linoleic. How many Hydrogen atoms are needed in the its catalytic hydrogenation? 2. Draw the structure of Phosphatidylserine that contains oleic and arachidonic acid. What role does it play in the cell?
- 1. What reactive groups are involved in the joining of glycerol and fatty acids?2. Which one of these reactive groups is part of the glycerol molecule?3. Which one of these reactive groups is part of the fatty acid molecule?4. If lots of acetyl-CoA is being formed in the mitochondrial matrix, a particular compound will be exported into the cytosol. a.What compound is this? b. What effect does this compound have on fatty acid synthesis? c. How does the compound produce this effect?1. The following unsaturated fatty acids can be found in seminal fluid and act on smooth muscles. a.Linoleic acids b.Oleic acids c.Prostaglandins d.Nervonic acids 2. The process involved when fatty acid chains covalently bonded to a glycerol molecule through an ester linkage is? a.hydrolysis b.oxidation c.phosphorylation d.esterification



