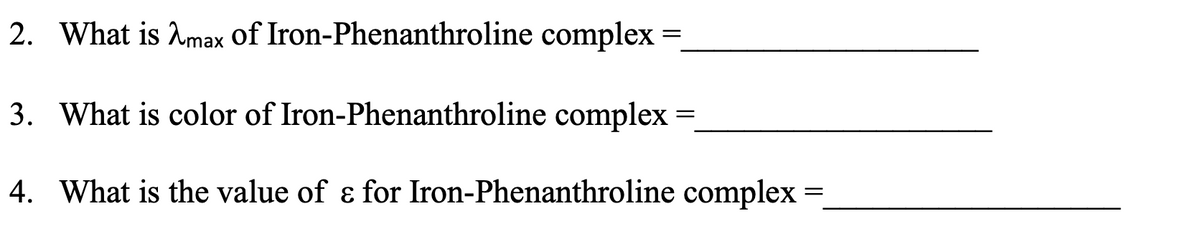
2. What is Amax of Iron-Phenanthroline complex = = 3. What is color of Iron-Phenanthroline complex = 4. What is the value of & for Iron-Phenanthroline complex =
Q: The zero order reaction A→ Products takes 63.5 minutes for the concentration of A to decrease from…
A: We know that, Rate of zero order reaction is independent on initial concentration of reactant.…
Q: Use the following information to answer the next question. 2HI(g) → H₂(g) + I₂(s) The standard…
A:
Q: Provide an IUPAC name for ball & stick - + labels W. (Specify (E) or (Z) stereochemistry, if…
A: Some of the key rules of IUPAC nomenclature include: Identify the longest continuous carbon chain…
Q: Draw the two possible products formed when this diene reacts with HBr. Do not include any byproducts…
A:
Q: The pH of a sodium acetate-acetic acid buffer is 4.40. Calculate the ratio CH₂COO [CH₂COOH] Note:…
A: Given, pH of the Sodium acetate acetic acid buffer = 4.40 [CH3COO-]/[CH3COOH] = ? Note: Ka for…
Q: Draw the skeletal (line-bond) structure of trans-4-methylpent-2- ene. 1 Drawing 1 1 1 1 I
A: Given that - Name of the organic compound - trans-4-methylpent-2-ene In this organic compound…
Q: You need to plan a reaction to produce 20 L of H2. What mass of potassium K will you to accomplish…
A:
Q: Predict the structure of the starting material used in the halogenation reaction below. I draw…
A: Product is given , we have to identify reactant
Q: Copper (II) sulfate reacts with zinc metal to form zinc sulfate and copper. What volume (in L) of…
A: Given that Copper (II) sulfate reacts with zinc metal to form zinc sulfate and copper. Molarity of…
Q: 12. Dehydrohalogenation of 2-bromobutane in the presence of a strong base proceeds via which of the…
A: In elimination reaction atom or group of atom removed from compound and new bond is formed.
Q: For the chemical reaction shown, 2 H₂O₂(1) + N₂H4 (1) 4 H₂O(g) + N₂(g) determine how many grams of…
A: To solve this problem we have use the stoichiometry of the balanced chemical equation.
Q: Note: Strictly follow the instructions for this question Example: OH CE CH3 H₂C (S)-2-butanol You…
A: To assign the absolute configuration, we need to first choose the carbon(s) with four different…
Q: 0.024 grams of solid silver chromate (Ag2CrO4) were added to 250 mL of water (final vol). How many…
A:
Q: Salicylic acid is the reactant that limits the amount of product in this experiment. If 2.02 g of…
A:
Q: What is the bond order of the following molecule, Y₂ ? Y₂ 0*2p *2p- 2p 02p AV AV 0*25 025 N
A: Bond Order = 1/2 (number of electrons in the bonding molecular orbital - number of electrons in the…
Q: hat are the appropriate reagents 1 O OEt 1
A: DIBAL-H is selective reducing agent used specially for the reduction of ester into aldehyde.
Q: The concentration of SO32 in a solution is determined by titrating it with a 0.1809 M permanganate…
A: Given, Concentration of the permanganate solution (MnO4-) = 0.1809 M = 0.1809 mol/L Note: 1000 mL =…
Q: At 455 K, AG = -7.8 kJ/mol for the reaction A (g) → 2 B (g). If the partial pressures of A and B are…
A:
Q: What invariant reaction is occurring at the point at 1493°C when (L+ 0 → y). Temperature, °C 1600 8-…
A:
Q: Write balanced half-reactions for the following redox reaction: 2+ BiO3(aq) + 3 H₂O(1)+2 Fe²+ (aq)…
A: The question is based on the concept of redox reactions. We need to write oxidation half and…
Q: #1) Complete and balance the following equations: Mg(OH)2(aq) + HCl(aq) → HI(aq) + H₂SO4(aq) →…
A: Balanced chemical equation: Balanced chemical equation can be define as the reaction in which number…
Q: We the UPAC Name of the structure bel WRITE IN CAPITAL LETTERS DO NOT INCLUDE SPACE IN BETWEEN THE…
A:
Q: Q5: 50 g of a 26% (m/m) solution was mixed together with 130 g of a 17% (m/m) solution. What is the…
A: To solve this problem we have to calculate the m/m% and volume of ethanoic acid. We use the formula…
Q: Solubility (g in 100g water) 100 80 60 40 20 0 10 1 20 1 1 30 3 40 ● 50 Temperature (°C) 60…
A: This is a solubility graph. It is used to find out the solubility of a solute per 100 grams of…
Q: 2. Solutions of [Cr(H₂O)]³ are pale green while the chromate ion [Cr(O)4]2 is intense yellow.…
A: Transition metals are a group of elements in the periodic table that are located in the d-block…
Q: Solubility (g/100 g H₂O) 160 140 120 100 80 60 40 20 OYb₂(SO4) O NaNO3 OKBr NaCl Solubility Varies…
A: The most soluble compound based on the given graph can be analyzed by plotting the points at 2oC and…
Q: 13. What Reagent(s) would be needed for the transformation shown below? А) МСРВА B) H3O+ C) CH3CO3H…
A: To solve this problem we have identify the reagent used to complete the given reaction
Q: Give the IUPAC name for this molecule. O CH3 T CH3CH₂CHCH₂CH₂CH IUPAC name:
A: To name any organic hydrocarbon, we need to count the total number of carbon atoms in longest…
Q: ▾ Part B -C=N
A: IUPAC nomenclature used for the systematic naming of the organic molecules.According to IUPAC…
Q: Molar masses for all compounds in the equation: K: 39.10 g/mol H₂O: 18.02 g/mol KOH:56.11 g/mol H₂:…
A:
Q: I. Newman projections of the different conformational isomers of butane Most stable: Least stable:…
A:
Q: (MGB A settling tank for a 2.5-million-gallon-per-day average overflow rate of 800 gallon/day-ft2.…
A: Solution- According to the question - Given - Discharge = 2.5-million-gallon-per-day overflow rate =…
Q: doform (HCl3, carbon with one hydrogen and three iodines) is a bright yellow solid. Which of the…
A: Iodoform test : The reaction of methyl ketones , iodine and a base gives a yellow colour…
Q: Evaluate the effectiveness of current safety measures used in the production and disposal of…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 9. What is the major organic product generated in the reaction sequence below? (A) (B) CI Br 1. K…
A: The question is based on organic reactions. we need to identify the product formed and explain…
Q: Calculate the energy required to heat 251.0 g of silver from -0.6 °C to 10.8 °C. Assume the specific…
A: Specific heat capacity - Amount of heat required to raise temprature by 1 degree of one gram of a…
Q: Draw the diagram of the structure of the micelles of a soap in hexane trapping a stain of sweet…
A: Micelle formation - Cleansing action of soap.
Q: 9. How many grams of Ca(OH)2 are needed to make 100.0 mL of 0.250 M solution?
A: Molarity -Moles of solute present in per liter volume of solution Molality =moles of solute…
Q: Specify reagents suitable for converting 3-ethyl-2-pentene to 2,3-epoxy-3-ethylpentane. Select the…
A: The given reaction is an example of epoxidation reaction. The correct reagent required for…
Q: 10. What is the major organic product generated in the reaction below? Ho HO..... .. (A) OH (B) 1)…
A: Ozonolysis of the double bond produces the ozonide intermediate which is further decomposed by the…
Q: Enter your answer in the provided box. A 2.49-L container of vinegar contains 6.2% (w/v) of acetic…
A:
Q: At 455 K, AG° = -28.5 kJ/mol for the reaction A (g) + 2 B (g) → 3 C (g). If the partial pressures of…
A: The question is based on the concept of chemical thermodynamics. we need to calculate Gibbs free…
Q: A chemistry graduate student is given 500. mL of a 0.70M propanoic acid (HC₂H,CO₂) solution.…
A:
Q: Suppose 0.900 mol of electrons must be transported from one side of an electrochemical cell to…
A:
Q: Using arrows to show the flow of electrons, write a stepwise mechanism for the following reaction…
A:
Q: Enough of a monoprotic weak acid is dissolved in water to produce a 0.01850.0185 M solution. The pH…
A: pH is equal to negative logarithmic value of Hydrogen ion concentration
Q: 2. 3. Нат b) HBC терва Всеу provide mechanism to form 2 major products вс вс NaCN DMSD NaOH до он -…
A: The missing boxes of the given reactions can be filled by completing the reaction. The reaction…
Q: 1. a. Write the line notation for the cell shown. b. Are electrons moving toward the Cu electrode or…
A: Copper is the anode, and silver is the cathode. b) electron is moving towards the silver electrode.
Q: A resonance structure of ozone (O3) is given blow. What is the formal charge on the central oxygen…
A:
Step by step
Solved in 2 steps

- Explain why the complex [Fe(H2O)6]3+ has Do = 14,000 cm-1, while [Fe(H2O)6]2+ has Do = 9,350 cm-1.1,Count up the 18-electron rule of the organometallic complex (Ferrocene) using covalent counting method.2. Draw the molecular orbital diagram for B2 and find the bond order?Inorganic Chemistry (i) Define spectrochemical series (ii) Explain the difference between weak-field and strong-field ligands
- 1) What role do the protons play in the transformation of [(SALEN)VO] to [(SALEN)Cl2]? --- Consider the number of anionic charges for all the ligands as well as the charge of the overall complex. Has the oxidation of vanadium changed in this reaction?Copy the structure of EDTA4- from your textbook and circle all six atoms that act as Lewis bases. Why does EDTA4- cease to function as a chelating ligand if acid is added to the solution? pls help asap!!copper complex crystallises straightaway when synthesising but the cobalt complex required the solution to be concentrated and cooled before it is filtered to obtain product. what does this tell you about the realtive solubilities of the copper and cobalt complexes.
- why [Cu(deen)2]X2(X = BF4-, ClO4-and NO3-) show thermochromic behavior, whereas most other bis-diamine Cu(II) complexes (e.g. ethylenediamine) do not exhibit this propertyConstruct an MO correlation diagram for the complex ion[Ni(NH3)6]2+ and populate the orbitals with electrons. Do youexpect p bonding to be important for this complex? Whatfeatures of this diagram correspond to those predicted usingcrystal field theory? What kind of bonding would VB theorypredict for d8 complexes, and how does MO theory accountfor the bonding more accurately, even qualitatively?Consider the [Ir2(dimen)4] 2+ complex. Notice that the dimen ligand is asymmetric. There is also a different, symmetric diisocyano ligand referred to as “TM4” (structure shown to the right) that forms the same sort of binuclear complex that dimen does. Answer the following questions with supporting justification: a. How many geometric isomers (ignore any optical ones) of [Ir2(TM4)4] 2+ are there? b. How many geometric isomers (ignore any optical ones) of [Ir2(dimen)4] 2+ are there? c. In the “twisted” deformational isomer, would you expect [Ir2(TM4)4] 2+ or[Ir2(dimen)4] 2+ to be able to rotate polarized light? Justify your answer.
- What is the expected number of Fe-Febondsper Fe atom in the complex ?Many transition metal complexes are said to follow the “18-electron rule,” meaning they form particularly stable complexes when the metal + ligand electrons contributing to the MOs add up to a total of 18 electrons. a. Use the full MO diagram for an octahedral metal complex with pi-acidic ligands we derived in class to justify why 18-electrons might be particularly stable. b. Use the 18-electron rule to predict which 1st row transition metal would be most likely to form an octahedral M(CO)6 (Note: there is no charge; the metal retains all its electrons.) c. Qualitatively, how would you expect the CO bond to change when it forms this complex? Would it be stronger, weaker, or the same compared to free CO? (Hint: think about what a ? −???? means and what the orbital does from the perspective of the M–CO interaction as well as the C– O interaction).Fill in the blanks : (i) When a ligand has a two-complexing or donor group in its structure, it is said to be————— (ii) ___________are compounds whose colour changes when they bind to a metal ion. (ii) If you double the wavelength, you will observe. ________the energy. (iv) In_____________ titration, we monitor changes in absorbance during the titration to find out the equivalence point. (v) Two liquids are_____________ if they form a single phase when they are mixed in any ratio. (vi) The efficiency of Chromatographic Columns can be described by____________Equation.



