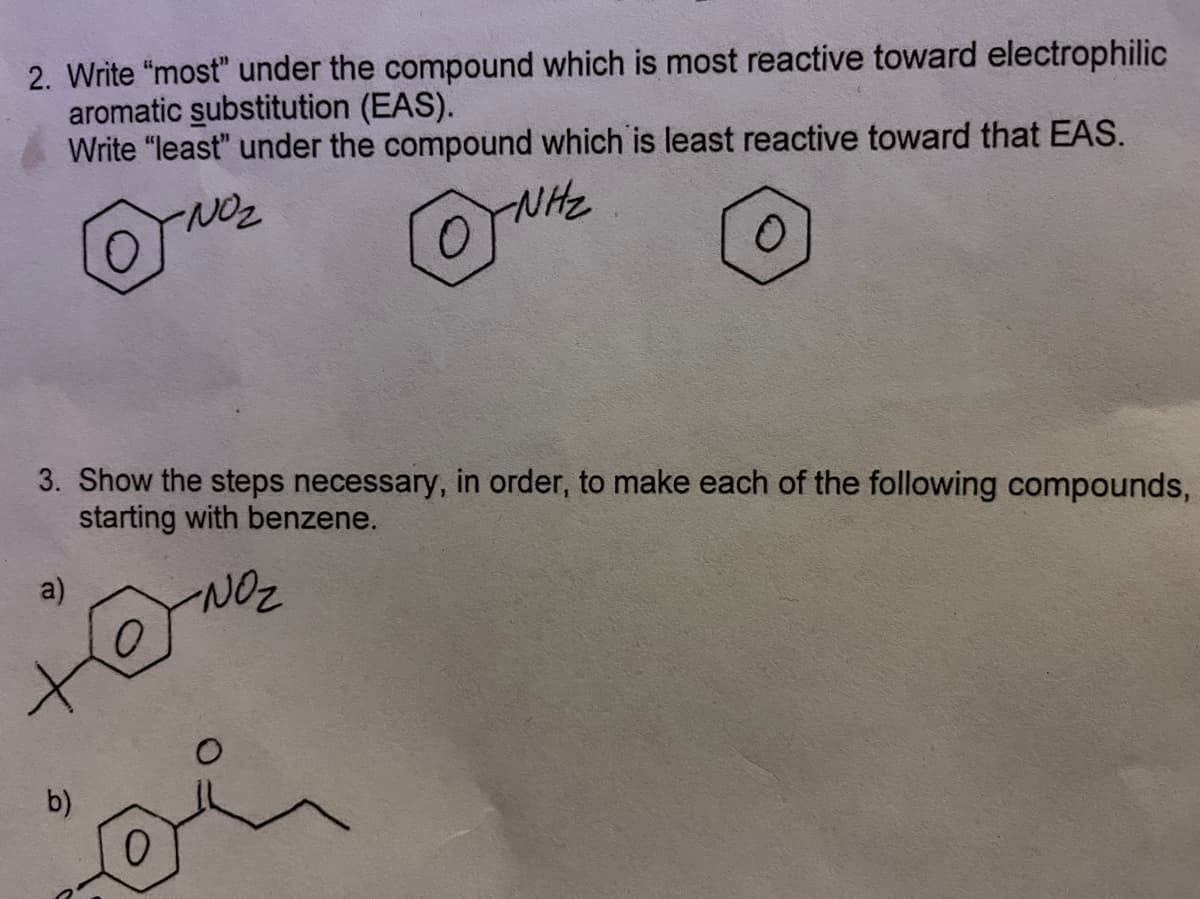
2. Write "most" under the compound which is most reactive toward electrophilic aromatic substitution (EAS). Write "least" under the compound which is least reactive toward that EAS. NHC
Q: The triphenylmethyl cation is so stable that a salt such as triphenylmethyl chloride can be isolated…
A: The species in which carbon carries a positive charge is called as carbocation. Carbocations are…
Q: Using the information below explain why CN- is a stronger Bronsted base even though in the reaction…
A: Bronsted base is the species that accept proton.
Q: The benzylic position has unique reactivity and resonance stability. Which structures can this…
A:
Q: Nucleophilic Aromatic Substitution Draw the curved arrow reaction for this experiment showing the…
A: Solutions We Know that These products can be obtained from following…
Q: Provide a short paragraph that relates the most essential elements of theory (reactivity,…
A: Electrophilic Aromatic Substitution Reaction is described below:
Q: We did not consider the possibility of water acting as a nucleophile and attacking the carbocation,…
A: Considered the electrophilic substitution at the benzene ring, when electrophile attack on benzene…
Q: 3) If the product of an SN1 or Free radical reaction contains a stereocenter then a mixture occurs.…
A: The correct answer is given below
Q: While designing a synthesis, define Reactions that convert one functional group to another—that is,…
A: It is referred to that reaction in which we are preparing a new compound using a few starting…
Q: example of reaction for each of the types of organic reactions: substitution, elimination, addition,…
A:
Q: Why alkyl groups considered as an ortho- , para-directing?
A: To find: Why alkyl groups are considered as an ortho-, para-direction
Q: The triphenylmethyl radical is highly delocalized by resonance. In the resonance hybrid, how many…
A: There are ten different positions have radical character
Q: ring with the effect of ……………… and are ……………………… directed towards electrophiles. Groups that are…
A: If any group increases electron density on benzene is ortho para directing. If it withdraw electron…
Q: Which is most reactive in electrophilic substitution? Which is least reactant in electrophilic…
A:
Q: A Moving to another question will save this response. Question 5 Which one of the following…
A: In the given compounds toluene,benzene, chlorobenzene,t-butyl benzene undergo a Friedel-crafts…
Q: what is Cannizzaro reaction, how does it work how do the reaction work based on the which one is…
A: This reaction takes place In those Aldehyde and ketone which do not have any acidic Hydrogen and…
Q: Which is more reactive for electrophilic addition ?
A: The compounds given are,
Q: Some of the groups pre-bonded to an aromatic ring cause electrophilic aromatic substitution to occur…
A: It is given in the statement that some of the attached groups on aromatic ring ( like benzene) make…
Q: Give two (2) examples of reactions for each of the types of organic reactions: substitution,…
A:
Q: Write any specific net reaction of your choice, which represents some type of reaction mechanism…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: Moecules with smaller amounts of electron delocalization will have HOMO/LUM0 gaps. This will cause…
A: Due to overlap of molecular orbital energy of HOMO increases and energy of LUMO decreases. Thus HOMO…
Q: Of the above compounds, which is least basic? Of the above compounds, which is least nucleophilic?…
A: Nucleophilicity is the tendency of the atom to donate electrons. Negative charge is more stable on…
Q: What kind of reaction is shown below? A C В Polar Organometallic Radical Pericyclic
A: Polar mean Positive and negative bonds organometallic Ex: Rmgx it is Reagent Radical…
Q: Write any specific net reaction of your choice, which represents some type of reaction mechanism…
A: Hello. Since the question contains more than three sub-parts, the first three sub-parts shall be…
Q: Select the member of each pair that shows the greater rate of SN2 reaction with KI in acetone.
A: The addition of nucleophile and removal leaving group takes place in simultaneously in SN2 reaction…
Q: Which reaction had the larger change in energy, the reaction with hydrochloric or acetic acid. Why…
A:
Q: 3. Rank the following in order by the rate in which they could participate in an SN1 reaction (“1"…
A:
Q: Rank the attached radicals in order of increasing stability
A: Free radicals refer to the type of reactive intermediate in which a homolytic cleavage of bond takes…
Q: Which statement(s) is/are true regarding leaving groups? i. nucleophilicity generally increases with…
A: THE OPTION D IS CORRECT. 1. Nucleophilicity follows basicity across a row, as basicity increases…
Q: Which will react slower in polar protic solvents
A: Polar protic solvent is polar solvent.
Q: Methylating agents are substrates that deliver methyl groups to nucleophiles, and so are themselves…
A: Methylating agent is chemical species which donates methyl ion to a nucleophile but also in some…
Q: a. Label the reactive features, highlight the most reactive one, then highlight what it needs. Also,…
A: Hydroboration reaction
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: The reaction given is,
Q: Select the member of each pair that shows the greater rate of SN2 reaction with KN3 in acetone.
A: SN2 reaction rate depends upon the steric effect. Higher the steric hindrance, lower would be the…
Q: 1) Which one more reactive to word nucleophilic substitution P- nitro toluene or O- ethyl toluene ?…
A: P-nitro toulene will be more reactive towards nucleophilic substitution than O-ethyl toulene because…
Q: (A) SNl reactions are stepwise and have intermediates. (B) The slow step in a Syl reaction is…
A: Statement A,B,C,D are correct and E statement is incorrect A) SN1 reaction is happens in two step…
Q: Explain and illustrate, why the Starting Material SM is more basic than pyrole
A:
Q: Using the resonance theory which carbocations is more stable ? And why ?
A: When a single Lewis structure fails to explain all the properties of a molecules resonance comes…
Q: What happens to the rate of an SN2 reaction under the following conditions? [RX] is halved, and…
A: Nucleophilic substitution bimolecular reaction or the SN2 is a type of reaction in which bonds are…
Q: Select the TWO most electrophilic carbons in this molecule. CI :
A:
Q: ) For your assigned letter rank the molecules provided from lowest (1) to highest (3) based on their…
A:
Q: True or false? Some of the groups pre-bonded to an aromatic ring cause electrophilic aromatic…
A: When a group is attached to aromatic ring, then depending on the type of group attached , it can…
Q: Indicate the true electrophile that Aromatic Ring in Nitratien
A: Nitration is a process by which a nitro group is added to an organic compound. Here, the organic…
Q: Moecules with larger amounts of electron delocalization will have HOMO/LUMO gaps. This will cause…
A: Molecule with large numbers of overlapping, will increase the energy of HOMO and decreases the…
Q: What is the correct order of the words in the blanks in the following sentences? If there are…
A: The different properties of the substituents on the benzene ring depends upon the type of group…
Q: By which substitution mechanism (SN1 or SN2) did the reaction occur? How did you know? For what…
A: The reaction is happened via SN1 mechanism. In SN1 mechanism first leaving group leaves in rate…
Q: Predict the site on each molecule that is most likely to undergo electrophilic aromatic…
A: (a)
Q: How do I rank these from best to poorest leaving group? Do I base it in the size of the…
A: We have to rank leaving group tendency :-
Q: Which reaction will give an alkene product that will favor anti-periplanar geometry?
A: Given : types of reaction
Q: APPLY STEREOCHEMISTRY WHEN APPLICABLE: Does each reaction occur from SN1 or 2? What are the organic…
A: the solution is as follows:
Q: What happens to the stereochemistry during and SN2 reaction? Why? Provide a reaction to illustrate…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Need help with Part 2: select all the appropriate resonance structures that result from the first step that is reproduced below.On the square, indicate the number of sites of unsaturation of the following molecules, show your work with one of the structures by applying the appropriate formula and showing the number (which should match the number on the square).OChem help with the following reaction schemes (see attached image) Please provide the bond line structures for the products obtained in the following reaction schemes... Notice in question #1 that A, B, and C are actually shown as C, B, and finally A in the actual reaction scheme so C correlates with PCC, and so on Question #2 is in order.
- By which substitution mechanism (SN1 or SN2) did the reaction occur? How did you know? For what reason does the substitution occur at only one of the bromine atoms, and in particular the bromine atom that it did?What is the correct answer here? Answer key says D but it doesn't make sense to me because the final one (5) is tertiary allylic.organic chemistry 1 Provide an overall order of carbocation stability, including every type of carbocations. (Not just primary< secondary<tertiary!) please help me this question, thank you!
- Please explain question 1 and 8 1. How many degrees of unsaturation are present in your unknown monosubstituted benzene starting material? 8. Based on all of the data provided, what is the identity of the “G” group? (Please NEATLY & CLEARLY draw its full Lewis structure in the space provided) Thank you!For each of the following, write the major product(s) and then draw out each step in the mechanism using curved arrows. Show ALL lone pair electrons and formal charges. Redraw ALL molecules as to show explicitly ALL bonds being broken or formed. Identify the molecular orbital (HOMO) of the nucleophile and the molecular orbital (LUMO) of electrophile involved in the nucleophilic attack. MO diagrams are not necessary..Kepone, aldrin, and chlordane are synthesized from hexachlorocyclopentadiene and other five-membered-ring compounds. Show how these three pesticides are composed of two five-membered rings.

