Q: The unknown compound with molecular formula C4H8O3, has infrared absorptions at 1710 and 2500 to…
A:
Q: Analysis of a sweet-smelling, neutral compound of carbon, hydrogen and oxygen produced the following…
A: Signals of proton NMR shows the number of equivalent protons present in the organic compound.
Q: The unknown compound that gives the following IR and NMR spectra has the formula CaH1,0. Based on…
A:
Q: Propose a structure for compound X (molecular formula C6H12O2), which gives a strong peak in its IR…
A: Given spectrum,
Q: (а) The IR spectrum below shows the important absorption bands for compound A (C7H1402). Its 'H NMR…
A:
Q: 4000 3000 2000 1500 1000 500 Wavenumbers (cm) 3H 2H 2H 2H Exchanges with Dz0 11 10 9 8 4. ppm 170…
A:
Q: A compound has a molecular formula of C5H802 and exhibits the following 13C NMR spectrum. 8 171.48,…
A: SOLUTION: Step 1: The given compound contains 5 C13 NMR signals, among the given structures B and C…
Q: The 1H- and 13C-NMR data of an ester of molecular formula C6H10O2 are given below. Also shown are…
A:
Q: Identify the unknown compound using the empirical formula C5H10O2, the IR spectrum and the nmr.
A: Given that, the empirical formula of the compound is C5H10O2 So, double bond equivalence (DOB) of…
Q: In addition to the NMR spectrum below, this compound, with formula C5H,0O2, has bands at 3450 cm 1…
A: The structure of the following is:
Q: 3000 * TRANSMITTANCE 6CCC LS62 1248
A: Provided molecular formula of compound=C4H10O Broad ir peak at =3339 cm-1 Provided 6H doublet at…
Q: Suggest structures
A: H-NMR spectra is the type of spectroscopy that identifies only the type and the number of hydrogen…
Q: :) Propose a structure for compound A that satisfies ALL the data provided: (GIVE REASONS FOR YOUR…
A: Note : MF = C5H10 (DBE= 5-10/2+1 = 6-5 = 1). Compound A do not decolorizes the bromine solution…
Q: Propose structures for the compound C5H10O whose 1H NMR spectra is attached
A:
Q: A compound has a molecular formula of CgH10 and exhibits the following 13C NMR spectrum. 8 144.24,…
A: The 13C given for compound has 6 signals. means the compound have 6 different types of carbons. So…
Q: 9. Which of the following structures is consistent with the IR spectra show E0 40 20 3500 3000 2500…
A: Infrared spectroscopy (IR spectroscopy or vibrational spectroscopy) is the measurement of the…
Q: A compound has a molecular formula of C5H802 and exhibits the following 13C NMR spectrum. 8 171.42,…
A: SOLUTION: Step 1: The molecular formula is C5H8O2. In C13 NMR spectrum, the number of signals is…
Q: Propose a structure for a compound of molecular formula C3H8O with an IR absorption at 3600–3200…
A: Calculation of double bond equivalence: DBE = C +1 – (H + X – N) / 2 Here, C is the number of carbon…
Q: An aromatic compound K, whose molecular formula is C8H11N, is examined in the laboratory to…
A: The chemical structure of the compound K, L and M are needed to predicted given that the compound K…
Q: Predict the structure of a compound based on this 13C NMR spectra. The chemical formula of this…
A: The given molecular formula: C4H6O2 The degree of unsaturation = C-H/2+N/2+1 Where C= Number of…
Q: Compound X of the molecular formula C7H10 has the 13C NMR spectrum (5 signals) shown below. On…
A: DBE = 7- (10/2)+1 = 8-5 = 3. Therefore the compound is having one ring and two double bonds.…
Q: From the following 1H-NMR and 13C-NMR spectra, assign a structure for a compound with a formula of…
A:
Q: 2 (a) Compound A has the molecular formula of CSH10O. The IR and 'H NMR spectra are given as…
A:
Q: Following are IR and 1H-NMR spectra of compound D. The mass spectrum of compound D shows a molecular…
A: The structural formulas for ions in the mass spectrum at m/z 118, 107, and 59 has to be given,
Q: Deduce the structures of compounds A and B, two of the major components of jasmine oil, from the…
A: IR absorbtion at 3091 - 2895 corresponds to aromatic double donds and 1743 corresponds to an ester…
Q: Thymol (molecular formula C10H14O) is the major component of the oil ofthyme. Thymol shows IR…
A: Given data: The molecular formula of the compound is C10H14O IR absorption bands are 3500–3200,…
Q: A hydrocarbon containing 85.5% carbon and 14.5% hydrogen is transparent above 210 nm in ultraviolet…
A:
Q: Propose a structure for a compound of molecular formula C7H14O2 with an IR absorption at 1740 cm−1…
A: The given compound has the molecular formula C7H14O2 and it shows an absorption band in the IR…
Q: 1H NMR 3. HO HO 4. (1) D PPM 9. HO 10 но 11 (2) J PPM
A: IR spectra deals results from the interaction of infrared radiation with matter by absorption. IR…
Q: Suggest structures given the 1H NMR spectra and formulas for each of the compound below. C3H7Cl
A:
Q: 1H NMR spectra and formulas
A: Given data, C9H10O The structure of the given proton NMR spectra can be identified as
Q: A compound with the molecular formula C6H11BrO2 shows an intense band at 1739 cm-1 in the IR…
A: Given: An intense IR spectrum at 1739 cm-1, then it should be a ester functional group. according to…
Q: Below is the 1H NMR spectrum for an ester with molecular formula C6H12O2. Draw the molecular…
A:
Q: Br çO,Me vs. čOH Br он он vs.
A:
Q: Propose a structural formula for compound A, C,H,N, given its IR and 'H-NMR spectra. Micrometers 25…
A: Degree of unsaturation is given as- DOU=#Cs+1-(#Hs-N)2 where #Cs: Number of carbon present. #Hs:…
Q: What are the major IR bands, 1H-nmr signals, And the m/z of the following compounds benzaldehyde
A: Given: benzaldehyde The structure of benzaldehyde is as follows,
Q: The IR spectrum, 1H NMR spectrum, and 13C NMR spectrum for the unknown compound with the formula…
A: IR, NMR together helps in interpretation of an organic compound.
Q: Calculate the IHD and explain (elucidate) the structure using the 1H and 13C NMR data.
A:
Q: The infrared spectrum of a compound has a strong absorption at 1669 cm. The mass spectrum of the…
A: Mass spectrum is a technique which is used to identify the compound structure. In this technique…
Q: The proton NMR spectrum of a compound with formula C8H14O2 is shown. The DEPT experimental results…
A: The given compound has a double bond equivalent of 2. The two strong bands obtained in the IR…
Q: 11. Provide a structure for Compound A, (CsH₁0O), based on its IR and NMR spectra. Show full…
A: Given the molecular formula of compound-A is C8H10O We are given the IR spectra and 1N NMR spectra…
Q: 11. Provide a structure for Compound A, (CsH₁0O), based on its IR and NMR spectra. Show full…
A: The molecular formula of the organic compound given is C8H10O. The degree of unsaturation =…
Q: Analyze the 1H AND 13C NMR spectrum of C8H9NO given below and draw the structure of the compound.
A: By looking at the no. of signals in 1HNMR and 13CNMR, we can infer the basic carbon framework. The…
Q: Deduce the structure of the compound that gives the following 1H, 13C, and IR spectra. Assign all…
A: IR spectrum : a. Medium peak at 3040 cm^-1 corresponding to aromatic C-H stretching vibration. b.…
Q: 13. The molecular formula of compound Y is CSH100. From its NMR spectra, determine the structure of…
A: Given molecular formula of Y: C5H10O Given NMR spectra: We have to find the structure of Y.
Q: There are several isomeric alkanes of molecular formula C6H14.Two of these exhibit the following…
A: Isomerism is the phenomenon in which distinct compounds with the same chemical formula exist side by…
Q: What is the structure of the compound with the formula C5H12O, if it has a strong broad IR signal…
A:
Suggest structures given the 1H NMR spectra and formulas for each of the compounds below.
C4H10O
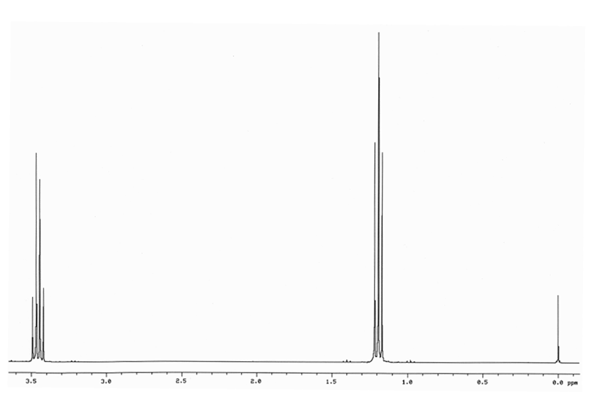
Step by step
Solved in 3 steps with 3 images


