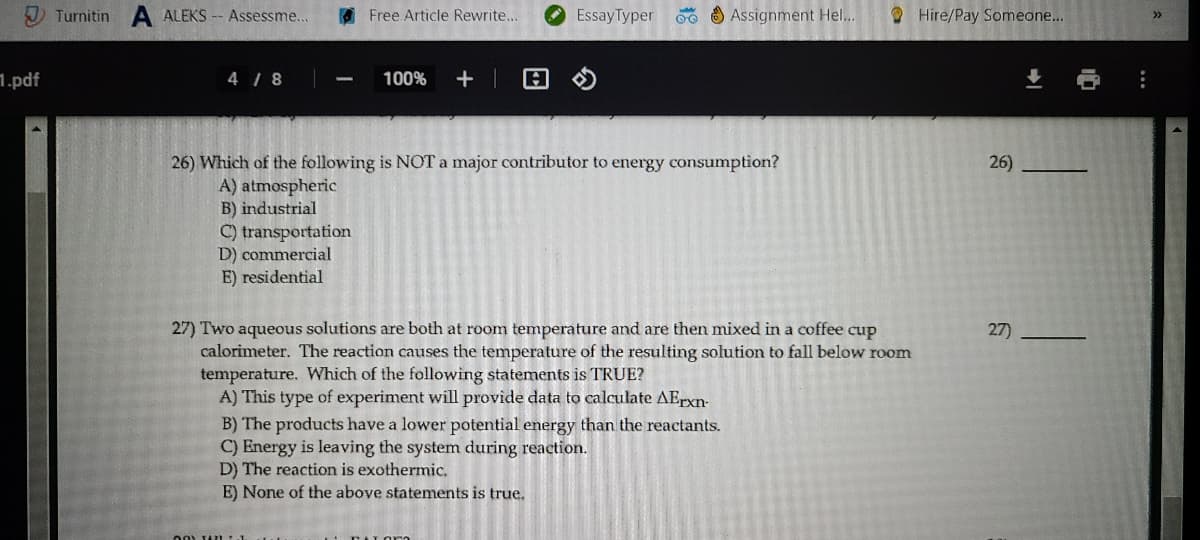
26) Which of the following is NOT a major contributor to energy consumption? A) atmospheric B) industrial 26) C) transportation D) commercial E) residential
26) Which of the following is NOT a major contributor to energy consumption? A) atmospheric B) industrial 26) C) transportation D) commercial E) residential
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 80AP
Related questions
Question
Transcribed Image Text:Turnitin
ALEKS -- Assessme...
O Free Article Rewrite...
Essay Typer
00 O Assignment Hel...
2 Hire/Pay Someone...
>>
1.pdf
4 / 8
100%
會
26) Which of the following is NOT a major contributor to energy consumption?
A) atmospheric
B) industrial
C) transportation
D) commercial
E) residential
26)
27) Two aqueous solutions are both at room temperature and are then mixed in a coffee cup
calorimeter. The reaction causes the temperature of the resulting solution to fall below room
temperature. Which of the following statements is TRUE?
A) This type of experiment will provide data to calculate AErxn-
B) The products have a lower potential energy than the reactants.
C) Energy is leaving the system during reaction.
D) The reaction is exothermic,
E) None of the above statements is true.
27)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning