Q: Standardization of a sodium hydroxide solution against potassium hydrogen phthalate (KHP) yielded…
A: Answer: This question is based on acid-base titration where with the help of given values of KHP we…
Q: emperature (°C) 22°c Titration 3 Titration 1 Titration 2 Volume of Saturated Calcium 10:00 ml…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: It is used to deliver a small quantity of distilled water 2. Reagents whose label of containers are…
A: To write one word for the given sentences:
Q: Complete the table. Questions IUPAC name Common name cinnamon bark acid that can be extracted…
A: Answer: In the given question we have to find out the main chemical compound present in the given…
Q: What is the mass of carbon present in 0.283 grams of propane (C3H8)?
A: Given data : Mass of propane = 0.283 gram To find : Mass of carbon present in 0.283 grams of…
Q: For each set of three structures, choose the structure that is not an isomer of the other two. но, C…
A:
Q: The redox potentials for the Zn2+/Zn and Cl2/Cl– couples are -0.764 V and 1.395 V, respectively.…
A:
Q: [Review Topica) [References) Use the References to access important values if needed for this…
A:
Q: Metals have properties that can be used for various applications. Determine the property of metals…
A: Metal electrodes have been found to carry the highest number of ions; thus, pure metal is the most…
Q: The following compounds contain a Carbonyl group, except Acetaminophen Citric Acid Phenol…
A: First, we see structure of the given molecules
Q: How can you find the moles and the KSP of the of acid and base of a titration experiment from the…
A: We have find out Moles of HCL, Moles of Analyte and KSP value.
Q: The IUPAC name for the following molecule is: Br но ÕH (3R, 4R)-3-bromo-4-hydroxyhexanal (3R,…
A: Every chiral center has a certain configuration that R or S. Configuration of chiral center used in…
Q: A 0.01kg plant extract in 2dm' of water was shaken with 0.1dm' ether. Upon checking, 1.7x10 kg of…
A: Mass of plant extract = 0.01 kg = 10 g
Q: Explain the reaction by observing the sets of molecular collisions below.|
A: Given:
Q: The tetragonal and hexagonal crystal systems produce the crystal form. O Tetrahedron Scalenohedral O…
A: Here option 'b' is correct option. 1. Tetrahedron will not be correct option as it forms with a…
Q: 7. Define back-titration. What is the concept behind this method?
A:
Q: A galvanic cell is powered by the following redox reaction: 2 MnO4(aq) + 4 H,0(1) + 3 Zn(s) 2+ 2…
A: Given reaction is : 2MnO4- (aq) + 4H2O (l) + 3Zn (s) ------> 2MnO2 (s) + 8OH- (aq) + 3Zn2+ (aq)…
Q: 1. Aqueous dilution of I0, results in the following reaction: 10. jac) + 2H,O H,lO. (ac) and K = 3.5…
A: GIVEN:-
Q: 2.49 g CuO + 5.05 g H2SO4 which reactant would limit the quantity of copper (II) sulfate that could…
A: Limiting reagent is the reagent which is consumed in the chemical reaction completely. It decides…
Q: e length
A:
Q: For each of the following reactions which of substitution products is correctly identified. There…
A: SN1 reaction always give enantiomeric product
Q: describe the position of the equilibrium when we mix equal amounts of N2, H2 and NH3 at 400K. The…
A:
Q: Chemistry The exchange current density for the electrode Pt(s) │|H2(g) │|H+(aq) is 0.79mA cm-2 at…
A: We have find out current density and anode current.
Q: Draw the structure of the given compound. Identify if the given name is correct of incorrect. If…
A: In this question, we have to draw the structure of the given compounds and also name the correct…
Q: Given an average carbon dioxide concentration of 400 ppmv, estimate the mole fraction, partial…
A: Carbon dioxide concentration = 400 ppmv C mgm3 = ppmv ×PR T ×mw C mgm3 =400 ×10.0821×298 ×44…
Q: 4. Which among the following refers to the value of internal energy. A 1025 J of heat energy is…
A: First law of thermodynamic says Q = ΔE + W
Q: Draw the attraction between a water molecule and a molecule of PCl3 First, draw one molecule and…
A: Let us discuss the structure of both tge molecules and then their bonding.
Q: Need solution to all if not answered I'll downvote solution Which of the reactions are…
A: Redox reactions are those reactions in which both oxidation and reduction take place simultaneously.…
Q: Draw the enolate anion and the ketone or aldehyde that would be needed to make this product through…
A: Given structure of alpha-beta unsaturated ketone is : Draw the enolate ion and the ketone or…
Q: 4. At 50°C, the vapor pressure of CS2 is 854 Torr. A solution of 2.0 grams of sulfur in 100.0 gm of…
A: Relative lowering vapor pressure (RLVP) :- The ratio of lowering of vapor pressure to vapor pressure…
Q: Q2. What is product for the following reaction? Click the correct structure Hint: This is called an…
A:
Q: Number of moles of sodium sulfate dissolved in 2 L of a 0.12 M sulfate, Na2SO4solution
A: Given :- molarity of Na2SO4 solution = 0.12 M Volume of solution = 2 L To calculate…
Q: Discuss in detail TWO types of invention that can be use to measure pH either quantitative and…
A: HYDROGEN ELECTRODE METHOD A hydrogen electrode is made by adding platinum black to platinum wire or…
Q: Compound with 103 x 6.74 molar (3) absorption What concentration of the compound is required to form…
A:
Q: If Z = 1.00054 at 0 °C and 1 atm and the Boyle temperature of the gas is 107 K, estimate the values…
A:
Q: 4- Propose a structure that is consistent with each set of 'H NMR data. IR data is provided for some…
A:
Q: PS#3 part 2- Synthesis: Given this group of simple compounds, devise a plan to synthesize each…
A: We have to snthesize the following given compounds from the given starting materials in multistep…
Q: Help only needed on D. Thank
A: We know epoxide ring due to ring strain is unstable and undergoes ring opening reaction even in weak…
Q: (3, 0, U, +2) 4 Write the kondensed ground state electron conng. for the following elements using…
A:
Q: d. After subtracting 2 electrons per is e. The number of electrons needed atom in the molecule is f.…
A: In NH₄⁺ ion, N atom forms 4 N-H bonds and thus it has 8 electrons in its outermost shell.…
Q: What is the correct IUPAC name of the structure presented. (Follow this format "Branch-Carbon…
A:
Q: To study the effects of a weakly acidic medium on the rate of corrosion of a metal alloy, a student…
A: First we would Calculate Kb value using Ka of acetic acid. Then first we write chemical equation…
Q: [M]=/
A: In presence of Alkylidine or metal carbene multiple bonded molecule, metathisis reaction is…
Q: For ferricyanide determination by voltammetry, 20 mL of non-electroactive KCl solution is added to…
A: Millimoles of ferricyanide added=Molar concentration of ferricyanide added×Volume of ferricyanide…
Q: vhat symmetry representation label is associated with it. Show all of your work. you will need to…
A: C-H asymmetric stretching (bu)3172 cm-1(IR intensity = 0.77)(Raman inactive) C-H symmetric…
Q: UESTION 4 All of the following are true of SN2 reactions except: The rate varies with the…
A: SN2 reaction nucleophilic substitution via 2nd order. This substitution reaction depends on both…
Q: Draw the attraction between a water molecule and a molecule of BrCl4F
A:
Q: Now let us look at a real amino acid, alanine. Fill in the chart below for each ionizable group. You…
A: alanine is an amino acid so it has both the basic amine group and acidic carboxylic acid. Here we…
Q: Draw the structure of an alcohol the best fits the description below. An open chain C5 alcohol that…
A: Given, Draw the structure of an alcohol the best fits the description below. An open chain C5…
Q: n רוו AROMATIC (10 e) ANTI-AROMATIC (8 e)
A: Given: Aromatic Anion (10e) and anti- aromatic cation (8 e)
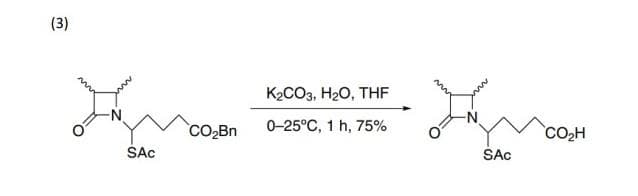
Step by step
Solved in 3 steps with 2 images

- 10.5mL (mw 106) of benzaldehyde and 2.9g(3.63mL, mw 40) of acetone react with 5g of NaOH and 25mL of Ethanol to form 6.7g of Dibenzalacetone. What is the theoretical, actual and percent yield?If 850 mL of syrup with a specific gravity of 1.5 contains 500 g of sucrose, what is itspercentage strength (w/w)?1. Which of the following compounds can form hydrogen bonds with water?a. octaneb. HClc. NaCld. acetic acid2. Maria wants to determine the percent water content of an unknown hydrate of AB. Given the following data:mass of empty crucible - 12. 2784gmass of AB · xH20 + crucible - 17.4960gmass of AB + crucible - 15.1314gCalculate the % water content of the hydrate3. From the above data, give the correct formula for the hydrate. (AB₂ Molar mass = 129.84 g/mol)
- Please help me with tyhis problem and the right sig fig because I got as the molar of the compound 199.886 so I rounded to 200 and it was wrong so what is the correct mount of sig fig I should use? thanksWhat is the final concentration of hydrocortisone in the mixture, as a ratio strength, if 5 g of hydrocortisone 2.5 % (w/w) ointment is added to 75 g of petroleum jelly? Round denominator of ratio strength to a whole numberis the A:B ratio just a place holder for the unknown compound can you use other variables in place of "A and B" also would H2O work as another formula instead of Na2O
- 1-Pentanol to 1-bromopentane Chemicals: - 60ml Conc. Sulfuric Acid - 100ml Saturated Sodium bicarbonate - 65ml 1-Pentanol - 78g sodium bromide - Distilled water - 58.42g 1-Bromopentane 1-Pentanol Sodium Bromide Sulfuric Acid 1-Bromopentane Formula C5H12O NaBr H2SO4 C5H11Br MW (g/mol) 88.15 102.894 98.078 151.04 Density (g/mL) 0.811 3.21 1.84 1.218 Boiling point (*C) 138 1,396 337 130 NaBr(aq) + H2SO4(aq) -> NaHSO4(aq) + HBr(aq) CH3(CH2)4OH(aq) + H+ Br- (aq) CH3(CH2)4OH2 (aq) + Br-(aq) CH3(CH2)4OH2 (aq) + Br-(aq) CH3(CH2)4Br(aq) + H2O(aq) How do I calculate the percent yield and identify the limiting reagent?For each of the following lists, rank the molecules with respect to the indicated properties from highest / best (1) to lowest /worst (4) by writing the numbers on the lines.What is the density of 47.0 wt% aqueous NaOH (FM 40.00) if 17.6 mL of the solution diluted to 1.80 L give 0.172 M NaOH?
- Stock iron(II) solution (200Ug mL-1 Fe) ferrous ammonium sulfate hexahydrate mass= 0.1437g, transfer it to a 100 ml beaker. add 15 ml approx of water and 15m1 'approx of dilute sulphuric acid (2M H2SO.). then transfer FeII to 100 ml flask makeup to the mark with water. calculate the moles of ferrous ammonium sulfate hexahydrate solution in unit ug/mL.4) please FINISH IN 3 SIG FIG19 g of unknown organic sample was dissolve in 640 mL of Dicloromethane (DCM). The boiling point of benzene was increased by 3.78oC. Determine the molecular weight of the unknown sample? Kb of DCM = 2.42oC/m Bb of benzene = 39.6 oC density of benzene = 1.33 g/mL at 25 °C Round your answer to the nearest whole number, no units required.



