Q: CaCl(aq) + 2 AgNO3(aq) Ca(NO3)hl ) + 2 AgCI( ) Select all the species that would be shown in the…
A: Balanced chemical reaction CaCl2 (aq) + 2 AgNO3 (aq) → Ca(NO3)2 (aq) + 2AgCl (s) In solution,…
Q: A flashbulb of volume 2.80 mL contains O2(g) at a pressure of 2.30 atm and a temperature of 27.0 °C.…
A: We have to calculate the mass of oxygen gas.
Q: For pentane, the AH° of vaporization is 26.22 kJ/mol and the AS° of vaporization is 87.88 J/mol·K.…
A:
Q: A 0.50-mol sample of oxygen gas is confined at 0 °C and 1.0 atm in a cylinder with a movable piston.…
A: According to the combined gas law, P1V1T1=P2V2T2 Where P1 and P2 are the initial and final…
Q: Draw the possible stereoisomers of 2-methylocta-4,6-dien-1-amine. Note that E-, Z- isomers of each…
A: The stereoisomers of a given compound are the combination of both optical and geometrical isomers E…
Q: Calculate the mass of dinitrogen tetroxide (N,O,) that contains a trillion (1.000 × 10'2) oxygen…
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: 300.0 mL of 0.400 mol/L KOH is added to 300.0 mL of 0.400 mol/L HF. The resulting neutralization…
A:
Q: . Write the charge balance for a solution containing 0.01M K;50, and 0.005M NaOH. In your charge…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 1. Provide the products of each transformation below. Show all stereoisomers. HBr NaOEt EIОН, heat…
A:
Q: 1. The polymer below resulted from 1,4-polymerization. Provide an unambiguous structural formula for…
A: #Q.1: Since the given polymer has only C and H, it is made of hydrocarbon monomers. Since there is…
Q: Use the diagram below to answer numbers 5-6 Solid Liquid Gas 5. Which of these conclusions can be…
A:
Q: Which of the followings are state functions? 1. Internal Energy 2. Gibbs free energy 3. Heat 4.…
A: Please find your solution below : State function is defined as the property which depends only on…
Q: 1. Any sample of matter has mass and takes up space. The main reason for this is because: * a. All…
A:
Q: substances dissolved in water shown below, determine whether it forms or does not form a buffer…
A: Buffer solution is the mixture of weak Acid/base with its conjugate base/acid. Acidic buffer is…
Q: 10.0 ml of .15M H3A (Ka1 = 2.5 x 104, Ka2 = 3.5 x 10-8, Ka3 = 5.0 x 10-12) is titrated with .10M…
A: The titration curve for the titration of 10.0 mL of 0.15 M triprotic acid H3A with 0.10 M NaOH…
Q: Which of the following is/are neutral? a. tap water b. Solution with pH = 6.5 c. Solution with pH…
A:
Q: What depth of mercury (in m) creates a pressure of 1.23 atm?
A: The depth of mercury (height) and pressure are related by the formula- P = density of mercury x g x…
Q: OH OH NAME: А. Br SHOW THE EXPANDED CONDENSED FORMULA:
A: We have one chemical compound with some functional groups. Nomenclature of such compounds is given…
Q: How many milliliters (mL) of a 0.630 M NaOH solution are needed to neutralize 20.0 mL of a 0.255 M…
A: The balanced chemical reaction for the acid base neutralization reaction is as below- 2NaOH + H2SO4…
Q: 1. The polymer below resulted from 1,4-polymerization. Provide an unambiguous structural formula for…
A: The monomer has one additional bond compared the single unit of a polymer
Q: Consider the molecule below. Of the indicated bonds, which would be expected to exhibit the lowest…
A: We have to find the bond with the lowest stretching frequency.
Q: 8:37 : Question 21 of 23 Submit Construct the expression for Ksp for solid Bilz in aqueous solution.…
A:
Q: 13. You are now well known as someone who can tell the target of a compound from only its structure.…
A: Interaction of small molecules with body biomolecules
Q: Biological reduction of Pt(IV) can occur which of the following? O Outer sphere mechanisms only O…
A: Ans. Option (e) is right. Both outer and inner space mechanisms. It is widely reported that the…
Q: The following reaction scheme is given: Aluminum metal added to a strong base aqueous solution…
A: A question based on qualitative analysis that is to be accomplished.
Q: Calories
A:
Q: 11.41 The aroma, taste, and general quality of wine are tied closely to the stage of development of…
A: We have to carry out the synthesis of (E)-2-hexenal starting from 1,1-dibromopentane.
Q: I need to write about the mechanism and how it happens in this lab please help me explain what…
A: In the above reaction hydroboration takes place where CHO group reduced to -CH2OH.
Q: • Calculate the frequency in Hertz, the energy in Joules, and the energy in electron volts of an…
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please…
Q: Calculate the ratio of the effusion rates of N2 gas and 02 gas.
A: Effusion rate of a gas is given by the Graham's law of effusion. According to the the Graham's law…
Q: Potassium ion is present in foods and is an essential nutrient in the human body. One of the…
A: Please repost the question to solve part 3. of the question.
Q: Which law of thermodynamics best explains why dissolving ammonium nitrate in water is spontaneous…
A: The first law of thermodynamics describes that the energy of the universe remains constant and it…
Q: What is the pH of a 0.35 M solution of trimethylamine, (CH3),N (Kb = 7.4 x 10 5)?
A: Given : concentration of (CH3)3N solution = 0.35 M Kb for (CH3)3N = 7.4 × 10-5 To calculate :- pH…
Q: A sample of argon gas occupies a volume of 8.67 L at 53.0°C and 405 torr. If the volume of the gas…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: F77612.SP 100.0- 90- 80 70 60- 50 40- 30 20- 10 0.0. 3500 3000 1300 1600 S400 1200 S000 00 4000.0…
A: IR spectrum is the fingerp[rint of a molecule
Q: 15 mg/L of compound X is
A:
Q: Provide complete mechanisms and the products for each step of the following Robinson annulation (3…
A: We have to carry out the given reaction. This is an example of Robinson's annulation reaction.
Q: Balance the following in an acidic solution: 103 + SO32-) - 12 + SO42-)
A: Balanced the given redox reaction under acidic solution---
Q: Tennis balls are usually filled with either air or N2 gas to a pressure above atmospheric pressure…
A: Given, volume=144 cm3 = 144 ml = 0.144 liter.. Mass (w) = 0.33 g Molar mass of N2 = 28 g/mol...…
Q: Illustrate all the locations where 2-methylocta-4,6-dien-1-amine could form hydrogen bonds with…
A: Structure of 2-methylocta-4,6-dien-1-amine
Q: Use the unbalanced neutralization below and the periodic table to calculate the answer. H,PO, (aq) +…
A: 1. Given ;- H3PO4(aq) +Mg(OH)2(aq) → Mg3(PO4)2(s) + H2O(l) Amount of H3PO4 = 7.49 g To calculate…
Q: To find the volume of a flask, it is first evacuated so it contains no gas. Next, 2.4 g CO2 is…
A: given :- mass of CO2 gas = 2.4 g temperature = 33°C pressure = 740.0 mm Hg to calculate :-…
Q: From a series of five data points concerning a gaseous reaction, a plot of In(K) vs (1/Temp)…
A: Concept: Van't half equation:
Q: mixture of 64.3 g of oxygen and 65.7 g of helium has a total pressure of 749 mmHg. What is the…
A:
Q: A Het ion corresponds to the principle quan- tum number n = 6. How many states are there? The last…
A: According to Aufbau principle, electrons are filled in lower energy states first. Order of energy :…
Q: How much heat will be absorbed when 13.7 g of nitrogen reacts with excess O2 according to the…
A:
Q: A 1.00 L solution contains propanoic acid (C3 Hs O2 H) with a concentration of 0.210 mol/L, and…
A:
Q: Styrene (shown below) has 8 t electrons but is aromatic. Why?
A:
Q: The Ksp of Pbl2 is 8.4 x 10^-9. The molar mass of Pbl2 is 461.03 g/mol. The mass solubility (g/L) of…
A:
Q: Calculate the gravimetric factor used to obtain the % Ca when if your final precipitate is…
A: Given that - Compound - CaC2O4-H2O And, atomic weights of different elements are - Ca = 40, C =…
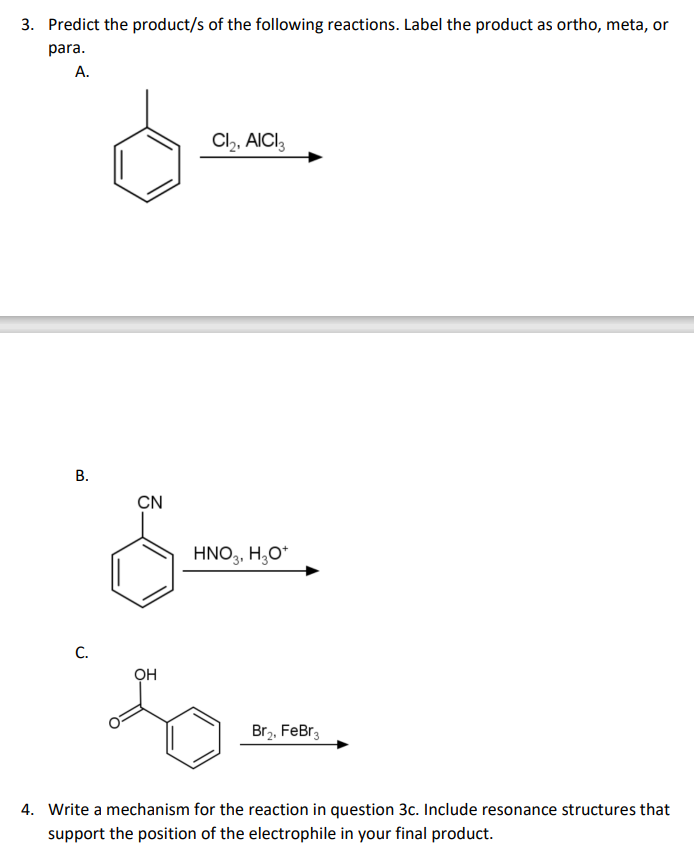
Step by step
Solved in 4 steps with 3 images

- the reactions should be all covered in organic chemistry loudon 6 th edition , for exmaple, clemensen/wolf -kishnerFill in what the structures in the boxes should be. This appears to be SN1 to get to the first box, E2 to get to the second, and electrophilic addition to get the final product. Assuming that that's correct, yes it's great I got that, but I'd like to confirm how the whole thing should look.I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?
- A haloalkane reacts with a strong base to yield only one alkene product. Select the possible structures that can give this result from Figure 9. * A B C DGiven this organic synthesis, is there any limiting and excess reagents? also describe the procedure is run, how is the reaction monitored?Is the order fo addition important? N-ETHYLALLENIMINE[Aziridine, 1-ethyl-2-methylene-] Submitted by Albert T. Bottini and Robert E. Olsen1.Checked by Thomas H. Lowry and E. J. Corey. 1. ProcedureCaution! This preparation should be carried out in a good hood to avoid exposure to ammonia. The operator should wear rubber gloves and protective goggles because 2-haloallylamines and ethylenimines can cause severe skin and eye irritation. A 2-l. three-necked flask is fitted with a sealed mechanical stirrer, a gas-inlet tube, and a dry ice condenser protected from the air by a soda-lime drying tube (Note 1). The system is flushed thoroughly with dry ammonia, and 32.8 g. (0.84 mole) of sodium amide (Note 2) is added to the flask. The system is again flushed with ammonia, the condenser is provided with dry ice covered by acetone, and 1.2 l. of liquid…What is the main organic product (B) of the reaction scheme in Figure 11?
- Assign the stereochemical configuration (E or Z) for the alkene below. Show your work, indicating clearly which groups are assigned high priority (e.g., through assigning the groups numbers, circling only the high priority groups, or labeling groups as high or low).In the beginning of this video https://www.youtube.com/watch?v=9Ng6Zv9oLzk, what reason is given on why the dienophile likes the diene?In SN2 reactions of haloalkanes, the order of reactivity is RI>RBr>RCl>RF. Alkyl iodides are considerably more reactive than alkyl fluorides, often by factors as great as 106. All 1-halo-2,4-dinitrobenzenes, however, react at approximately the same rate in nucleophilic aromatic substitutions. Account for this difference in relative reactivities.

