30. Select the letter of each image (A, B, C, etc.) that best reflects the microstructure at 700°C for each composition sample listed in the table below. (Assume that copper-rich phases are dark.)
30. Select the letter of each image (A, B, C, etc.) that best reflects the microstructure at 700°C for each composition sample listed in the table below. (Assume that copper-rich phases are dark.)
Welding: Principles and Applications (MindTap Course List)
8th Edition
ISBN:9781305494695
Author:Larry Jeffus
Publisher:Larry Jeffus
Chapter26: Welding Metallurgy
Section: Chapter Questions
Problem 11R: Using Figure 26-18, answer the following questions: a. At approximately what temperature does a 70...
Related questions
Question
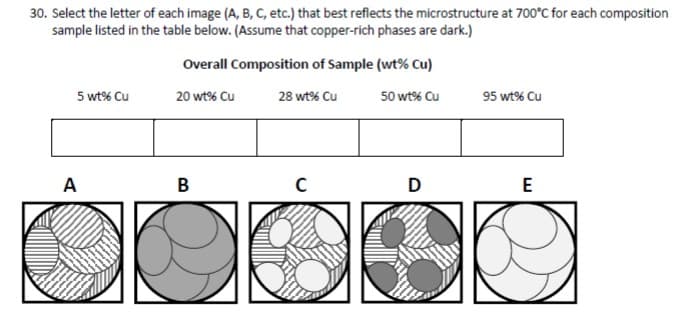
Transcribed Image Text:30. Select the letter of each image (A, B, C, etc.) that best reflects the microstructure at 700°C for each composition
sample listed in the table below. (Assume that copper-rich phases are dark.)
Overall Composition of Sample (wt% Cu)
5 wt% Cu
20 wt% Cu
28 wt% Cu
50 wt% Cu
95 wt% Cu
A
B
D
E
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Welding: Principles and Applications (MindTap Cou…
Mechanical Engineering
ISBN:
9781305494695
Author:
Larry Jeffus
Publisher:
Cengage Learning

Welding: Principles and Applications (MindTap Cou…
Mechanical Engineering
ISBN:
9781305494695
Author:
Larry Jeffus
Publisher:
Cengage Learning