Q: In the determination of iron in fortified cereals by atomic absorption, 0.9133 g of sample was…
A: A question based on absorption that is to be accomplished.
Q: Give TWO (2) reasons and ONE (1) example the transition metals act as good catalysts.
A: Elements that belong to d-block of the periodic table are called d-block elements or transition…
Q: sodium hypochlorite (NaOCl) was dissolved in a solution buffered to pH = 6.20 . find the ratio…
A:
Q: Use the data below to calculate AH for the reaction N,0, + 3C0 → N‚0 + 3CO, AH = ? - 110 - 393 + 81…
A: Note: As per our guidelines, we are supposed to answer only the first question (question number 2),…
Q: In a pharmaceutical industry a drug Paracetamol (151.163 g/mol) is determined in a sample. A 0.0315…
A: The given data in above Molar mass of paracetamol = 151.163 g /mol Mass of sample = 0.0315 g…
Q: Write the name, stereochemistry and magnetic behaviour of the following : (At. nos. Mn = 25, Co =…
A: i) K4[Mn(CN)6] : IUPAC name : Potassium Hexacyano manganate (II) Geometry : Octahedral Magnetic…
Q: A 0.973-g sample of canned tuna was analyzed by the Kjeldahl method. A volume of 21.15 mL of 0.1174…
A: 1. Moles of HCl required to titrate the liberated ammonia =Volume of HCl consumed (in L)×Molar…
Q: Given that the ratio between U235 and Pb207 analyzed from the igneous intrusion (K) is 1:3 and…
A:
Q: Give ionic and net ionic equations for the following (see picture)....
A: The actual balanced equation is given below.
Q: A 2.559 g sample containing both Fe and V was dissolved under conditions that converted the elements…
A: According to the mole concept, in terms of mass, the amount of substance in moles is equal to the…
Q: A 0.600 g sample of steel is dissolved in acid, and the manganese is oxidized to permanganate. The…
A: Given → Mass of the sample of steel = 0.600 g Volume of solution = 100 mL Wavelength = 520 nm Length…
Q: Which one of the following statements below is false and explain what is wrong:
A: Give statements,Ionization interference can occur with alkali metals at relatively low temperature…
Q: B- Calculate the ( Ag ) mg analyte to mg precipitate for Ag;PO4 (FW P= 30.97) (FW Ag=107.87), (FW O…
A:
Q: Which particles are termed as the most effective scatterers?
A: Scattering is a physics concept that refers to a wide variety of physical processes through which…
Q: A solution of sodium thiosulfate was standardized by dissolving 0.1310 g KIO3 in water, adding a…
A:
Q: D- A sample of non-pure magnetite( Fe304) weighed 0.5g convert by the chemical reaction to Fe203…
A:
Q: This problem requires values in your textbook's specific appendices, which you can access through…
A: Here we are required to find the standard cell potential of the given electrochemical reaction
Q: What is the unit of absorbance? b)When he exponential trend line of iron concentration vs time…
A: a) Absorbance has not unit. It is dimensionless.
Q: 05. Experiments show that 3.5 grams of XY, can be dissolved in a liter of saturated solution. Which…
A: Answer :- (E) 137 Atomic weight of X = 137…
Q: A 0.64 g sample containing KCl ( mw = 74.6 ) is dissolved in 50mL of water and titrated to the…
A: The %W/W of KCl in the solution has to be given,
Q: Which of the following is an inner transition element? A) Es B) Os C) Na D) F E) none of these
A: Here given option and we are asked which element is belong to inner transition element.
Q: GROUP II FLOW CHART Al", Cr". Fe". Mn", Ni", Co" colorless, purple, yellow, pale pink, green, pink…
A: The chart given for the prediction of group III ions is,
Q: Calculate how many grams of ruthenium (MW = 101.07 g/mol) were in the sample
A: Given A 2.345 g sample of a ruthenium ore was dissolved and diluted to 100.00 mL. 4.000 mL of this…
Q: A 0. 6875 g rock sample containing an unknown amount of Ca was dissolved in HCl and diluted to…
A: According to Beer-Lamberts law, the absorbance of a solution is directly proportional to its…
Q: Calculate the concentration in ppm of cadmium in an unknown which exhibits an absorbance of 0.179.
A: Using given data a calibration curve generated in Excel by plotting Absorbance versus Concentration.…
Q: Mn0 + SH + SFe Mn1 S 41,0a I he reducng agent is (A) Mn (B) H) Feani (D) Mn0a)
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 25.0 g of an impure sample containing cobalt was dissolved in acid and diluted to 1000.00 mL. 6.00…
A: The final 250.00 mL sample had absorbance equal to 0.410
Q: Which metal is used as needle for scanning tunneling microscopy? Rh O Ag Pt OW
A: Given, Which metal is used as needle for scanning tunneling microscopy?
Q: 214 82 -->
A:
Q: For the Buffers experiment, your group was tasked to prepare a 250.0 ml, 0.20 M pH 3.00 buffer from…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Zn2+(aq)+2e¯¬Zn(s) E = -0.76 Fe3+(aq)+3e¯→Fe(s) E = -0.036 Use appropriate data to calculate AG° for…
A:
Q: If a second brass washer of 0.6003 grams had been dissolved in nitric acid, diluted to 50.00 mL and…
A: Solution
Q: enow.com/ilrn/takeAssignment/takeCovalentActivity.do?locatorwassignment-take nd 10 Magnesium metal…
A: Answer : C. Magnesium burns in air. because Malleability and ponded into narrow ribbons are the…
Q: Refer to the posted calibration curve ... Note that this calibration curve can be read to the 4th…
A: The calibration curve given is, Given: Mass of sample used = 0.4567 g. Volume of sample = 50.00 mL…
Q: I know the answer is D and we'll use the M1V1=M2V2 formula.
A: Given:Number of moles of CuCl2 = 0.592 molVolume of solution = 258 mL = 0.258 L
Q: roblem Set #9 (Ch 19) Adaptive Follow-Up ercise 19.121 Item 2 I Review I Constants I Periodic Table…
A: We will equal gm equivalent of both An and NO3-
Q: d pKa values.
A:
Q: (223) 226 l227 Question 5/17 ميز هذا السؤال A 0.600 g sample containing arsenic (As) was converted…
A:
Q: Calculate the mass in grams of Al (FM = 26.98) present in an %3D unknown sample if 35.79 mL of…
A: In calmagite end point , metal ions and EDTA form complex in 1 : 1 . So we would use this concept…
Q: Use the literature value of Keq = 130 to answer this question. A solution is prepared by combining…
A: (a) Millimoles of Fe(NO3)3 =Molar concentration of Fe(NO3)3×Volume of Fe(NO3)3 =0.0500 M×4.00…
Q: A 0.2219 g sample of pure iron (MM = 55.847 g/mol) wire was dissolved in acid, reduced to the +2…
A: Given data, Mass of iron = 0.2219 g Molar mass of iron = 55.847 g/mol Volume of Cerium (IV) =…
Q: 0.650 mg of Rb,SeO4 (FM 313.89) is dissolved in 2.000 L of water. What are: (a) |M Rb2SeO4 (b) M Rb*…
A: Since you have posted a question with multiple sub-parts, we will solve first two subparts for you.…
Q: Correctly match the following related transition metals with their applications. heme myoglobin iron…
A:
Q: Potentially Useful Information Spectrochemical Series: | < Br° < SCN° < Cl° < NO3¯ < F° < OH° <…
A: The given compound is: Na4[Co(C2O4)(CN)4] The charge of the complex [Co(C2O4)(CN)4] = - 4
Q: A volume of 2.5 ml was taken from a hypothetical sample containing (3 ) valent antimony and (3 )…
A:
Q: 6. Do you see any nuclei in the specimens? What accounts for the colors of the cells? Explain.
A: Answer - According to the question - Given - S.aureus usually acts as a commensal of the human…
Q: 9. Write balanced equations for (a)238 U(a, n)'Pu, (b) * N(a, p)0, (c) Fe(a, B)Cu Write bolanged…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: The following reaction is used for the analytical determination of uranium in acidic solution: UO2+…
A:
Q: For pH values of 2.00, 6.00, and 10.00, calculate the alpha values for each species in an aqueous…
A:
Complete the following reactions
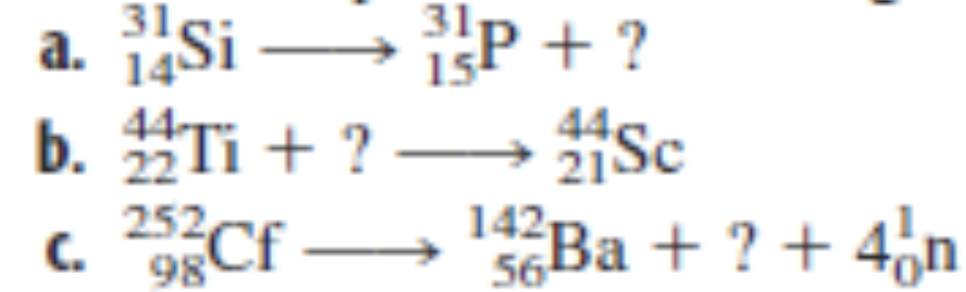
Step by step
Solved in 2 steps with 1 images

- Why not 3?Please explain how to find the highest O.N and why is for example number a) Cr +6 and not +7. Thank you!A 0.8780 g sample containing only NaCl and KCl was treated with AgNO3. The AgClformed had a mass of 1.913 g. Calculate the %Na and %K in the sample. (Na=22.9897,Cl=35.453, K=39.0983, Ag=107.868, N=14.0067, O=15.9994)
- A 0.7336-g sample of an alloy that contains copper and zinc is dissolved in 8 M HCl and diluted to 100 mL in a volumetric flask. In one analysis, the zinc (At. Mass = 65.38 g/mol) in a 25.00-mL portion of the solution is precipitated as ZnNH4PO4, and isolated as Zn2P2O7 (FM = 304.70g/mol), yielding 0.1163 g. The copper (At. Mass = 63.55 g/mol) in a separate 25.00-mL portion of the solution is treated to precipitate CuSCN (121.63 g/mol), yielding 0.2383 g. Calculate the %w/w Zn and the %w/w Cu in the sample.I wanna know how to make V = abc(1 − cos2α − cos2β − cos2γ + 2 cos α cos β cos γ)1/2 (the volume of triclinic). When a≠b≠c, α≠β≠γ ≠90°A mixture containing only BaO (153.3264) and CaO (56.0774) weighs 3.00 g. The oxides areconverted to the corresponding sulfates, which weigh 5.00 g. Calculate the % Ba and %Ca in theoriginal mixture. Ba (137.327); Ca (40.078); BaSO4 (233.896); CaSO4 (136.1406)
- 26. A 1.500 g sample of iron ore, labeled A, was processed accordingly to give 0.5215 g of pure Fe2O3 (159.69 g/mol). A second sample of iron ore, labeled B, weighed 1.750 g and gave 0.5097 g of pure Fe2O3.What is the %Fe in Ore B?What is the %Fe3O4 in Ore B?4.75 Describe the basic steps involved in gravimetricanalysis. How does this procedure help us determine theidentity of a compound or the purity of a compound ifits formula is known?Toxic Cr(VI) can be precipitated from an aqueous solution by bubbling SO2 through the solution. How much SO2 is required to treat 3.00x10^8L of 4.00x10^-2 mM Cr(VI)? 2CrO4^-2 + 3SO2 + 4H^+ —> Cr2(SO4)3 + 2H2O ___kg
- A 67.2 g sample of gold and palladium allo contains 3.40x10^23 atoms. What is the mass percentage of the gold in the alloy. And what is the mass percentage of the palladium in the alloy? Do not give handwriting solution.Help with Part A B and C: Ksp Values: Ionic CompoundFormulaKsp Aluminum hydroxideAl(OH)31.8×10–5 Aluminum phosphateAlPO46.3×10–19 Barium carbonateBaCO35.1×10–9 Barium chromateBaCrO41.2×10–10 Barium fluorideBaF21.0×10–6 Barium hydroxideBa(OH)25×10–3 Barium sulfateBaSO41.1×10–10 Barium sulfiteBaSO38×10–7 Barium thiosulfateBaS2O31.6×10–6 Bismuthyl chlorideBiOCl1.8×10–31 Bismuthyl hydroxideBiOOH4×10–10 Cadmium carbonateCdCO35.2×10–12 Cadmium hydroxideCd(OH)22.5×10–14 Cadmium oxalateCdC2O41.5×10–8 Cadmium sulfide*CdS8×10–28 Calcium carbonateCaCO32.8×10–9 Calcium chromateCaCrO47.1×10–4 Calcium fluorideCaF25.3×10–9 Calcium hydrogen phosphateCaHPO41×10–7 Calcium hydroxideCa(OH)25.5×10–6 Calcium oxalateCaC2O42.7×10–9 Calcium phosphateCa3(PO4)22.0×10–29 Calcium sulfateCaSO49.1×10–6 Calcium sulfiteCaSO36.8×10–8 Chromium(II) hydroxideCr(OH)22×10–16 Chromium(III) hydroxideCr(OH)36.3×10–31 Cobalt(II) carbonateCoCO31.4×10–13 Cobalt(II) hydroxideCo(OH)21.6×10–15 Cobalt(III) hydroxideCo(OH)31.6×10–44…What would happen to CaBr2 + Ni(NO3)3 --> NiBr3 + Ca(NO3)2 , if another CaBr2 was added?

