4. Comment on the following: a. Whereas D-glucose is virtually unaffected by heating in dilute aqueous acid (apart from undergoing mutarotation), D-idose form the anhydrosugar X. Explain the different behaviour of the sugars. он он он HO, OH но, „OH HO.. OH H+ HO CH2OH "CH,OH gluco se idose
4. Comment on the following: a. Whereas D-glucose is virtually unaffected by heating in dilute aqueous acid (apart from undergoing mutarotation), D-idose form the anhydrosugar X. Explain the different behaviour of the sugars. он он он HO, OH но, „OH HO.. OH H+ HO CH2OH "CH,OH gluco se idose
Cardiopulmonary Anatomy & Physiology
7th Edition
ISBN:9781337794909
Author:Des Jardins, Terry.
Publisher:Des Jardins, Terry.
Chapter7: Acid–base Balance And Regulation
Section: Chapter Questions
Problem 11RQ
Related questions
Question
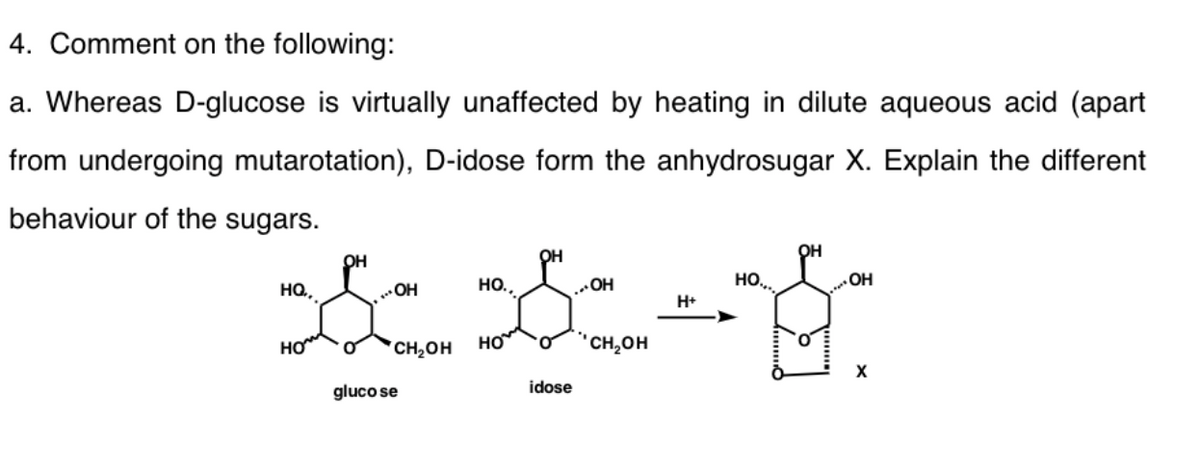
Transcribed Image Text:4. Comment on the following:
a. Whereas D-glucose is virtually unaffected by heating in dilute aqueous acid (apart
from undergoing mutarotation), D-idose form the anhydrosugar X. Explain the different
behaviour of the sugars.
он
OH
HQ,
OH
но.,
OH
HO..
H+
HO
HO
*CH¿OH
но
"CH,OH
gluco se
idose
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Cardiopulmonary Anatomy & Physiology
Biology
ISBN:
9781337794909
Author:
Des Jardins, Terry.
Publisher:
Cengage Learning,


Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Cardiopulmonary Anatomy & Physiology
Biology
ISBN:
9781337794909
Author:
Des Jardins, Terry.
Publisher:
Cengage Learning,


Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax