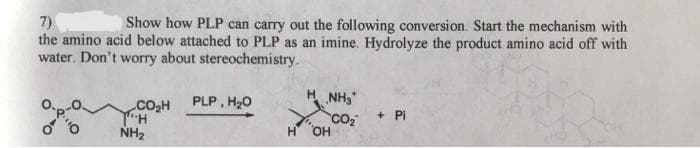
7) the amino acid below attached to PLP as an imine. Hydrolyze the product amino acid off with water. Don't worry about stereochemistry. Show how PLP can carry out the following conversion. Start the mechanism with PLP, H20 H NH3 CO2H + PI NH2 H OH
Q: hich of the two will first elute during a process? and Why? 1,3,5 trimethyl benzene vs benzene,…
A: Chromatography is a technique used for the separation of compounds or molecules present in a sample.…
Q: It is often stated that the phosphate-phosphate bonds in ATP are "high energy," but in fact, they…
A: ATP is an energy currency of cells that contain nucleoside, which consists of nitrogenous base…
Q: Use arrow-pushing to show how the glycerol phosphate reacts with the base :8 and NAD* to form DHAP.…
A: Cellular respiration is a catabolic pathway of the process of metabolism, where a series of chemical…
Q: The reaction below is an exampie ol an) reaction. H-C-OH H--OH H-C-OPo, H-C-Oro Hydrolysis…
A: Isomers have different physical and chemical properties, although they have a same molecular…
Q: Which of the following reactions are considered isomerization reaction? (the letters designate…
A: Isomerization reaction is a chemical during which a compound is transformed into its isomeric forms.…
Q: Which of the following is the result of the hydrolysis of 5'-CTAGTTC-3' at the b side? (А) рСр, 3…
A: Hydrolysis of DNA is the addition of water molecules to break the pyrophosphate bond between the…
Q: an inorganic ion. Such as metal ion, that improves the fit of an enzyme with its substrate is a(n)?
A: Enzymes are the molecules with speed up the rate of a biological reaction without being consumed in…
Q: In a solution with pH ~7 most of the amino acids are form zwitterions. Name the amino acids which…
A: Amino acids: a. Amino acids are the building block of proteins that contain a carboxylic acid group…
Q: Why do we refer to the conversion of six molecules of carbon dioxide (six carbon atoms) to one…
A: The Calvin cycle is the sequence of reactions involving the reaction of carbon dioxide to form…
Q: In titration of Amino Acids: Show the reaction involved in the titration showing all three species…
A: Amino acids have the ionizable groups. The predominant ionic form of the amino acids is determined…
Q: In chymotrypsin the active site histidine Forms a tetrahedral intermediate O a. O b. Acts as a…
A: Serine protease is part of the catalytic triad and consists of the serine, aspartic acid, and…
Q: HOCH, OH OH OH OH
A: Biopolymers or large biomolecules, such as nucleic acids are needed for all known types of life.…
Q: Which of the following is the result of the hydrolysis of 5'-CTAGTTC-3' at the a side?…
A: Nucleic acids are composed of nucleotides. Nucleotides are composed of a phosphate group, a pentose…
Q: Which of the following reactions takes place by dehydration synthesis? Which of them takes place by…
A: Some polymerization reactions are accompanied with the loss of water molecules at each step. Such…
Q: ОН HOH,C_| Н C | н н-с- Но HN / III -Н соо- ОН ОН HC-C Н N-Acetylneuraminic acid (Neu5Ac) (a sialic…
A: Different Isomeric forms of simple sugars (called monosaccharides like glucose and galactose) are…
Q: Riboflavin gives rise to cofactors: Select one: O a. All of them O b. FAD; FADH2 O c. NAD+; NADH d.…
A: Introduction: Riboflavin is likewise known as Vitamin B2. It is an important food supplement.…
Q: The blood brain barrier protects the brain from harmful molecules and also prohibits certain drugs…
A: Hydrophilic materials are those that have a unique solubility in water and expand across it,…
Q: Which factor is least likely to result in protein denaturation? disruption of weak interactions by…
A: Protein denaturation is the process by which a protein loses its tertiary structure and therefore…
Q: Which of the following is true regarding the glycosidic bond between the pentose sugar and…
A: DNA and RNA are nucleic acids composed of nucleotide units. Nucleotides are composed of pentose…
Q: Question attached
A: pka of NADH > pka of the phenetic -OH therefore NADH will deprotmate the phenolic -OH to produce…
Q: working, oxygen gas bubbles will be released. The chemical reaction that occurs is the following:…
A: pH is a very important criteria for determining the activity of an enzyme because an enzyme only…
Q: Which of the following statements about folic acid is not true? a. Folic acid is activated by folate…
A: Folic acid is water-soluble vitamin B12 which was an essential nutrient necessary for protein and…
Q: Several of these reactions are energetically unfavorable and will only occur if coupled to a second,…
A: Energetically reactions have two types of conditions: Energetically Favorable Condition: The…
Q: In the physiological pH range, the nitrogen in the imidazole ring can be easily…
A: The nonessential amino acid Histidine has physiological pH of 6 due to the presence of the imidazole…
Q: In the elimination-addition mechanism for phosphate group transfer reactions, the intermediate would…
A: Phosphate ester and anhydride phosphorus atom reactions form the chemical basis for many of the most…
Q: I’m not sure which answer is true for all that answer this question can you please help?
A: ATP is called the energy currency of the cell. ATP's full form is adenosine 5' triphosphates. ATP…
Q: What is the role of water in the following biological reaction/system? 00 R. R. Glu HN R' Glu HN R'…
A: Carboxypeptidase is a zinc containing proteolytic enzyme. Activation of bound water…
Q: The structure below shows a tripeptide at physiological pH with the protonatable groups labeled with…
A: There are twenty amino acids are present, which join with each other via a covalent bond to form a…
Q: Two macromolecules, such as proteins, can adhere tightly and specifically to each other. How can…
A: Proteins when binds to a molecule it does so with great specificity and is tightly bound. There are…
Q: Your protein has a pI of 8.34 and is in a pH 7 buffer. Which of the chromatographs below would you…
A: *Cation exchange chromatography is an ion exchange chromatography used to separate molecules based…
Q: Determine the type of biochemical reaction that occurs in the reaction shown below. O redox…
A: In the given question, a cyclic pyranose structure is given. It is a pyranose because it has 6…
Q: Urease can catalyze the hydrolysis of Urea, H,N – C - NH, but not H H the hydrolysis of diethyl urea…
A: A chemical reaction is a process that converts one or more substances into another substance.…
Q: Consider the following molecule. a. Name it.b. Use the three-letter symbols for the amino acids…
A: Amino acids are organic compounds that contain the amino (–NH2) and carboxylic acid (–COOH)…
Q: H20 f) OH Chemistry Steps Chemistry Stes Br Chemistry Steps Chemistry Steps Chemistry Steps Br h)…
A: Solution is attached below.
Q: A (-) charge in the transition state can be stabilized by a catalyst, which is usually a(n): a.…
A: A negative charge in the transition state can be stabilized by a BASE. Incorrect options- a. anion…
Q: Which of the following statement is FALSE ? pKa values of active site residues can be shifted from…
A: Enzymes are the proteins that catalyse the reactions by lowering the activation energy of the…
Q: The iodine number of a fatty acid indicates: Group of answer choices how much iodine to…
A: Fatty acids are carboxylic acids with a long chain of hydrocarbons.
Q: Protein synthesis is which type of reaction? Group of answer choices A. ester synthesis is anabolism…
A: Introduction: Those molecules that are helpful in the formation and function of the living body are…
Q: Given the balanced equation with an unknown compound represented by X, which compound is represented…
A: Sugars such as glucose, sucrose, fructose, etc., can be converted to alcohol (ethanol) by a…
Q: How do the ligand (substrate) and the protein/enzyme predominantly interact? O sigma bonds O…
A: Note: We are authorized to answer one question at a time since you have not mentioned which question…
Q: During the formation of ester bond, which of the following takes place? O a. Hydrogen atom is gained…
A: The formation of an ester bond between glycerol molecule and fatty acid converts them to a lipid…
Q: 6. Decrease in temperature, reactant concentration, pH, or co-enzymes can slow reaction rate 6. 7.…
A: Enzymes are biocatalysts that increase the rate of reaction. Co-enzyme is a substance that increases…
Q: Which of the following has the sugar found in RNA? A. D. Но- OH HN. ÓH но ОН ОН В. E. NH2 H. OH…
A: RNA have the following nitrogen bases:- nitrogen bases in the RNA are - 1) Uracil , 2) Adenine , 3)…
Q: Which of the following alters proteins and membranes? Alcohols O lodine O All other choices O…
A: Many critical roles in the body are performed by certain complex molecules called proteins. For the…
Q: What form of the tripeptide is most abundant at pH 4.00? O ILY CO ILY2+ O ILY+ O ILY O ILY-
A: Tripeptide is an oligopeptide that contains three amino acids. It has two peptide linkages in it. A…
Q: Which of the following represents a synthesis reaction? Multiple Choice AB +XY + AY+ XB AB A+B AB +…
A: In biological system the enzymes are responsible for doing the chemical reactions in a cells. The…
Q: P P₂ 2 E*-P₁-P₂ E*-P₂ E* E E-S₁ E-S₁-S₂ Which of these 2 options represents a Sequential Ordered…
A: Enzymes are very important ad highly effective catalysts, having the ability to enhance the reaction…
Q: In a lab experiment involving temperature and a glycolytic enzyme isolated from E. coli, you notice…
A: Enzymes are the biocatalyst that increase the rate of reaction by decreasing the activation energy.…
PLP or Pyridoxal phosphate is an enzyme which has the ability to act as an electrophilic catalyst that stabilizes different types of carbanionic reaction intermediates. It catalyzes myriad reactions and forms internal lysine derived aldimine.
Step by step
Solved in 2 steps with 1 images

- In titration of Amino Acids: Show the reaction involved in the titration showing all three species at low, neutral, and high pH.Use the given experimental data to deduce the sequence of anoctapeptide that contains the following amino acids: Ala, Gly (2 equiv),His (2 equiv), Ile, Leu, and Phe. Edman degradation cleaves Gly from theoctapeptide, and carboxypeptidase forms Leu and a heptapeptide. Partialhydrolysis forms the following fragments: Ile–His–Leu, Gly, Gly–Ala–Phe–His, and Phe–His–Ile.Based on our pka's table, pKa1 of -COOH group of Cys = 1.71 pKa2 of -NH2 group of Cys = 10.78 pKa3 of side chain group of Cys = 8.33 If pKa is different, will the buffer change??
- Which of the following is the result of the hydrolysis of 5'-CTAGTTC-3' at the a side? 2 pC, 3 pT, pA, pG 2pCp, 3 pT, pA, pG, pC-OH pCp, 3 Tp, Ap, Gp, C-OH pCp, 3 pT, pA, pG, pC-OHDraw the tripeptide gly-glu-phe with all ionizable groups in their predominant state at pH 7.0.The pl of alkaline phosphatase is 4.5; the pl of the DEAE cellulose is 10.5. We used a buffer of pH 7.4 to run a DEAE-cellulose as anion exchange column? Explain how did this work?
- Write the reversible reactions with its corresponding enzymes of Dehydration via Hydrolysis reaction. A.) A tripeptide between lysine, alanine and argininelooking at ABG's, if ph is normal, Bicarb is alkaline, and paCO2 is acidic...what do you say about it? its not clearly in any of the 4 states, partially compensated, etc pH 7.37 HCO3 45 PaCO2 66.Draw the structure of the products formed when Arabinose shown above undergoes the following reaction: Tollen's reagent (include side product) Benedict's reagent (include side product) Oxidizing enzyme to produce alduronic acid Reducing agent like NaBH4 with Ethylamine (CH3CH2NH2) do everything for upvote. no upvote if incomplete
- The Keq for the reaction of water with water is called Kw H2O + H2O ⇔ H3O+1 + OH-1 [H3O+1][OH-1] Kw = ––––––––––––– = [H3O+1][OH-1] = (1 x 10-7)2 = 1 x 10-14 [H2O]2The investigators reported that the sphingosine kinase enzyme attached to the DEAE column when Tris was used as a buffer, but not when a phosphate buffer at the same pH was used. Explain the difference in behavior. The structure of the conjugate base of the Tris buffer is shown below. Hint: consider what charges all these molecules have at pH 7.4Oils can be converted to fats by a commercial process catalyzed bynickel which is called partial hydrogenation. In relatively mild conditions (180ºC and pressures of about 1.33 atm) a sufficient number of double bonds are hydrogenated and solidify liquid oils. This solid material, margarine, has a consistency similar to butter. Come up with a practical reason why oils don'tthey are completely hydrogenated during commercial hydrogenation processes.

