Q: tarting with any alcohols (SIC) and any inorganic reagents required make the target molecule. Show…
A: Concept introduction: Retrosynthesis analysis is an important tool for Designing any organic…
Q: 4. What starting materials would you use to prepare the following via a Diels-Alder reaction?
A: 2 equivalent 1,3-butadiene and 1 equivalent 1,4-benzoquinone give the described product in…
Q: 5. 6. 7. Write the Lewis Structure for SC1₂ (S is the center atom) Write the Lewis Structure for…
A:
Q: MISSED THIS? Read Section 18.2 (Pages 788 - 799); Watch KCV 18.2B, IWE 18.2. Use the…
A:
Q: The equilibrium constant, K, for the following reaction is 3.78E-2 at 530 K. PCI5(9) PC13(g) +…
A: Given: Initial volume of the container is 9.30L. New volume of the container is 16.6L. Equilibrium…
Q: HEMWORK When a calcium chloride solution is mixed with a lead(II) nitrate solution, a precipitate…
A: Steps to write a net ionic equation and complete ionic equation for a molecular reaction :…
Q: Which of the following transport systems allows small molecules such as urea and O₂ to cross the…
A: By the passive diffusion process small molecule cross the cell membrane without expanding energy .…
Q: 1. Keeping in mind the definition of a buffer, prec Start with 50 mL of buffer at pH 9.00. Add b.…
A: Solutions- Buffer- It is defines as the buffer solution is a solution able to maintain its Hydrogen…
Q: t takes 614. kJ/mol to break a carbon-carbon double bond. Calculate the maximum wavelength of light…
A: Answer: Relation between wavelength of light and energy of photon is shown below: E=hCλ Here:…
Q: Part F 4- V₂07¹ Express your answer as signed integers separated by a comma. View Available Hint(s)…
A: In the given compound, oxygen is in -2 oxidation state and vanadium is in its highest oxidation…
Q: Calculate the standard enthalpy change for the reaction at 25 ∘ C. Standard enthalpy of formation…
A: Calculate standard enthalpy change for the reaction at 25°C.
Q: Which of the following substances is/are never in a Bronsted-Lowry acid in an aqueous solution? KOH…
A: A Bronsted-Lowry acid is a substance which donates a proton or H+ ion to the other compound
Q: In the reaction CH3 COOH+H₂O=H3O++CH3COO- the base constant K₁ of the acetate ion CH3COO is…
A:
Q: What is the pH of a 0.750 M solution of NaCN (Ka of HCN is 4.9 × 10-10)?
A: pH is the negative logarithm of hydrogen ion concentration. It can be given as pH=-log [H+] NaCN…
Q: Consider the malate dehydrogenase reaction from the citric acid cycle. Given the listed…
A:
Q: Calculate the volume in milliliters of a 2.59M nickel(II) chloride solution that contains 175. g of…
A:
Q: Determine the molar solubility with the generic formula AB2 if ksp=2.56*10^-2
A:
Q: For a gas obeying Dieterici's equation of state for 1 mole is p(V - b) = RTe-a/RTV (i) (ii) Prove…
A: We have find out the answer.
Q: b) C₂H₂O FILTRE 11 10 HSP-04-272 9 8 00 7 6 5 ppm w 1380 bs, IH 9.2H 3 3,6H 1 1, 3H O. 0
A: DBE is double bond equivalent. It is very helpful to get rough idea about how many multiple bonds…
Q: B Current Attempt in Progress X Incorrect Draw the major product(s) for the following reaction. If a…
A: We have been given an organic reaction and we have been asked to draw the major product of given…
Q: Draw one of the two enantiomers of the major product from this reaction. Use a dash or wedge bond to…
A: given
Q: The molar concentration of H3O+ in pure water at 25 °C is: O(A) 0.0 M (B) 10-14 M O(c) 10-7M O (D)…
A:
Q: 14. What is the molar mass of Ca? 16. 078 15. What is the molar mass of H₂SO4? What is the mass of 3…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
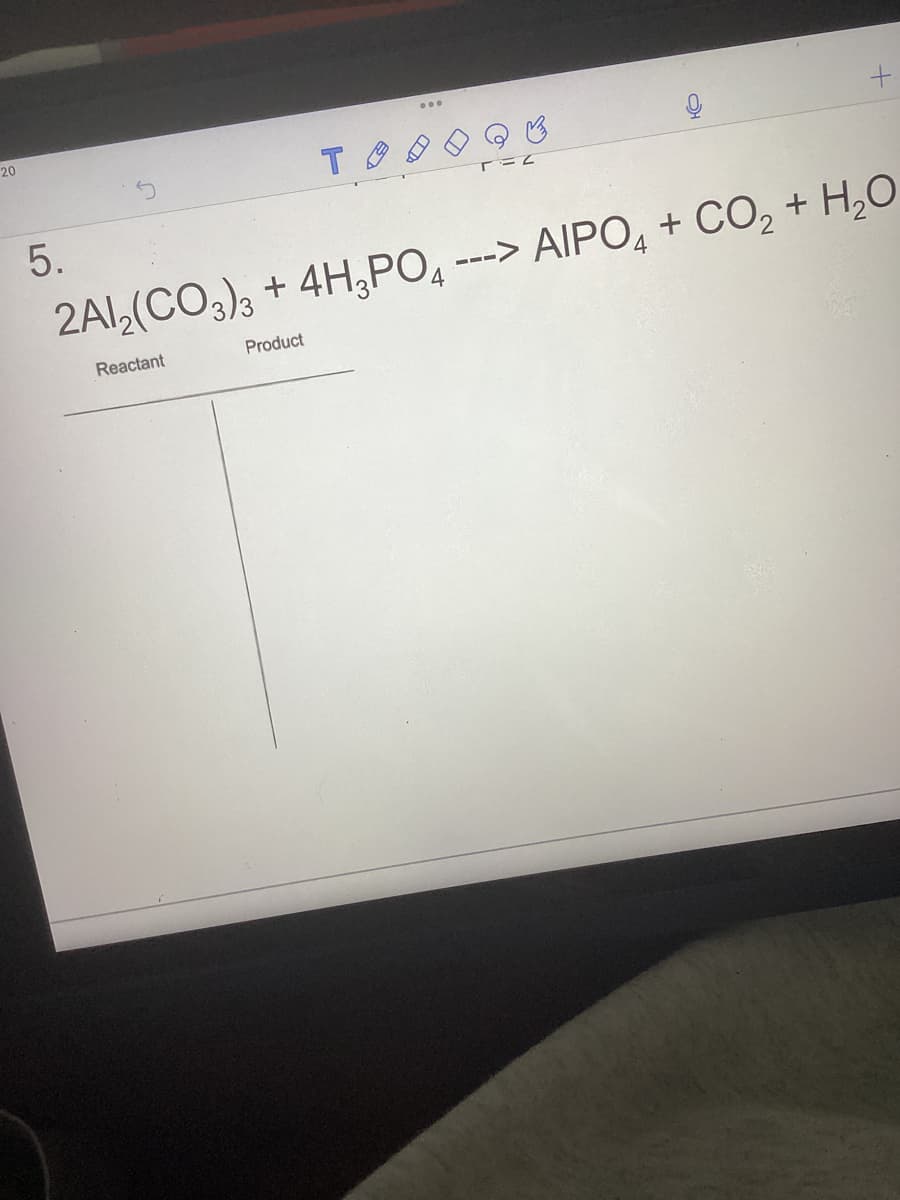
Q: C5H12 + 60₂- 602 ---> 6CO₂ + H₂O Reactant Product
A: Since Balanced reaction means that both side number of atom present in equal number. Thus,
Q: Macmillan L Cl name: Incorrect MAR OH CI CI
A: The question is based on the concept of IUPAC naming. we need to identify the IUPAC name of the…
Q: Write the reaction in water for H2CO3? Be careful to use the proper arrow. If none, write N.R.
A: An acid is a substance which when dissolve in water give H+ ions. For weak acid there exist an…
Q: MS Relative Intensity 100 80 60 40 20 0 10 20 30 40 50 60 70 m/z 80 90 100 M+ 110 120 m.w. 106 2-p…
A: The question is based on the concept of mass spectroscopy . we need to analyse the spectra &…
Q: A block of wood has the measurements 78 cm X 15.1 cm X 243.1 cm has a mass of 1.716 x 10° grams.…
A:
Q: Identify the product for this reaction. excess H₂ /Rd.C 1 atm/25°C
A:
Q: 2. A student performed the experiment as described, using 5.00 mL of an aqueous 3.00% H2O2 solution,…
A: The basic chemical concept like mole concept, mass percentage, ideal gas law etc. are necessary to…
Q: or the reaction: AgI(s) + 1/2Br2(g) → AgBr(s) + 1/2I2(s), ΔH° = –54.0 kJ…
A: Answer: Enthalpy of reaction is always equal to the difference between enthalpy of formation of…
Q: CH₂CH3 H3C-C-CH3 :Br: NA H H3C For the reaction above, draw curved arrows to show the movement of…
A: Draw the curved arrows that depict the mechanisms by which the reactions occur is one of the most…
Q: Choose the major product obtained from Diels-Alder reaction of cycloaddition of 1,3-cyclopentadiene…
A: When a conjugated diene reacts with dienophile it forms six member ring as a product
Q: It takes 151. kJ/mol to break an iodine-iodine single bond. Calculate the maximum wavelength of…
A:
Q: The potential of electrodes Cu2+ (aq) | Cu(s) is ϕ1 = 0.337 V , and Cu2+ (aq), Cu+ (aq) | Pt is…
A: We need to calculate a) the value of Ecell for electrode Cu+(aq) | Cu(s). b) the equilibrium…
Q: Based upon the limiting reagent, calculate the theoretical yield of magnesium oxide, MgO, for the…
A: Given: Ignition of magnesium metal in air for this experiment To determine: Based upon the limiting…
Q: For the reaction that represents the formation of water: 2 H2 (g) + O2 (g) → 2H₂O based on the moles…
A: For the reaction that represents the formation of water: 2H2(g) + O2(g) → 2H2O (l) , we have to…
Q: at undergoes: H₂C HC H₂C- Refer to the fat structure below. Draw the possible products if the 6.…
A: Solution: We know fatty acid on saponification we get soap (alkali salt of fatty acid) and glycerine…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: The unbalanced redox reaction given is ClO2 + Cr3+ → ClO3− + Cr2+
Q: Which compound below is predicted to be the most acidic? OH OH да вовн OH 0 OH
A: carboxylic acid stronger than phenol Due to the resonance in carboxylic acids, the negative charge…
Q: a. On the basis of the general solubility rules, choose the substance that is not likely to be…
A: Here, we have to find the compounds that are not likely to be soluble in water according to the…
Q: What is the K₂ reaction of HCN? Ka reaction: The K₂ of HCN is 6.2 x 10-10. What is the K, value for…
A: It is based on the concept of acid and base Here we are required to find the expression of acid…
Q: Question 8 of 20 Write the formula of the conjugate acid of HCO₂™.
A: an acid is a species which donates Proton while a base accepts proton. an acid after donating…
Q: 10. What is the major product in the following reaction? of A. I 1. NaOEt, EtOH, 25 °C do ď B. II 2.…
A:
Q: Carbon monoxide and water vapor are formed when carbon dioxide and hydrogen react. CO2(g) + H2(g) ⇌…
A:
Q: Please do not round off intermediate calculations. Thank you.
A:
Q: Consider the reaction: 2 CO(g) + O₂(g)=2 CO₂(g). ↑ The reaction is allowed to reach equilibrium in a…
A: As per Le Chatelier's principle, when there is any change in external factor in equilibrium…
Q: What is the pka range of the alpha proton of the carbonyl compound?
A: The alpha proton of a carbonyl compound indicates the hydrogen atom attached to the carbon atom…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- What mass of 47CaCO3 must be orderedA 0.9056 g sample of KBrO3 (MM=167.0) was dissolved in dilute HCl and treated with an unmeasured excess of KI. The liberated iodine required 37.21 mL of Na2S2O3. Calculate the molar concentration of sodium thiosulfate. 1 mol BrO3- = 3 mol I2 = 6 mol S2O32- 0.8744 M 2.623 M 0.8791 M 0.2915 M31. A sample of meat scrap weighing 2.000 g is digested with concentrated H2SO4 and a catalyat. The resulting solution is made alkaline with NaOH and the liberated ammonia distilled into a 50.0 mL of 0.6700 N H2SO4. The excess then requires 30.10 mL of 0.06520 N NaOH for neutralization. What is the percentage of nitrogen in the meat?
- A batch of raw milk was tested and found to contain a bacterial population of 8xl06cfu/ml. It is to be pasteurised at noc for 33 seconds. The average D value at 52°C for the mixed population is 11 min. The z value is 10°C.i)Explain the terms "D value" and "z value".ii)Calculate how many organisms will be left after pasteurisation.iii)Hpw much time will be needed at 62°C to accomplish the same degree oflethality?iv. How much time will be needed at 82°C to achieve the same degree oflethality?Why does Cv≠ Cp?600g of molybdenum (C-0.25) is heated from 250K to 650K. What is delta H?
- 23a, no need to explain too muchThe data below give the concentration of thiol (mM) in the blood lysate of the blood of two groupsof volunteers, the first group being ‘normal’ and the second having rheumatoid arthritis (Banford, J.C.,Brown, D.H., McConnell, A.A., McNeil, C.J., Smith, W.E., Hazelton, R.A. and Sturrock, R.D., 1983,Analyst, 107: 195): Normal: 1.84, 1.92, 1.94, 1.92, 1.85, 1.91, 2.07Rheumatoid: 2.81, 4.06, 3.62, 3.27, 3.27, 3.76 Verify that 2.07 is not an outlier for the ‘normal’ group.Fluoride treatment strengthens tooth enamel by converting hydroxyapatite, Ca5(PO4)OH, into fluorapatite, Ca5(PO4)3F. Fluoraparite is stronger and more resistant to acid than hydroxyapatite. If the solubility of fluorapatite is 6.1 x 10^-8 mol/L, what is its Ksp? Ca5(PO4)3F(s) -> 5Ca^2+ + 3PO4^3- + F-

