5.) Below is the valence electron molecular orbital diagram and the corresponding potential energy diagram for the Cl2 diatomic molecule. 'n 5- Зр, Зр, Зр. 3p, 3p, 3P. CIP) + CI 2- CIP) + CI 3s 3s 10 12 Bond length / Å a) Referring to the potential energy diagram on the left, indicate whether the following transitions are allowed or not allowed? Explain your choice in each case. 'LE'E b) The MO diagram on the right depicts the ground state electron configuration for the Cl2. Use the information from this configuration to show that the ground state term symbol is '2: c.) Redraw the MO diagram with electron occupancy to show the excited state electron configurations represented by the y and I „ term symbols that label the states in the potential energy level diagram. Energy Energy/eV
5.) Below is the valence electron molecular orbital diagram and the corresponding potential energy diagram for the Cl2 diatomic molecule. 'n 5- Зр, Зр, Зр. 3p, 3p, 3P. CIP) + CI 2- CIP) + CI 3s 3s 10 12 Bond length / Å a) Referring to the potential energy diagram on the left, indicate whether the following transitions are allowed or not allowed? Explain your choice in each case. 'LE'E b) The MO diagram on the right depicts the ground state electron configuration for the Cl2. Use the information from this configuration to show that the ground state term symbol is '2: c.) Redraw the MO diagram with electron occupancy to show the excited state electron configurations represented by the y and I „ term symbols that label the states in the potential energy level diagram. Energy Energy/eV
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter6: Quantum Mechanics And Molecular Structure
Section: Chapter Questions
Problem 26P: Following the pattern of Figure 6.21, work out the correlation diagram for the BeN molecule, showing...
Related questions
Question
Answer second question
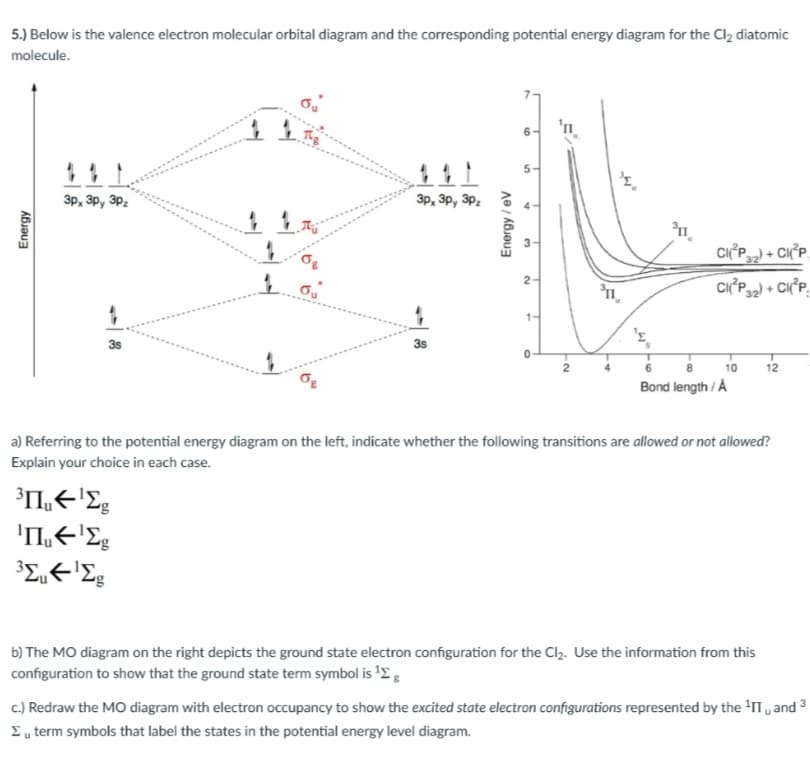
Transcribed Image Text:5.) Below is the valence electron molecular orbital diagram and the corresponding potential energy diagram for the Cl2 diatomic
molecule.
'n
5-
3p, 3p, 3Pz
3p, 3p, 3Pz
n.
CIP) + CIP.
2-
CI°P + CI°P,
1-
3s
3s
12
Bond length / Å
a) Referring to the potential energy diagram on the left, indicate whether the following transitions are allowed or not allowed?
Explain your choice in each case.
b) The MO diagram on the right depicts the ground state electron configuration for the Clz. Use the information from this
configuration to show that the ground state term symbol is '2 :
c.) Redraw the MO diagram with electron occupancy to show the excited state electron configurations represented by the 'II , and
Eu term symbols that label the states in the potential energy level diagram.
Energy
Energy / ev
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,