Q: If the same mass of Na,sO4 (MW: 142 g/mol) and K2SO4 (MW: 174 g/mol) were weighed and used for the…
A: Given: Mass of Na2SO4 and K2SO4 taken is the same (assuming 100 g).
Q: 4:[a] A mixture containing only KCI and NaBr is analyzed by the Mohr method. A 0.3172-g sample is…
A:
Q: A compound, KBrOx, where x is unknown, is analyzed andfound to contain 52.92% Br. What is the value…
A:
Q: Consider thé following statement: Statement I: Matrix is equal to blank plus the analyte. Statement…
A: Matrix: Matrix refers to the components of a sample other than the analyte of interest. Analyte: a…
Q: Lactic acid is what causes the muscles the feeling of soreness or fatigue. In vitro study was done…
A:
Q: A hydrochloric acid solution is standardized by titrating 0.4541 g of primary standard…
A: Given:The mass of primary standard tris(hydroxymethyl)aminomethane = 0.4541 g. Volume = 35.37 mL =…
Q: Choi is fond of carbonated beverages so he crafted his own soda formulation called "Sodalicious."…
A: Since you have asked a question with multiple subparts, we will answer only first 3 subparts for…
Q: 4. What is the pereentage purity of a sample of Al, (SO4)3 if a 0.5000-g sample produced a…
A: Given : Mass of Al2(SO4)3 sample = 0.5000 g And mass of BaSO4 precipitated = 1.000 g
Q: What volume of 0.305 M AgNO3 is required to react exactly with 155.0 mL of 0.274 M Na2SO4 solution?…
A: The balanced chemical reaction between the silver nitrate and sodium sulphate is shown below.
Q: Assuming that the uncertainty in the readings does not change, why is it not acceptable to base…
A: Reducing uncertainty in titration is important to determine the desired concentration or the…
Q: How many grams of precipitate will form when Na2SO4 reacts with 25 mL of 0.50 M Ba(NO3)2?
A: The balanced reaction would be: Na2SO4 (aq) + Ba(NO3)2 → BaSO4 (s) + 2 NaNO3(aq)
Q: A solution is made by mixing 500.0 ml. of 0.04794 M Na, HASO, with 500.0 mL of 0.02554 M NaOH.…
A: The given data contains, Volume of Na2HAsO4 =500 mlmolarity of Na2HAsO4=0.04794 MVolume of NaOH…
Q: ) 0.100 g of Na½CO3 is added to a 200.0 mL solution of 1.00 x 103 M Ca(NO3)2. Calculate Q. -x 106
A: First of all we have to calculate concentration of the species
Q: c. You have titrated the first sample of your unknown sample. Using the average molarity of the…
A: From part-(b), the average molarity of potassium permanganate, KMnO4(aq) is 8.108*10-3 M #Trial-1…
Q: If 12.50 mL of a KOH(aq) solution is required to titrate 0.218 g of KHP, what is the molar…
A: weight in g = 1 molmolecular weight in g×weight in g = Number of molsVolume in…
Q: A hydrochloric acid solution is standardized by titrating 0.5008 g of primary standard tris…
A: Given that : The mass of primary standard tris (hydroxymethyl) aminomethane, C4H11NO3 = 0.5008 g…
Q: mass of KIO3 = 0.9290g titrated volume Na2S2O3 = 28.88mL standardisation sodium thiosulfate
A:
Q: What is the exact concentration of the NaOH titrant in molarity? Express your answer in 3…
A: Since you have asked a question with multiple subparts, we will answer only 3 subparts for you. In…
Q: calculate the mass of primary standard sodium chloride (MM=58.44) necessary to react with 25 ml of…
A: Molarity is a term used for concentration which shows the moles of solute dissolved per liters of…
Q: During the standardization of barium hydroxide, a 0.3259-g of primary-standard-grade benzoic acid…
A:
Q: 0.6025-g sample was dissolved, and the Ca 2+ and Ba 2+ ions present were precipitated as BaC2O 4 .…
A: Note the dehydration equation of calcium oxalate and barium oxalate.…
Q: The 500.0-mg sample of impure Na, CO3 (FM: 105.96) required 22.00 mL of the HCI standard solution…
A: In order to solve this question, we will have to write down the reaction of sodium carbonate and…
Q: 2.5 ml of an unknown dye having a molarity of 3.6 x 10° M is diluted to a final volume of 20.0 ml.…
A: This is a dilution problem of mole concept. On dilution , the concentration of a substance…
Q: An impure sample of barium hydroxide of mass 1.6524g was allowed to react with 100mL of 0.200M…
A: Molar mass of Ba(OH)2= 171.34 g/mol Mass of impure sample of Ba(OH)2=1.6524g Volume of HCl=100ml…
Q: A 0.4435-g sample of impure Na2CO3 (FW 105.99) was analyzed by the Volhard method. After adding…
A:
Q: A sample of freshly precipitated and filtered silver chloride weighs 459.0 mg. If 1.00 % of the…
A: The chemical equation representing the decomposition reaction of silver chloride is shown below.
Q: 2每張試題卷得必填寫(學號)、(姓名) The ethyl acetate concentration in an alcoholic solution was determined by…
A: Question 11 is not related with the given problem.
Q: 05 ± 2.0e-4 (Analytical Balance) Total volume of Sample Stock, mL: 75 ± 0
A: For phenolphthalein Endpoint Using the molarity equation; M1V1 (for Hcl)=M2 V2 (Noda Ash)(5.05×10-2…
Q: A sample containing BTEX What is the appropriate quantitative and qualitative analysis method for it…
A: BTEX i.e Benzene, toulene, ethyl benzene and xylene are volatile organic compounds and are very…
Q: You collected the following data from a titration experiment using a 0.129M standardized NaOH…
A: Please have a look on the solution:
Q: A 0.1530 g of pure KIO3 was dissolved in 50 mL distilled water. This was then added with KI and 6N…
A: Normality of sodium thiosulfate solution can be calculated from the Molarity. So first we calculate…
Q: A permanganate solution is prepared by dissolving 20.0123 g KMNO4 in 500 mL of distilled water and…
A:
Q: 100.0 mL of 0.60 M aqueous Pb(NO3)2 is mixed with excess aqueous KI. What mass of precipitate is…
A:
Q: Please calculate the volume in mL of a solution containing 35.5 grams of sodium sulfate with a…
A:
Q: In the titration of a volume of 50 mL of a hydrochloric acid solution with Solution density 1.045 g…
A: Density is measure of mass per unit volume. Density =MassVolume Molarity is the ratio of moles of…
Q: The tartaric acid, H2C4H4O6 (150.087 g/mol ) in 100.0 mL wine sample was determined using sodium…
A: The question is based on the concept of titrations. we are titrating a weak acid with a strong…
Q: Choi is fond of carbonated beverages so he crafted his own soda formulation called “Sodalicious.”…
A: We have two different variants of soda formulations which are: 1. Sodalicious by Choi. 2. Soda Pop…
Q: If the same mass of Na2SO4 (MW: 142 g/mol) and K2SO4 (MW: 174 g/mol) were weighed and used for the…
A: The balanced equation for the formation of the precipitate, BaSO4(s) is: Ba2+(aq) + SO42-(aq)…
Q: A 35.0 mt sample of 0.225 M HBr was dituted to 42.3 ml, what is the Corrc. of diluted Her ?
A: Concentration of diluted HBr can be calculated using dilution formula.
Q: 4. A 0.5843-g sample of a plant food preparation was analyzed for its N content by the Kjeldahl…
A:
Q: How can you prepare 0.5 N of 500 mL Hydrochloric Acid if you Sp. Gr. 1.184, percentage ratio 37% and…
A: Molecular weight of HCl = 1 +35.5 = 36.5 g/mol Specific gravity = 1.184 density of HCl solution =…
Q: The indicator of Moher regent is. KMN04. О Ксго4.
A: Mohr's method: Mohr's method is a direct method of titration. In this method potassium chromate…
Q: Under these conditions the acid acetylsalicylic (MW, 180) reacts according to: CH3COO-C6H4-COOH + 2…
A: To analyze an aspirin sample, 0.3470 g of crushed tablets are weighed and treated with 50.00 mL…
Q: 5. A0.104 M NAOH solution is used to titrate a solution containing 0.124 g KHP (MM = 204.2) and…
A:
Q: If this lab’s procedure were carried out using 28.06 mL each of 0.385 M K2CrO4 and 0.161 M Pb(NO3)2.…
A: Volume of K2CrO4 = 28.06 ml = Volume of Pb(NO3)2 Molarity of K2CrO4= 0.385 M Molarity of Pb(NO3)2 =…
Q: ind the concentration of Pb?* ions in a solution made by adding 5.00 g of lead (1) iodide to 500. mL…
A:
Q: 2.) 0.100 g of Na2CO3 is added to a 200.0 ml solution of 1.00 x 103 M Ca(NO3)2. Calculate Q. Q = x…
A:
Q: "Gravimetric form" in gravimetric assay of Barium-ions is: BaSO4 BaCO; BaO BaF2
A: Gravimetric analysis: gravimetric analysis i an analytical a method of quantitative chemical…
Q: A permanganate solution is prepared by dissolving 20.0123 g KMNO4 in 500 mL of distilled water and…
A: Given: The reaction is as follows, 5H2C2O4+2MnO4-+6H+⇌10CO2+2Mn2++8H2O Molarity of oxalic acid =…
Q: 1.00 grams of table salt was dissolved in 100 mL water and excess AgNO3 reagent was added and…
A: Given, The mass of table salt 1.00 grams dissolved in 100 mL water and excess AgNO3 reagent was…
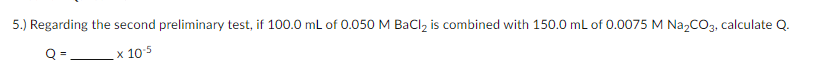
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 3 images

- If the titer during the standardization procedure of HCI is 32.50 ml by using 0.2115 grams of sodium carbonate as its standard, what is the percent error if the target concentration is 0.1250 N?For a titration to be effective, the reaction must be rapid and the yield of the reaction must essentially be 100%. Is Kc > 1, < 1, or ≈ 1 for a titration reaction?Calculate the volume in milliliters of 0.362 M KOH necessary to titrate 28.38 mL of 0.279 M diprotic sulfuric acid, H2SO4, to a phenolphthalein end-point. Report the answer to a precision of 2 decimal places.
- If 35.0 mL of a 0.0800 M HNO3, 25.0 mL of a 0.0500 M KSCN, and 55.0 mL of a 0.0500 M Fe(NO 3)3 are combined, what is the initial concentration of Fe3+ in the mixture?What mass of Ba(OH)2 is present in a sample if it is titrated to its equivalence point with 44.20 mL of 0.1000 N H2SO4? Note: Present complete solutions for the following problem. Express your final answers up to two (2) decimal places.list the apparatus involved in volumetric or titrimetric analysis
- In the experiment conducted to determine the amount of acetic acid in vinegar, 8.2 mL of NaOH standard solution adjusted to 0.098 M was spent besides the phenolphthaleley indicator. Accordingly, which of the following is the amount of acetic acid in the vinegar sample in grams? (Ma (CH3COOH): 60.05 g / mol)Sulfamic acid ( +H 3NSO 3 - , 97.094 g/mol) is a primary standard that can be used to standardize NaOH. +H 3NSO 3 - + OH - → H 2NSO 3 - + H 2O What is the molarity of a sodium hydroxide solution if 34.26 mL reacts with 0.3337 g of sulfamic acid?During a titration, the initial reading of a burette was recorded at 11.5 ml and the final reading is recorded at 34.2 ml. How much titrant was used in the analysis?
- Determine the required grams of a solid organic acid (KHP – 204.2 g/mol) that used as a primary standard to standardize and neutralize a base such as NaOH which is 1.005 mol/L. the volume you can assume0.5 g mixture of (0.1 N KCI and 4 points KBr) is required to 58.03 mL of 0.1 N AGNO3 to precipitate both bromide and chloride as AgCl and AgBr. Calculate the weight percentage for each of the AgCl and AgBr in a mixture if you know that the molecular weight of KCI = 74.5 and of KBr = 119 * %3DQ.5. During a titration, a fellow student weighs out 0.078 g of KHP. The final volume of ash water added to reach the endpoint was 9.20 mL. How many moles of KHP were used in this experiment?

