552 Cytoplasm АТР Malate Citrate + H* он CHAPTER 18 Oxidative Phosphorylation Inner mitochondrial membrane FIGURE 18.39 Mitochondrial transporters. Transporters (also called carriers) are transmembrane proteins that carry specific ions and charged metabolites across the inner mitochondrial membrane. Matrix Phosphate Malate Phosphate ADP АТР-ADP translocase Dicarboxylate Tricarboxylate carrier Phosphate carrier carrier
552 Cytoplasm АТР Malate Citrate + H* он CHAPTER 18 Oxidative Phosphorylation Inner mitochondrial membrane FIGURE 18.39 Mitochondrial transporters. Transporters (also called carriers) are transmembrane proteins that carry specific ions and charged metabolites across the inner mitochondrial membrane. Matrix Phosphate Malate Phosphate ADP АТР-ADP translocase Dicarboxylate Tricarboxylate carrier Phosphate carrier carrier
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter18: Glycolysis
Section: Chapter Questions
Problem 1P: Characterizing Glycolysis List the reactions of glycolysis that a. are energy consuming (under...
Related questions
Question
The rate of oxygen consumption by mitochondria increases markedly when ADP is added and then returns to its initial value when the added ADP has been converted into ATP (Figure 18.39). Why does the rate decrease?
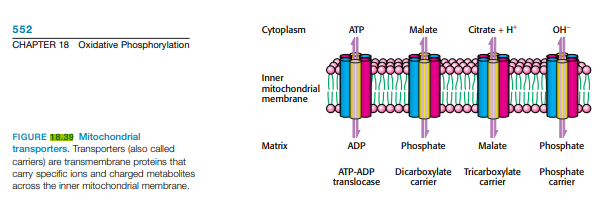
Transcribed Image Text:552
Cytoplasm
АТР
Malate
Citrate + H*
он
CHAPTER 18 Oxidative Phosphorylation
Inner
mitochondrial
membrane
FIGURE 18.39 Mitochondrial
transporters. Transporters (also called
carriers) are transmembrane proteins that
carry specific ions and charged metabolites
across the inner mitochondrial membrane.
Matrix
Phosphate
Malate
Phosphate
ADP
АТР-ADP
translocase
Dicarboxylate Tricarboxylate
carrier
Phosphate
carrier
carrier
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning