7. (1) What would you expect to be the order of reactivity from the following pyridine Derivatives towards electrophilic substitution (i.e. list from most reactive to the least reactive) and why? (ii) Can you also indicate which position would be favoured for such www substitution in each of those and why? O,N, `NO2
7. (1) What would you expect to be the order of reactivity from the following pyridine Derivatives towards electrophilic substitution (i.e. list from most reactive to the least reactive) and why? (ii) Can you also indicate which position would be favoured for such www substitution in each of those and why? O,N, `NO2
Chapter24: Amines And Heterocycles
Section24.9: Heterocyclic Amines
Problem 24P
Related questions
Question
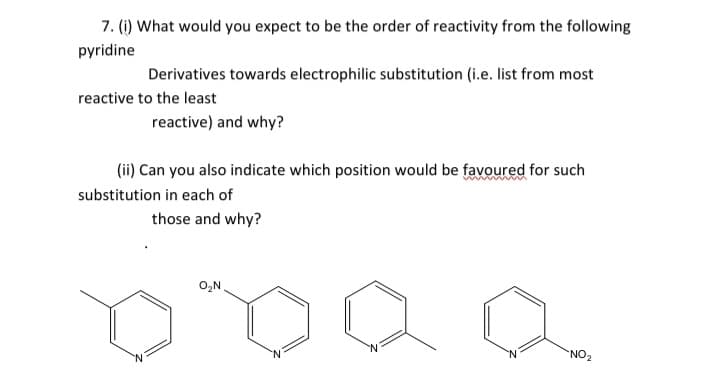
Transcribed Image Text:7. (1) What would you expect to be the order of reactivity from the following
pyridine
Derivatives towards electrophilic substitution (i.e. list from most
reactive to the least
reactive) and why?
(ii) Can you also indicate which position would be favoured for such
substitution in each of
those and why?
O,N
`NO2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

