Q: Phosphoric acid, H3PO4, is a polyprotic acid. What is the SECOND ionization reaction for phosphoric…
A: The question is asking for the second ionization reaction of phosphoric acid (H3PO4) in water.…
Q: Which of the following is a complete balanced equation for the reaction between a nonmetal oxide and…
A: The objective of the question is to identify the correct balanced chemical equation for the reaction…
Q: Question 29 Predict the FINAL (?) product for each of the following reaction or synthetic chain: A…
A: Thank you.
Q: Calculate the K, and AG° for the following reaction at 25 °C: 2 AgNO, (aq) + Zn(s) Zn (NO,), (aq) +…
A: Step 1 The given reaction is: 2AgNO3 aq + Zns ⇌ Zn(NO3)2 aq+2Ags Equilibrium constant (Kc) of a…
Q: In carbohydrate metabolism, the hexose monophosphate shunt is used for: Question 67 options:…
A: The objective of the question is to understand the role of the hexose monophosphate shunt (also…
Q: The first enzyme to rise in acute pancreatitis is ________________, and the enzyme which stays…
A: The objective of the question is to identify the enzymes that are affected during acute…
Q: None
A:
Q: The following data was obtained by measuring the volume of a trapped sample of gas at different…
A: Temperature T is plotted along x axis and volume V is plotted along y axis . The graph is shown in…
Q: DRAW THEM OUT 1. Predict the major organic product(s) for each of the following reactions. Be sure…
A: Step 1:
Q: Question 12 Choose the correct chemical structure for every single acronym used below: Bnl ? A B C D…
A: Option c: This option is correct
Q: Ethylene oxide, C2H4O, may be made by passing ethylene, C2H4, and air over a catalyst at 2500C. When…
A: The given reaction is the reaction between ethylene, water and CO to form ethylene glycol. The…
Q: Describe how you could achieve a higher degree of separation for the bands of food dyes on a TLC…
A: The objective of the question is to understand how to achieve a higher degree of separation for the…
Q: None
A: In order to balance an equation we must keep the number of atoms on both sides of the reation equal.…
Q: 2. Show how you would use the Wittig reaction to synthesize the following compounds. Start from the…
A: Step 1: Step 2: Step 3: Step 4:
Q: These disorders are associated with inborn errors of metabolism: Question 13 options:…
A: Option a: This option is incorrect because Types 1 and 2 diabetes mellitus can impact metabolism,…
Q: What is the pH of the solution that results when the following solutions are combined? 0.020 L of…
A: Thank you,Please rate my response.
Q: Question 25 Predict the FINAL (?) product for each of the following reaction or synthetic chain: 1.…
A: Step 1:
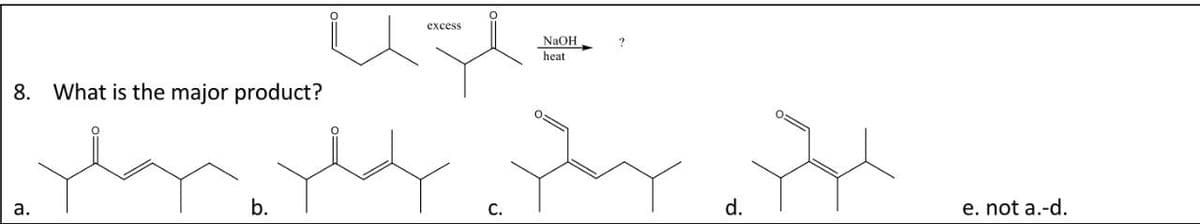
Q: help question 8
A:
Q: What is calibration and why is it essential in relation to food analysis? Provide examples.
A: What is calibration ? Calibration is comparing the measurements of an instrument or piece of…
Q: A weak base has a K₁ of 5.6x104, determine the pKь of the base. 3.25 6.36 7.64 10.75
A:
Q: A chemist was trying to synthesize compound C over the course of two steps, but after the first step…
A:
Q: Chemistry
A: Step 1: Step 2: Step 3: Step 4:
Q: Calculate the pH of solution of the following weak acid. Use Table 14.2 and Appendix H in your…
A: Step 1:Amphiprotic ion is a specie that can accept as well as donate the H+ ion. It means it can act…
Q: Balance the following equation in basic solution using the lowest possible integers and give the…
A: Ag+ + I- -----> Ag + IO3- Step 1:Determine oxidation number of each element:Ag+ has…
Q: Predict the product of the following reactions. Part E CH3NH2 H₂O Part F Part G Ph SH འས་ཀ་ པ་ NaOH…
A:
Q: provide A and B both answer with explanation
A: In case of any query feel free to ask.
Q: Which of the following is a complete balanced reaction for an Arrhenius base in water? NaOH (s) 2…
A: The objective of the question is to identify the correct balanced reaction for an Arrhenius base in…
Q: d.) Calculate the molality of the Nace and MgCl2.6H2O solutions.
A: Mass of MgCl2.6H2O =3.6 gMass of NaCl = 1.5 gVolume of water = 25 mLDensity of water = 1 g/mLMolar…
Q: Question 9 Please predict the products for each of the following reactions: A ABCD 1. Na 2. PrBr…
A:
Q: The mass of the salt is 5.006, volume of water is 100ml. Initial temperture is 20 and final…
A: Step 1: Using the specific heat capacity formula, the enthalpy change of the solution is calculated…
Q: What is the pH of a 0.40 M solution of NaOCI? (Ka for HOC1 = 3.50 x 10 -8) a) 2.48 b) 6.54 c) 7.45…
A: Use the Henderson-Hasselbalch equation to find the pH of the solution. The Henderson-Hasselbalch…
Q: At what temperature (in degrees Celsius) will xenon atoms have the same average speed that Br2…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: An unidentified covalent molecular compound contains only carbon, hydrogen, and oxygen. When 5.60 mg…
A: Step 1:Let formula of the compound is CxHyOzGeneral combustion reaction isCxHyOz + (x + y/4 - z/2)…
Q: Convert from rectangular to spherical coordinates. (Use symbolic notation and fractions where…
A: The objective of the question is to convert the given rectangular coordinates (6,-6√3,12√3) into…
Q: In the following acid-base equilibria of weak acids in water, label the acid (A), the base (B), the…
A: The objective of the question is to identify the acid, base, conjugate acid, and conjugate base in…
Q: Calculate creatinine clearance from the following results.urine creatinine=75 mg/dLurine volume=850…
A: The objective of this question is to calculate the creatinine clearance rate, which is a measure of…
Q: A solution contains 8.30×10-2 M ammonium bromide and 0.455 M ammonia.The pH of this solution is
A: The objective of this question is to calculate the pH of a solution containing ammonium bromide and…
Q: Which of the following is a correct molecular equation for the reaction of a metal and an acid? K…
A: The objective of the question is to identify the correct molecular equation for the reaction of a…
Q: If the total pressure inside the container is 400 mmHg , what is the partial pressure of each…
A: Thank you.
Q: 2. Treatment of the ketone shown below with ZnBH4 in diethyl ether provides a 94:6 mixture of B and…
A: Given that ZnBH4 reduces the ketone to give a 94:6 mixture of B and C, we can infer that B is the…
Q: show the mechanism please
A: The reaction is the Friedel-Craft acylation reaction. First, acyl chloride reacts with AlCl3 and…
Q: Please fast expert answer solutions and dwor computer figure
A: In order to determine an aromatic compound, it must be: cyclicplanarComplete delocalization of π…
Q: ton Draw the mechanism for each reaction ་ 1. он MAJ 2. Ugot 1.-04 2. H₂0 HO Nu HO OH HO 244 S…
A:
Q: How many grams of solid ammonium bromide should be added to 1.50 L of a 0.0879 M ammonia solution to…
A: The objective of this question is to determine the amount of solid ammonium bromide that needs to be…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1. CH2=CHLİ 2. H3O+ Drawing H
A: The reaction seen in the image is a Grignard reaction followed by an acid-base reaction. Grignard…
Q: Consider three hypothetical solids:AX,AX2 and AX3 (each X forms X). Each of these solids has the…
A: Solution : Solubility is defined as the maximum amount of solute dissolved in an aqueous…
Q: None
A: Let's delve into the intricacies of the chemical structures depicted in the image you…
Q: An air-gap parallel plate capacitor whose plates have an area of A=0,38m² are separated by a…
A: Step 1:Step 2:
Q: Which of the following aqueous solutions are good buffer systems? (Select all that apply.) 0.20 M…
A: The objective of this question is to identify which of the given aqueous solutions are good buffer…
Q: For each of the substituted benzene molecules below, determine the inductive and resonance effects…
A: Step 1:Step 2:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images




