Q: Name three other products applied in industry obtained from phenol and its uses.
A: 1) asprin is applied in industry phenol Is used in the synthesis of aspirin Phenol is first allowed ...
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the tem...
A:
Q: D-Glucose, cyclic (D-Glucopyranose)
A: A hemiacetal is a carbon connected to two oxygen atoms, where one oxygen is an alcohol (OH) and the ...
Q: Reaction 3: An aqueous solution of sodium hydroxide reacts with an aqueous solution of hydrogen chlo...
A: This is a neutralisation reaction between strong acid HCl and strong base NaOH. Neutralisation react...
Q: 4) (a) For the following structure, using curved arOWs draw all resonance contributors (only those w...
A: Resonance is a phenomenon in which shifting of electron takes place from one atom to another atom to...
Q: What volume, in L, would 10.0 moles of nitrous oxide occupy at 65.1 °C with a pressure of 6.40 atm?
A:
Q: Calculate the pH of a solution saturated in calcite and in equilibrium with atmospheric CO2. The tem...
A: The Ksp expression for the solubility of CaCO3 can be expressed as: Ksp=Ca2+CO32- Ca2+=Ksp/CO32-………(...
Q: help me draw 13C NMR spectrum of the image below
A: The 13C NMR Spectra of the compound are given below.
Q: Consider the reaction: 4 PH3 (g) → P4 (g) + 6 H2 (8). If, in a certain experiment over a specific ti...
A: reaction given in question, 4PH3(g) → P4(g)+ 6H2(G)HERE, data is given 0.036 mole of PH3 , Volum...
Q: 26. The lipid shown below is a(n) (a) Monoglyceride (b) Phospholipid (c) Steroid (d) Diglyceride (e)...
A:
Q: 8. Balance the following redox reaction using the half-reaction method. Cro,? + SO,?- Cr(OH); + SO? ...
A:
Q: Question 16 PCI5 (s) + H20(1)- POCI (1) + 2HCI(ag) When 53.22 g of phosphorus pentachloride reacts w...
A:
Q: SiO2 + C SiC + Cu The unbalanced equation above shows carbon and silicon dioxide reacting to form si...
A: Given mass of each reactant = 22 g standard values molar mass of SiO2 = 60 g/mol molar mass of C = 1...
Q: Given the following unbalanced equation, select the correct net ionic equation. CaCO3(s) + HCI(aq) -...
A:
Q: Galactosamine is a major component of cartilage. Using organic chemistry describe using diagrams the...
A: Answer - Galactosamine - Galactosamine is a hexosamine derived from galactose with the molecular for...
Q: The following indicator exhibits the following colors in its acid and conjugate base forms. HIn(aq) ...
A: The solution is as follows:
Q: If a single C-C bond has a bond energy of 346 kJ/mol, could a single photon in the visible range wit...
A: We have to check the given length of light dissociate the C-C single bond or not
Q: Aqueous hydrochloric acid (HCI) will react with solid sodium hydroxide (NAOH) to produce aqueous sod...
A:
Q: tion produced Hydrogen gas with a final Temperature, Pressure and Volume of 35 °C, 775 mmHg, and 2.3...
A:
Q: Questions: 1. Write a balanced equation for the titration of NaOH with: a. Citric acid (acid in cand...
A: “Since you have asked multiple question, we will solve the first question for you. If you want any s...
Q: 6. 2.3x 102 formula units FeCl, to grams
A:
Q: Q7. A gas mixture that has the following composition in percent by volume: 10.2% CO2, 21.0% CO, 18.5...
A: Let the total composition of the mixture be 100 mL. The volume of CO2, CO, H2, CH4 and N2 can now be...
Q: What mass of glucose (CH;2Og) must dissolve in 1045 g of ethanol to make a 0.66 m solution? Answer:
A:
Q: For the analysis below, find one step within design of experiment, interpretation and calculation of...
A: Answer : Based on the unit of molar absorptivity, unit is L/(cm*mole) in another way we can write th...
Q: Consider the element Work (Wr) with an atomic number of 33 and an atomic mass of 81.03 g/mol. The f...
A:
Q: (4). A buret in the “The soluble chloride determination by gravimetry” lab is employed as ____ ...
A:
Q: CH3 ((i CH3-C=CH2CI Gilman rcagent ? BF, 1) LDA →? 2) CH31 -78 °C 1) LIAIH4 → ? 2) H30® Cliv) Ph-COO...
A:
Q: Which of these compounds is most soluble in water and why? (A) CH4; it forms hydrogen bonding with w...
A: CH4 do not form any hydrogen bonding and it is also a non polar compound so it is insoluble in wate...
Q: of 1.00 x10-4 M Ag* and 5.00 x10-5 M Cro42, Ag2CrO4 precipita= of 1.0 x10-4 M Ag* and 1.0 x10-4 M I0...
A: As we know, Ksp is less than Qsp , precipitate will form. But in case of Ag2CrO4 and Ag(IO3) both ...
Q: Choose the process that will not disturb the following equilibrium. Co(g) + H20(g)=CO2(g) + H2(g)
A: Temperatures, pressure and volume effect the equilibrium. According to Le-Chatlier's principle,when ...
Q: Reaction 2: Solid sodium hydroxide reacts with aqueous hydrogen chloride solution to form water and ...
A: Three exothermic reactions involved in this neutralisation reaction are:- 1) dissolution of NaOH in...
Q: Which of the following is TRUE regarding catabolic reactions: a. They require an input of energy...
A: The correct option is:
Q: 32. The two principal polysaccharides of starch are and
A: Note : According to our guidelines we are supposed to answer only one question. Kindly repost other...
Q: Activity Series Practice Predict whether or not each of the following reactions will occur. Write th...
A: Explanation : Chemical Reaction : The process that when one or more compound are change to their p...
Q: PART A - ELECTRON CONFIGURATIONS AND ORBITAL NOTATIONS Use the patterns within the periodic table to...
A:
Q: A 69.5 g piece of iron at 525°C is put into 627 grams of water at 15.0°C. What is the final temperat...
A: Given Mass of iron = 69.5 g Mass of water = 627 g Initial temperature of iron = 525°C Initial tempe...
Q: A 10.00 mL aliquot of a 0.0100 M Ca solution is titrated with 25.00 mL of EDTA 0.00500 M at a pH of ...
A: Answer: b) there is an excess of Y4- Full Solution given in step 2. please check
Q: The calibration curve was generated using known concentrations of five solutions of a newly discover...
A: The amount of a substance dissolved in a specified volume of the solvent produces a solution of a pa...
Q: 5. Calculate the solubility (g/L) of Ag3PO4 (Mw = 419 g/mol). K«p = 1.8 x 10-18
A:
Q: Data of Candy: Mass of candy: 6.1115g Concentration of NaOH (M): 0.09639 Volume of NaOH solution use...
A:
Q: Experimenters started with excess copper(II) chloride, but a limited amount (855 mg) of potassium io...
A: Using the sticheometric coefficient to calculate theoretical yield. We start with limiting reagent....
Q: Please refer to the monosaccharides below when answering questions (a) – (1). CHO CHO СНО СНО CHO Но...
A: Enantiomers are optical isomers that are non-superimposable mirror images of each other. Epimers are...
Q: Consider the titration of 150.0 mL of a 0.225 M solution of (C2H5)NH with 0.45 M HCl(aq). Kb= 4.3 ...
A: (C2H5)2NH is a weak base with Kb = 4.3*10-4. HCl is a strong acid. Hence (C2H5)2NH and HCl will unde...
Q: How do chemists predict the shapes of molecules? A. Due to the attraction between electrons, valenc...
A: Statement1: Due to the attraction between electrons, valence electrons will be arranged as close to ...
Q: Benzene, C6H6, consists of a six-member ring of sp2 hybridized carbon atoms. Each carbon atom has on...
A: Sox carbon in benzene have 6 unhybridized p orbital these atomic orbital combine to form Six molecul...
Q: 29. Which of the lipids in Figure LP 1 is likely to have the highest melting point? II III Figure LP...
A:
Q: In the reaction between hydrogen gas and chlorine gas to give hydrogen chloride gas spontaneous, und...
A:
Q: ii. Draw the arrows for STEPS 3-4. In STEP 3, the D-glucosyloxy anion reacts with water. In STEP 4, ...
A:
Q: The combustion of 1.00mol of glucose (C6H12O6) liberates 2820 kJ of heat. If 1.64g of glucose is bur...
A:
Q: 25.0 mL solution of HNO₃ is neutralized with 36.8 mL of 0.250 M Ba(OH)₂. What is the concentration o...
A:
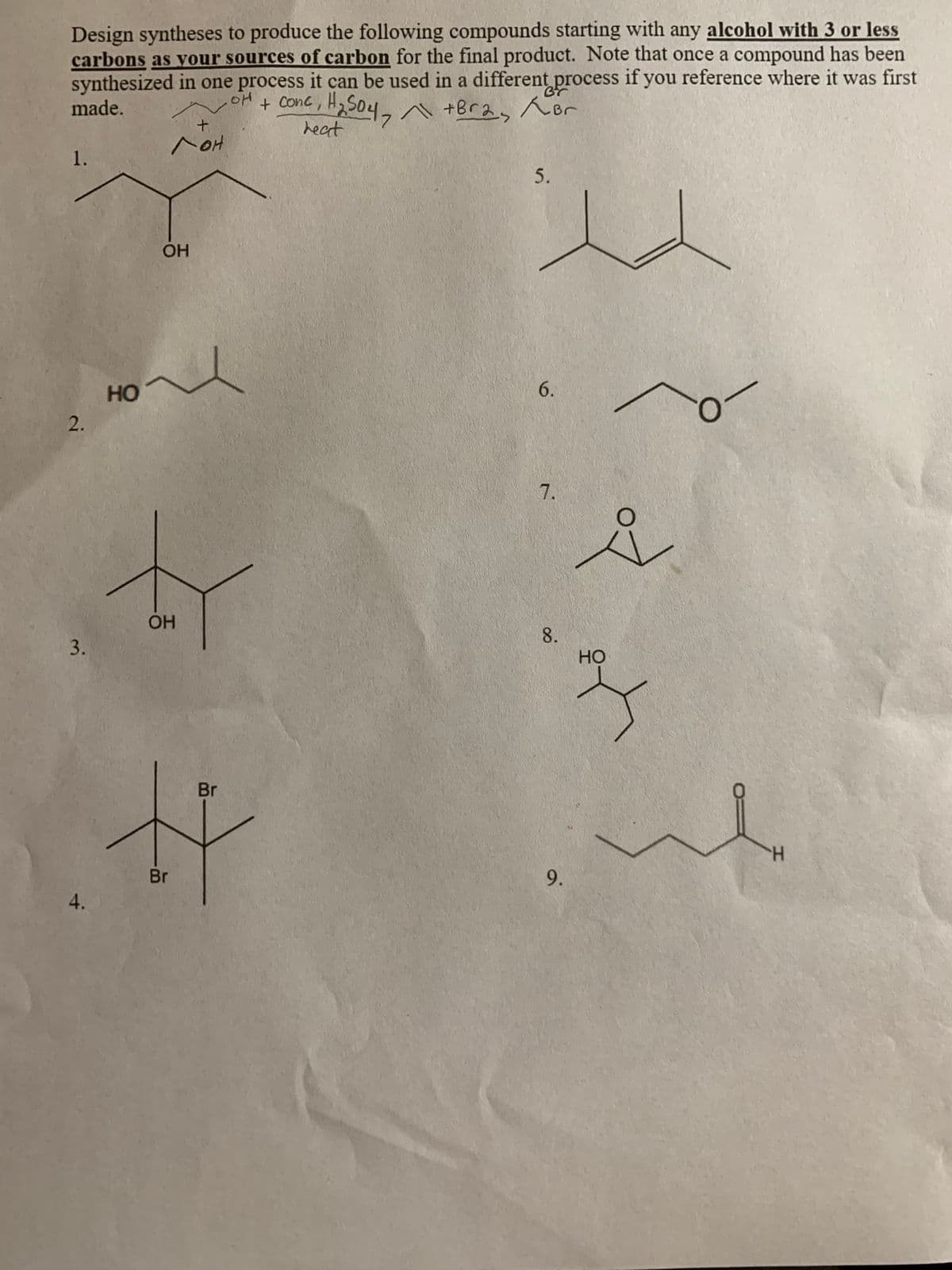
Number 9 please
Step by step
Solved in 2 steps with 2 images

- Knowing the properties of organic molecules, students will now design experimental set-ups that will maximize the yield and purity of the target organic molecule. The students must prove that their choice of materials is available for purchase by providing a CAS number or a catalogue from a supplier (example: Sigma Aldrich). Also, included in this part is the discussion of the Chemistry behind the synthesis as well as safe laboratory techniques and practices to synthetic design.Which of the following statements is not correct? Oxymercuration-demercuration for alcohol synthesis is complicated by carbocation rearrangement. Oxymercuration-demercuration for alcohol synthesis follows Markonikov's Rule. Sodium borohydride is used in the last step of oxymercuration-demercuration to oxidize the hydroxyalkyl mercury compound. All of the above. Both the 1st and the 3rd statements.Show a possible stepwise synthesis of 2, 3–dichloropentane starting with ethanol and propanol as the only sources of carbon atoms and any other inorganic reagents, solvents and laboratory equipment. Name the type of reaction that is occurring in each step of the synthesis.
- To practice working through the early parts of a multistep synthesis, devise syntheses of pentan-3-one from alcohols containing no more than three carbon atoms1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the aboveHi there, can someone please help me solve this problem? Please make sure to clarify all steps taken to go about solving for the products and explain various terms and rules that should be known or correlate with the question. I'm using these questions to study for our final exam and need some extra clarification on rules and steps. Thank you very much in advance!
- What is the strongest IMF in pure 2-octanone? I know it is a ketone and has some polarity due to this, but the carbon chain so long, I wonder if london dispersion forces are stronger?Which of these compounds is most soluble in water at pH 7.0? (Note that the non-ionized forms are shown, and remember that ionization will impact solubility!) a CH3-CH2-HCO-O-CH3 (an ester) b CH3-CH2-CH2-CH2-CHO c CH3-CH2-CH2-CH2-CH3 d CH3-CH2-CH2-CH2-NH2Based on the results of the KMnO4 and Lucas tests on the representative alcohols (alcohol, phenol, ether), give a generalization on how the different types of alcohols (1°, 2°, and 3°) can be differentiated from each other. Permanganate Test Result: Positive: 1-butanol, 2-butanol, phenol Negative: tert-butyl alcohol, disopropyl ether Lucas Test Positive: 2-butanol & tert-butyl alcohol Negative: 1-butanol
- Which of these compounds is most soluble in water at pH 7.0? (Note that the non-ionized forms are shown, and remember that ionization will impact solubility!) a CH3-CH2-CH2-CH2-CH2OH b CH3-CH2-CH2-CH2-COOH c CH3-CH2-CH2-CH2-CH3 d CH3-CH2-CH2-CH2-CHOConsidering the elimination reactions, followed by the deprotonation of water, evaluate the following statements.i- Three equivalents of the base are needed to obtain the terminal alkyne.ii- The alkyl dialect is geminal.iii- The speed of obtaining the alkynes depends only on the concentration of the base.iv- Obtaining alkyne from alkyl dialects is accompanied by two successive elimination reactions. In the first elimination reaction it is possible to use NaOH. However, in the second, it requires a very strong base, such as liquid ammonia. It is correct to say that: i and ii, just i and iii, just i and iv, just i, ii and iv, only all except i. nahTwo Compounds: 3-methylbutyric acid and 4,5-dimethyldecane Suppose you took your two compounds, dissolved them in tertbutyl methyl ether and then added them to a separatory funnel. Now suppose you add in aqueous sodium bicarbonate. _____________________ will be dissolved in the terbutyl methyl ether. ________________________ will be dissolved in the aqueous sodium bicarbonate layer. Draw the EXACT chemical structure that will exist in each of the layers after shaking with sodium bicarbonate. (Is it neutral or charged?)

