a) AADase-catalyzed decarboxylation of acetoacetate proceeds through an enzymatic Schiff base. Draw out the mechanism for this conversion of acetoacetate to acetone, showing enzymatic lysine (Lys, or K) involvement.
a) AADase-catalyzed decarboxylation of acetoacetate proceeds through an enzymatic Schiff base. Draw out the mechanism for this conversion of acetoacetate to acetone, showing enzymatic lysine (Lys, or K) involvement.
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter19: The Tricarboxylic Acid Cycle
Section: Chapter Questions
Problem 18P
Related questions
Question
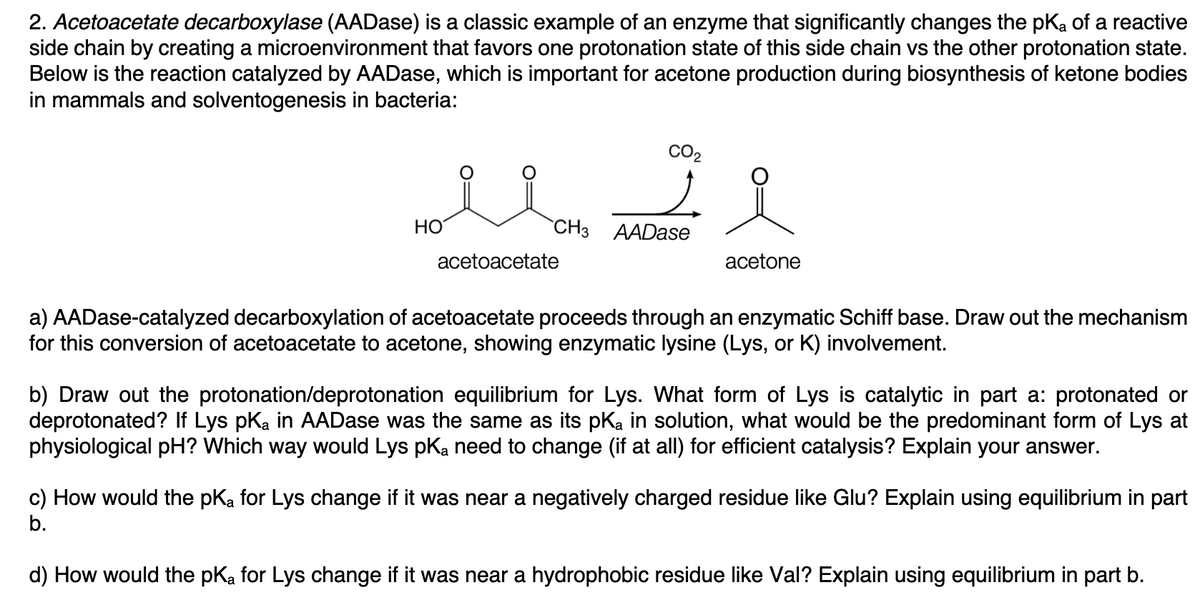
Transcribed Image Text:2. Acetoacetate decarboxylase (AADase) is a classic example of an enzyme that significantly changes the pKa of a reactive
side chain by creating a microenvironment that favors one protonation state of this side chain vs the other protonation state.
Below is the reaction catalyzed by AADase, which is important for acetone production during biosynthesis of ketone bodies
in mammals and solventogenesis in bacteria:
CO2
HO
"CHз АADase
acetoacetate
acetone
a) AADase-catalyzed decarboxylation of acetoacetate proceeds through an enzymatic Schiff base. Draw out the mechanism
for this conversion of acetoacetate to acetone, showing enzymatic lysine (Lys, or K) involvement.
b) Draw out the protonation/deprotonation equilibrium for Lys. What form of Lys is catalytic in part a: protonated or
deprotonated? If Lys pKa in AADase was the same as its pKa in solution, what would be the predominant form of Lys at
physiological pH? Which way would Lys pKa need to change (if at all) for efficient catalysis? Explain your answer.
c) How would the pka for Lys change if it was near a negatively charged residue like Glu? Explain using equilibrium in part
b.
d) How would the pKa for Lys change if it was near a hydrophobic residue like Val? Explain using equilibrium in part b.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning