A biochemist carefully measures the molarity of glycerol in 84.0 mL of photobacterium cell growth medium to be 38.2 uM. Unfortunately, a careless graduate student forgets to cover the container of growth medium and a substantial amount of the solvent evaporates. The volume of the cell growth medium falls to 9.21 mL. Calculate the new molarity of glycerol in the photobacterium cell growth medium. Round each of your answers to 3 significant digits. db OAM Submit Assignment Continue © 2021 McGraw Hill LLC. AlI Rights Reserved. Terms of Use Privacy Center Accessibility MacBook Air 44 DII DD 20 F10 FB F6 F2 F3 F4 F1 & @ 23 %24 7 9. * CO ( CO
A biochemist carefully measures the molarity of glycerol in 84.0 mL of photobacterium cell growth medium to be 38.2 uM. Unfortunately, a careless graduate student forgets to cover the container of growth medium and a substantial amount of the solvent evaporates. The volume of the cell growth medium falls to 9.21 mL. Calculate the new molarity of glycerol in the photobacterium cell growth medium. Round each of your answers to 3 significant digits. db OAM Submit Assignment Continue © 2021 McGraw Hill LLC. AlI Rights Reserved. Terms of Use Privacy Center Accessibility MacBook Air 44 DII DD 20 F10 FB F6 F2 F3 F4 F1 & @ 23 %24 7 9. * CO ( CO
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter1: Matter And Measurements
Section: Chapter Questions
Problem 53QAP: Magnesium sulfate (MgSO4) has a solubility of 38.9 g/ 100 g H2O at 30C. A solution is prepared by...
Related questions
Question
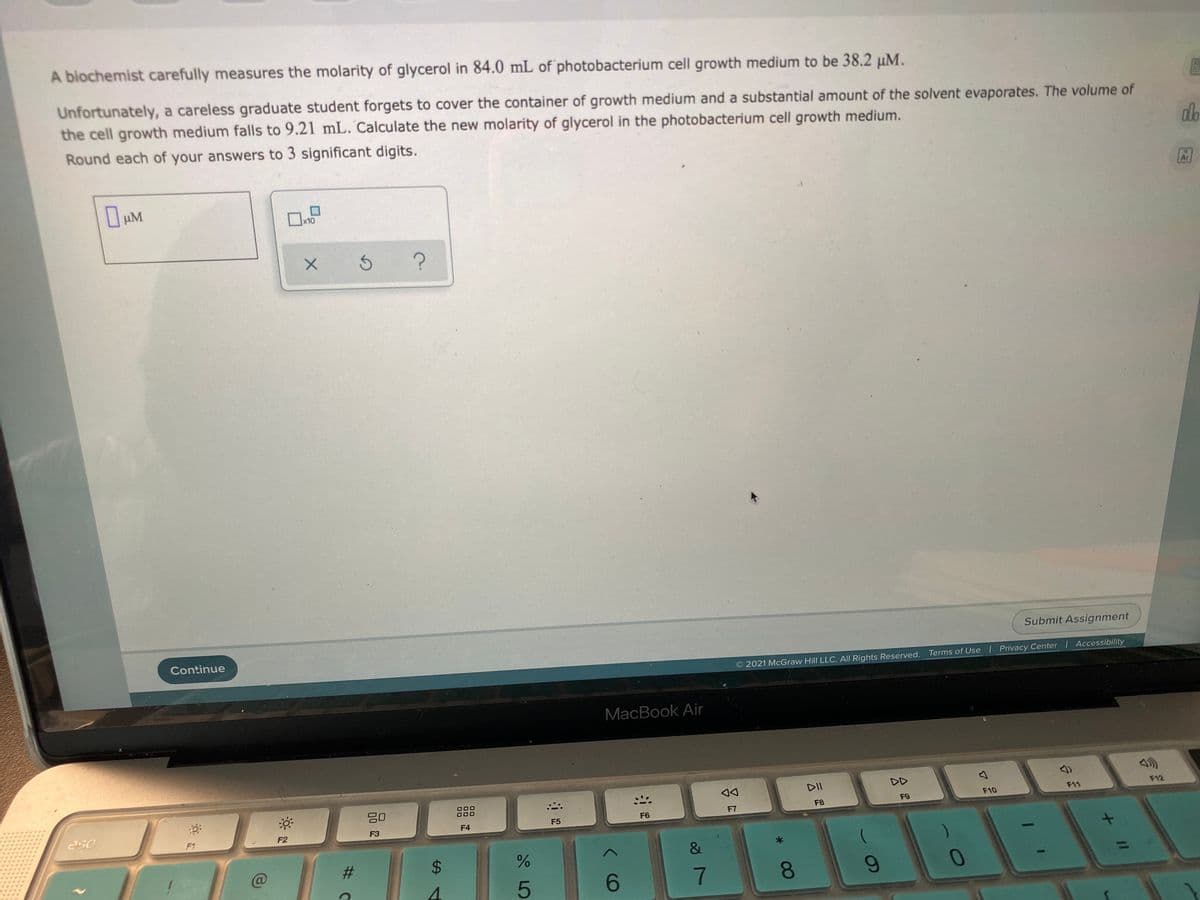
Transcribed Image Text:A biochemist carefully measures the molarity of glycerol in 84.0 mL of photobacterium cell growth medium to be 38.2 µM.
Unfortunately, a careless graduate student forgets to cover the container of growth medium and a substantial amount of the solvent evaporates. The volume of
the cell growth medium falls to 9.21 mL. Calculate the new molarity of glycerol in the photobacterium cell growth medium.
Round each of your answers to 3 significant digits.
do
x10
Continue
Submit Assignment
O 2021 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Center Accessibility
MacBook Air
DII
DD
000
F10
F11
F12
esc
F8
F9
F4
F5
F6
F7
F1
F2
F3
@
$4
6
7.
8.
%23
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning