(a) C4H,CIO IR: 1810 cm-1 TMS 10 8. O ppm Desplazamiento químico (8) (b) C5H;NO2 IR: 2250, 1735 cm-1 TMS 10 9. 8. 7 5 3. 2 1 O ppm Desplazamiento químico (8) Intensidad Intensidad
(a) C4H,CIO IR: 1810 cm-1 TMS 10 8. O ppm Desplazamiento químico (8) (b) C5H;NO2 IR: 2250, 1735 cm-1 TMS 10 9. 8. 7 5 3. 2 1 O ppm Desplazamiento químico (8) Intensidad Intensidad
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter32: Radiochemical Methods
Section: Chapter Questions
Problem 32.12QAP
Related questions
Question
Kindly assign structures for compounds with the following spectra.
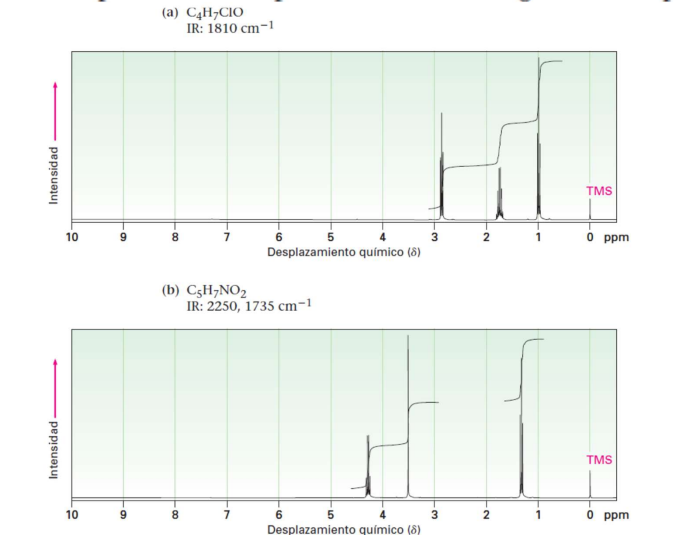
Transcribed Image Text:(a) C4H¬CIO
IR: 1810 cm-1
TMS
10
9.
8.
6
3
O ppm
Desplazamiento químico (8)
(b) C5H¬NO2
IR: 2250, 1735 cm-1
TMS
10
8.
7
6
1
O ppm
Desplazamiento químico (8)
Intensidad
Intensidad
3.
2-
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
