A chemical engineer must calculate the maximum safe operating temperature of a high-pressure gas reaction vessel, The vessel is a stainless-steel cylinder that measures 22.0 cm wide and 26.4 cm high. The maximum safe pressure inside the vessel has been measured to be 6.50 MPa. For a certain reaction the vessel may contain up to 0.502 kg of carbon dioxide gas. Calculate the maximum safe operating temperature the engineer should recommend for this reaction. Write your answer in degrees Celsius. Round your answer to 3 significant digits. temperature: C
A chemical engineer must calculate the maximum safe operating temperature of a high-pressure gas reaction vessel, The vessel is a stainless-steel cylinder that measures 22.0 cm wide and 26.4 cm high. The maximum safe pressure inside the vessel has been measured to be 6.50 MPa. For a certain reaction the vessel may contain up to 0.502 kg of carbon dioxide gas. Calculate the maximum safe operating temperature the engineer should recommend for this reaction. Write your answer in degrees Celsius. Round your answer to 3 significant digits. temperature: C
Chapter5: Gases
Section: Chapter Questions
Problem 74E: Urea (H2NCONH2) is used extensively as a nitrogen source in fertilizers. It is produced commercially...
Related questions
Question
100%
Please reply with the right answer. Thank you
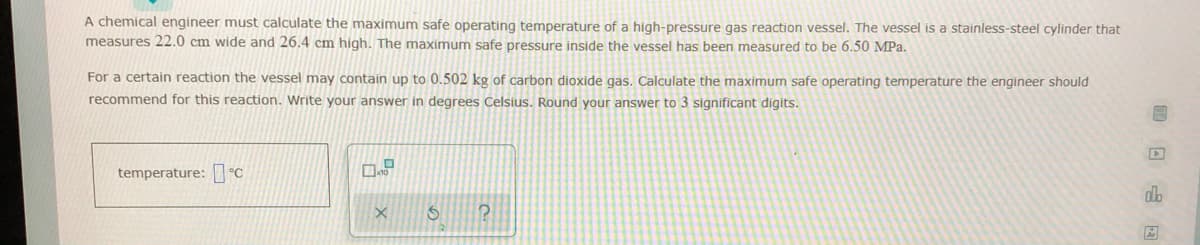
Transcribed Image Text:A chemical engineer must calculate the maximum safe operating temperature of a high-pressure gas reaction vessel. The vessel is a stainless-steel cylinder that
measures 22.0 cm wide and 26.4 cm high. The maximum safe pressure inside the vessel has been measured to be 6.50 MPa.
For a certain reaction the vessel may contain up to 0.502 kg of carbon dioxide gas. Calculate the maximum safe operating temperature the engineer should
recommend for this reaction. Write your answer in degrees Celsius. Round your answer to 3 significant digits.
temperature: °C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning