A compound consists of X and Y atoms. Its solid has the cubic unit structure shown below. Based on the information given here, which of the following statements is correct?
A compound consists of X and Y atoms. Its solid has the cubic unit structure shown below. Based on the information given here, which of the following statements is correct?
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter13: Introduction To Symmetry In Quantum Mechanics
Section: Chapter Questions
Problem 13.8E
Related questions
Question
A compound consists of X and Y atoms. Its solid has the cubic unit structure shown below. Based on the information given here, which of the following statements is correct?
1. The empirical formula of this compound is X2Y3; X-atoms form the BCC (body center cubic) unit structure.
2. The empirical formula of this compound is X9Y6; X-atoms form the FCC (face center cubic) unit structure.
3. The empirical formula of this compound is X3Y2; X-atoms form the BCC (body center cubic) unit structure.
4. The empirical formula of this compound is X2Y3; X-atoms form the FCC (face center cubic) unit structure.
5. The empirical formula of this compound is X9Y6; X-atoms form the BCC (body center cubic) unit structure.
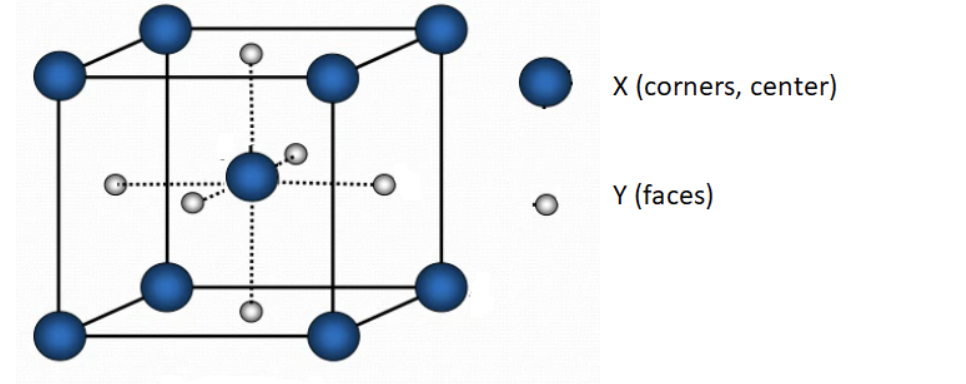
Transcribed Image Text:X (corners, center)
Y (faces)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning
