A compound containing only carbon, hydrogen, and oxygen is analyzed using combustion analysis. When 0.501 g of the compound is burned, 0.711 g of carbon dioxide and 0.436 g of water are collected. Given that there are 0.0161 moles of carbon and 0.0484 moles of hydrogen produced in the reaction, calculate the mass of oxygen in the original compound. You will need to subtract the mass of the carbon that ended up in the carbon dioxide and the mass of the hydrogen that ended up in the water from the mass of the original compound.
A compound containing only carbon, hydrogen, and oxygen is analyzed using combustion analysis. When 0.501 g of the compound is burned, 0.711 g of carbon dioxide and 0.436 g of water are collected. Given that there are 0.0161 moles of carbon and 0.0484 moles of hydrogen produced in the reaction, calculate the mass of oxygen in the original compound. You will need to subtract the mass of the carbon that ended up in the carbon dioxide and the mass of the hydrogen that ended up in the water from the mass of the original compound.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter2: Chemical Formulas, Equations, And Reaction Yields
Section: Chapter Questions
Problem 13P
Related questions
Question
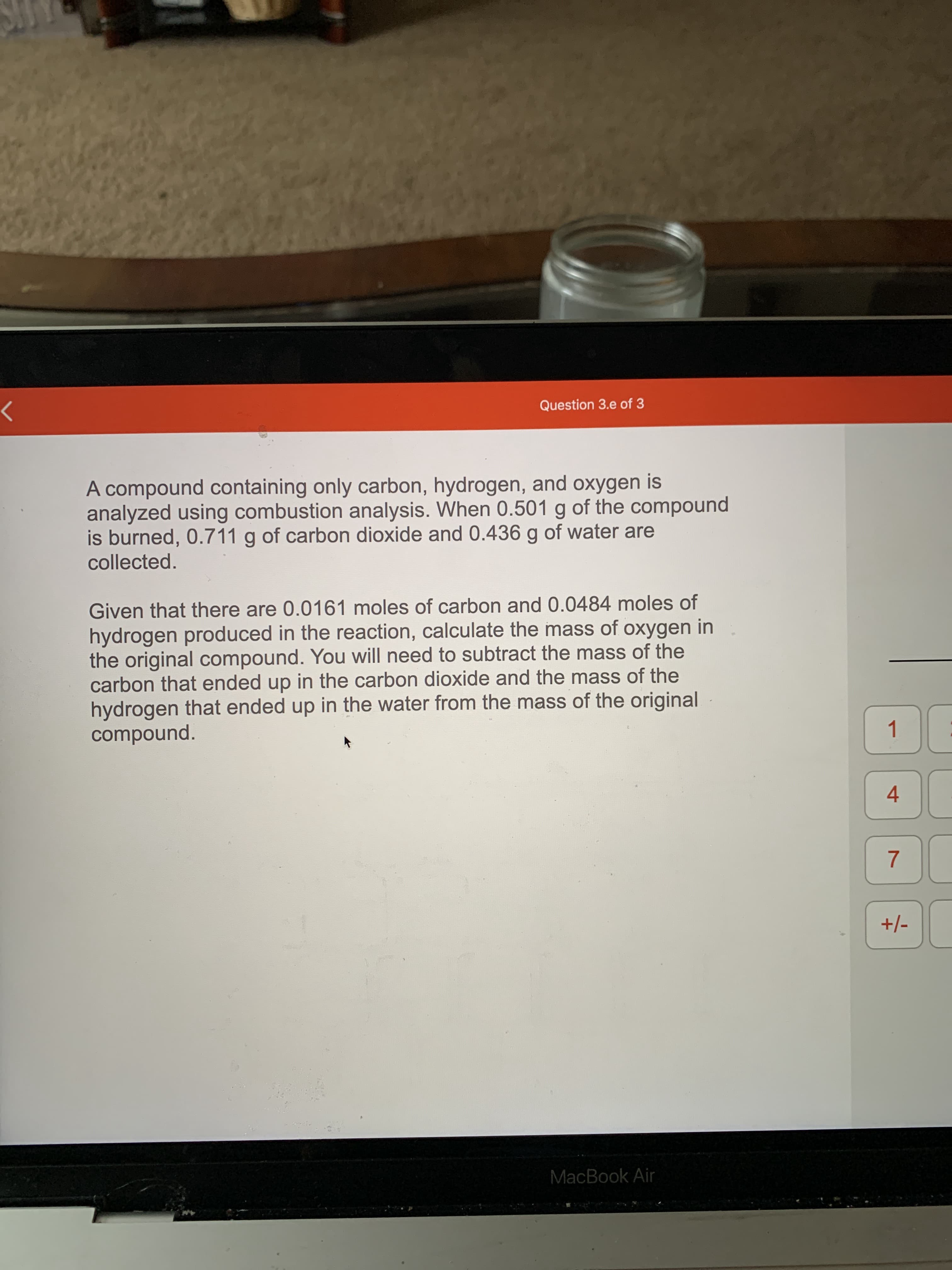
Transcribed Image Text:A compound containing only carbon, hydrogen, and oxygen is
analyzed using combustion analysis. When 0.501 g of the compound
is burned, 0.711 g of carbon dioxide and 0.436 g of water are
collected.
Given that there are 0.0161 moles of carbon and 0.0484 moles of
hydrogen produced in the reaction, calculate the mass of oxygen in
the original compound. You will need to subtract the mass of the
carbon that ended up in the carbon dioxide and the mass of the
hydrogen that ended up in the water from the mass of the original
compound.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
